Chapter: Biochemistry: Proteins
Amino acids
Amino acids
Amino acids are the simplest units of a protein
molecule and they form the building blocks of protein structure. The general
formula of an amino acid can be written as,
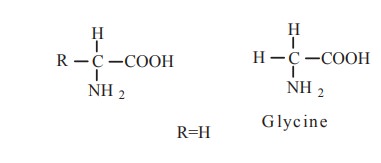
An amino acid is an amino carboxylic acid. R is
the side chain or residue and it represents the group other than -NH2
and -COOH. It may be a hydrogen atom (H) or a methyl group (-CH3) or
an aliphatic group or an aromatic group or a heterocyclic group. The amino
acids are classified based on the nature of R groups.
D and L amino acids
Based on the position of amino group on the
asymmetric carbon atom, amino acids exist in two types. They are D and L amino
acids
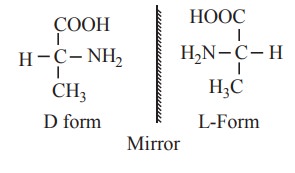
The amino acid having the NH2 group
on the right is called D-amino acid. The amino acid having the NH2
group on the left is called L-amino acid. These isomers are the mirror images
of each other.
All amino acids are α amino acids because the
NH2 group is attached to the a carbon atom which is next to the COOH
group. Examination of the structure of an amino acid except glycine, reveals
that the α carbon atom has four different groups attached to it, thus making it
asymmetric. Because of the presence of asymmetric carbon atom, amino acids
exist in two optically active forms, dextrorotatory and levorotatory.
Dextrorotatory, compounds rotate plane
polarised light in the clockwise direction. Levorotatory, compounds rotate
plane polarised light in the anti clockwise direction. The direction of optical
rotation of an amino acids indicated by the symbol + and - (+ indicates dextro
and - indicate levo).
It has been found that L-amino acids are more
common than D forms and most of the naturally occuring amino acids are L-amino
acids. Therefore L-amino acids are called natural aminoacids. Since the L amino
acids are more common, the letter “L” is usually omitted, while representing
L-amino acids.
Amino acids are widely distributed in plants
and animals.
Related Topics