Air | Chapter 11 | 8th Science - Acid rain | 8th Science : Chapter 11 : Air
Chapter: 8th Science : Chapter 11 : Air
Acid rain
Acid rain
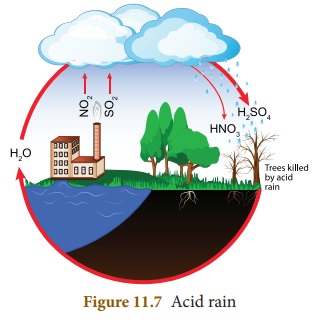
Rain water is actually the purest
form of water. However, pollutants such as oxides of nitrogen (N2O,
NO2) and sulphur (SO2, SO3) in the air
released by factories, burning fossil fuels, eruption of volcanoes etc. ,
dissolve in rain water and form nitric acid and sulphuric acid which adds up to
the acidity of rain water. Hence, it results in acid rain.
Acid rain has pH less
than 5. 6 whereas pH of pure rain water is around 5. 6 due to dissolution of
atmospheric CO2 in it.
Effects of Acid rain
Acid rain affects us in many ways.
Some of the consequences are given below.
* It irritates eyes and skin of
human beings.
* It inhibits germination and growth
of seedlings.
* It changes the fertility of the
soil, destroys plants and aquatic life.
* It causes corrosion of many
buildings, bridges etc.
Preventive measures
Acid rain and its effects can be
controlled by the following ways.
* Minimizing the usage of fossil
fuel such as petrol, diesel etc. ,
* Using CNG (Compressed Natural
Gas).
* Using non-conventional source of
energy.
* Proper disposal of the industrial
wastes.
Related Topics