Chapter: Medical Physiology: Adrenocortical Hormones
Abnormalities of Adrenocortical Secretion
Abnormalities of Adrenocortical Secretion
Hypoadrenalism-Addison’s Disease
Addison’s disease results from failure of the adrenal cortices to produce adrenocortical hormones, and this in turn is most frequently caused byprimary atrophy of the adrenal cortices. In about 80 per cent of the cases, the atrophy is caused by autoimmunity against the cor-tices. Adrenal gland hypofunction is also frequently caused by tuberculous destruction of the adrenal glands or invasion of the adrenal cortices by cancer. The dis-turbances in Addison’s disease are as follows.
Mineralocorticoid Deficiency. Lack of aldosterone secre-tion greatly decreases renal tubular sodium reabsorp-tion and consequently allows sodium ions, chloride ions, and water to be lost into urine in great profusion. The net result is a greatly decreased extracellular fluid volume. Furthermore, hyponatremia, hyperkalemia, and mild acidosis develop because of failure of potassium and hydrogen ions to be secreted in exchange for sodium reabsorption.
As the extracellular fluid becomes depleted, plasma volume falls, red blood cell concentration rises markedly, cardiac output decreases, and the patient dies in shock, death usually occurring in the untreated patient 4 days to 2 weeks after cessation of mineralo-corticoid secretion.
Glucocorticoid Deficiency. Loss of cortisol secretion makesit impossible for a person with Addison’s disease to maintain normal blood glucose concentration between meals because he or she cannot synthesize significant quantities of glucose by gluconeogenesis. Furthermore, lack of cortisol reduces the mobilization of both proteins and fats from the tissues, thereby depressing many other metabolic functions of the body. This sluggishness of energy mobilization when cortisol is not available is one of the major detrimental effects of glu-cocorticoid lack. Even when excess quantities of glucose and other nutrients are available, the person’s muscles are weak, indicating that glucocorticoids are needed to maintain other metabolic functions of the tissues in addition to energy metabolism.
Lack of adequate glucocorticoid secretion also makes a person with Addison’s disease highly susceptible to the deteriorating effects of different types of stress, and even a mild respiratory infection can cause death.
Melanin Pigmentation. Another characteristic of mostpeople with Addison’s disease is melanin pigmentation of the mucous membranes and skin. This melanin is not always deposited evenly but occasionally is deposited in blotches, and it is deposited especially in the thin skin areas, such as the mucous membranes of the lips and the thin skin of the nipples.
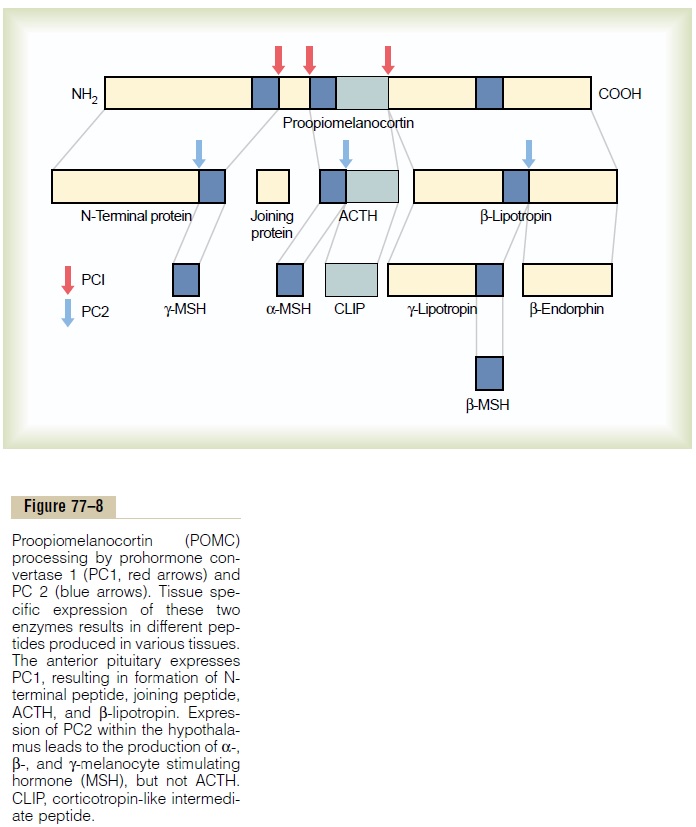
The cause of the melanin deposition is believed to be the following: When cortisol secretion is depressed, the normal negative feedback to the hypothalamus and anterior pituitary gland is also depressed, therefore allowing tremendous rates of ACTH secretion as well as simultaneous secretion of increased amounts of MSH. Probably the tremendous amounts of ACTH cause most of the pigmenting effect because they can stimulate formation of melanin by the melanocytes in the same way that MSH does.
Treatment of People with Addison’s Disease. An untreatedperson with total adrenal destruction dies within a few days to a few weeks because of weakness and usually circulatory shock. Yet such a person can live for years if small quantities of mineralocorticoids and glucocorti-coids are administered daily.
Addisonian Crisis. As noted earlier, greatquantities of glucocorticoids are occasionally secreted in response to different types of physical or mental stress. In a person with Addison’s disease, the output of glucocorticoids does not increase during stress. Yet whenever different types of trauma, disease, or other stresses, such as surgical operations, supervene, a person is likely to have an acute need for excessive amounts of glucocorticoids and often must be given 10 or more times the normal quantities of glucocorticoids to prevent death.
This critical need for extra glucocorticoids and the associated severe debility in times of stress is called an addisonian crisis.
Hyperadrenalism-Cushing’s Syndrome
Hypersecretion by the adrenal cortex causes a complex cascade of hormone effects called Cushing’s syndrome. Most of the abnormalities of Cushing’s syndrome are ascribable to abnormal amounts of cortisol, but excess secretion of androgens may also cause important effects. Hypercortisolism can occur from multiple causes, including (1) adenomas of the anterior pituitary that secrete large amounts of ACTH, which then causes adrenal hyperplasia and excess cortisol secretion; (2) abnormal function of the hypothalamus that causes high levels of corticotropin-releasing hormone (CRH), which stimulates excess ACTH release; (3) “ectopic secretion” of ACTH by a tumor elsewhere in the body, such as an abdominal carcinoma; and (4) adenomas of the adrenal cortex. When Cushing’s syndrome is sec-ondary to excess secretion of ACTH by the anterior pituitary, this is referred to as Cushing’s disease.
Excess ACTH secretion is the most common cause of Cushing’s syndrome and is characterized by high plasma levels of ACTH as well as cortisol. Primary overpro-duction of cortisol by the adrenal glands accounts for about 20 to 25 per cent of clinical cases of Cushing’s syn-drome and is usually associated with reduced ACTH levels due to cortisol feedback inhibition of ACTH secretion by the anterior pituitary gland.
Administration of large doses of dexamethasone, a synthetic glucocorticoid, can be used to distinguish between ACTH-dependent and ACTH-independentCushing’s syndrome. In patients who have overproduc-tion of ACTH due to an ACTH-secreting pituitary adenoma or to hypothalamic-pituitary dysfunction, even large doses of dexamethasone usually do not suppress ACTH secretion. In contrast, patients with primary adrenal overproduction of cortisol (ACTH-independent) usually have low or undetectable levels of ACTH. The dexamethasone test, although widely used, can sometimes give an incorrect diagnosis, because some ACTH-secreting pituitary tumors respond to dexamethasone with suppressed ACTH secretion. Therefore, it is usually considered to be a first step in the differential diagnosis of Cushing’s syndrome.
Cushing’s syndrome can also occur when large amounts of glucocorticoids are administered over pro-longed periods for therapeutic purposes. For example, patients with chronic inflammation associated with dis-eases such as rheumatoid arthritis are often treated with glucocorticoids and may develop some of the clinical symptoms of Cushing’s syndrome.
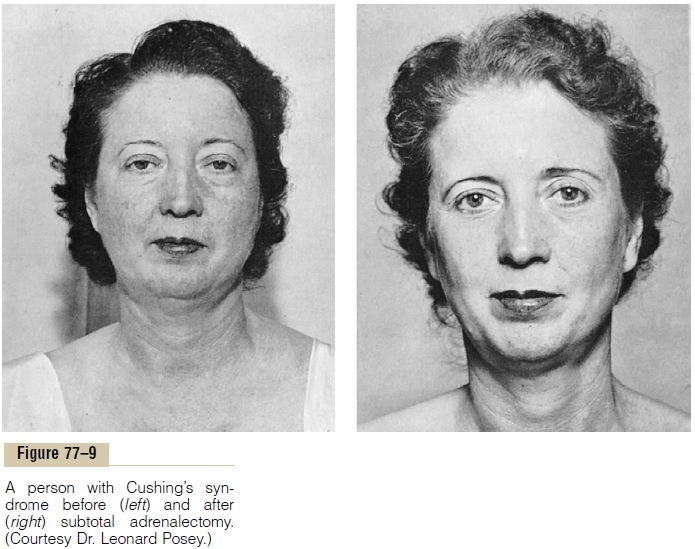
A special characteristic of Cushing’s syndrome is mobilization of fat from the lower part of the body, with concomitant extra deposition of fat in the thoracic and upper abdominal regions, giving rise to a buffalo torso. The excess secretion of steroids also leads to an ede-matous appearance of the face, and the androgenic potency of some of the hormones sometimes causes acne and hirsutism (excess growth of facial hair). The appearance of the face is frequently described as a “moon face,” as demonstrated in the untreated patient with Cushing’s syndrome to the left in Figure 77–8. About 80 per cent of patients have hypertension, pre-sumably because of the slight mineralocorticoid effects of cortisol.
Effects on Carbohydrate and Protein Metabolism. The abun-dance of cortisol secreted in Cushing’s syndrome can cause increased blood glucose concentration, some-times to values as high as 200 mg/dl after meals-as much as twice normal. This results mainly from enhanced glu-coneogenesis and decreased glucose utilization by the tissues.
The effects of glucocorticoids on protein catabolism are often profound in Cushing’s syndrome, causing greatly decreased tissue proteins almost everywhere in the body with the exception of the liver; the plasma pro-teins also remain unaffected. The loss of protein from the muscles in particular causes severe weakness. The loss of protein synthesis in the lymphoid tissues leads to a suppressed immune system, so that many of these patients die of infections. Even the protein collagen fibers in the subcutaneous tissue are diminished so that the subcutaneous tissues tear easily, resulting in devel-opment of large purplish striae where they have torn apart. In addition, severely diminished protein deposi-tion in the bones often causes severeosteoporosis with consequent weakness of the bones.
Treatment of Cushing’s Syndrome. Treatment of Cushing’ssyndrome consists of removing an adrenal tumor if this is the cause or decreasing the secretion of ACTH, if this is possible. Hypertrophied pituitary glands or even small tumors in the pituitary that oversecrete ACTH can sometimes be surgically removed or destroyed by radiation. Drugs that block steroidogenesis, such as metyrapone, ketoconazole, and aminoglutethimide, orthat inhibit ACTH secretion, such as serotonin antago-nists and GABA-transaminase inhibitors, can also beused when surgery is not feasible. If ACTH secretion cannot easily be decreased, the only satisfactory treat-ment is usually bilateral partial (or even total) adrena-lectomy, followed by administration of adrenal steroids to make up for any insufficiency that develops.
Primary Aldosteronism (Conn’s Syndrome)
Occasionally a small tumor of the zona glomerulosa cells occurs and secretes large amounts of aldosterone; the resulting condition is called “primary aldostero-nism” or “Conn’s syndrome.” Also, in a few instances, hyperplastic adrenal cortices secrete aldosterone rather than cortisol. The effects of the excess aldosterone are discussed in detail earlier. The most important effects are hypokalemia, slight increase in extracellular fluid volume and blood volume, very slight increase in plasma sodium concentration (usually not more than a 4 to 6 mEq/L increase), and, almost always, hypertension. Especially interesting in primary aldosteronism are occasional periods of muscle paralysis caused by the hypokalemia. The paralysis is caused by a depressant effect of low extracellular potas-sium concentration on action potential transmission by the nerve fibers.
One of the diagnostic criteria of primary aldostero-nism is a decreased plasma renin concentration. This results from feedback suppression of renin secretion caused by the excess aldosterone or by the excess extra-cellular fluid volume and arterial pressure resulting from the aldosteronism. Treatment of primary aldos-teronism is usually surgical removal of the tumor or of most of the adrenal tissue when hyperplasia is the cause.
Adrenogenital Syndrome
An occasional adrenocortical tumor secretes excessive quantities of androgens that cause intense masculiniz-ing effects throughout the body. If this occurs in a female, she develops virile characteristics, including growth of a beard, a much deeper voice, occasionally baldness if she also has the genetic trait for baldness, masculine distribution of hair on the body and the pubis, growth of the clitoris to resemble a penis, and deposi-tion of proteins in the skin and especially in the muscles to give typical masculine characteristics.
In the prepubertal male, a virilizing adrenal tumor causes the same characteristics as in the female plus rapid development of the male sexual organs, as shown in Figure 77–9, which depicts a 4-year-old boy with adrenogenital syndrome. In the adult male, the viriliz-ing characteristics of adrenogenital syndrome are usually obscured by the normal virilizing characteristics of the testosterone secreted by the testes. It is often dif-ficult to make a diagnosis of adrenogenital syndrome in the adult male. In adrenogenital syndrome, the excre-tion of 17-ketosteroids (which are derived from andro-gens) in the urine may be 10 to 15 times normal. This finding can be used in diagnosing the disease.
Related Topics