Chapter: Biotechnology Applying the Genetic Revolution: Gene Therapy
Adeno Associated Virus
ADENO-ASSOCIATED
VIRUS
Because of the problems with
using adenovirus discussed above, other DNA viruses have been considered as
vectors. Although none are yet widely used, the adeno-associated virus (AAV)
shows considerable promise. AAV is a defective or “satellite” virus that
depends on adenovirus (or some
herpes viruses) to supply some necessary functions. Consequently, it is usually
found in cells that are infected with adenovirus. Unlike adenovirus, AAV seems
to be entirely harmless.
The benefits of using AAV are
as follows:
(a) It does not stimulate
inflammation in the host.
(b) It does not provoke
antibody formation and can therefore be used for multiple treatments.
(c) It infects a wide range of animals, as
long as an appropriate helper virus is also present. It can therefore be
cultured in many types of animal cells, including those from mice or monkeys.
(d) It can enter nondividing cells of many
different tissues, unlike adenovirus.
(e) The unusually small size of the virus particle
allows it to penetrate many tissues of the body effectively.
(f) AAV integrates its DNA into a single site
in the genome of animal cells (the AAVS1 site on chromosome 19 in humans). This
allows the therapeutic gene to be
permanently integrated.
One drawback is that the AAV
genome is small (4681 nucleotides of single-stranded DNA) and the virus can
carry only a relatively short segment of DNA. (AAV is unusual in packaging both
plus and minus strands into virus particles. Although each virus particle
contains only one ssDNA molecule, a virus preparation contains a mixture of
particles, half with plus and half with minus strands.) On entering a host
cell, the DNA is converted to the double-stranded replicative form, or RF,
which is used for both replication and transcription. In the absence of helper
virus, AAV integrates into the host chromosome and becomes latent.
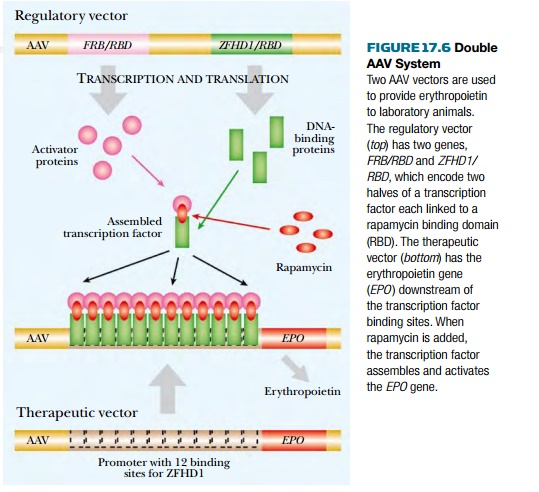
Genes that are permanently
integrated need to be carefully regulated. This may be tackled by using two AAV
vectors. Mice and monkeys have been experimentally treated with a double AAV
system that provides erythropoietin, a protein required for development of red
blood cells. One AAV vector carries the gene for erythropoietin with a promoter
that must be activated by a transcription factor. The second AAV vector carries
an artificial regulatory system (Fig. 17.6). This consists of two genes
encoding hybrid proteins, each with one domain of the transcription factor. The
other domain binds rapamycin (used as an immunosuppressant). In the presence of
rapamycin, the two hybrid proteins associate via their rapamycin binding
domains to form a functional transcription factor. This activates
erythropoietin expression.
After delivery of the two
vectors to mice, there was no production of erythropoietin. But when the
animals were injected with rapamycin, the transcription factor was assembled
and the erythropoietin gene was activated. The levels of erythropoietin
increased up to 100-fold and the number of red blood cells rose. Even after
several months, injection of rapamycin triggered a sharp rise in erythropoietin
levels. Preliminary studies are now being performed in cystic fibrosis patients
with AAV vectors carrying the CFTR gene.
Related Topics