Chapter: Microbiology and Immunology: Virology, Virus: Laboratory Diagnosis of Viral Diseases
Viral Assays - Methods of Laboratory Diagnosis of Viral Diseases
Methods of Laboratory Diagnosis
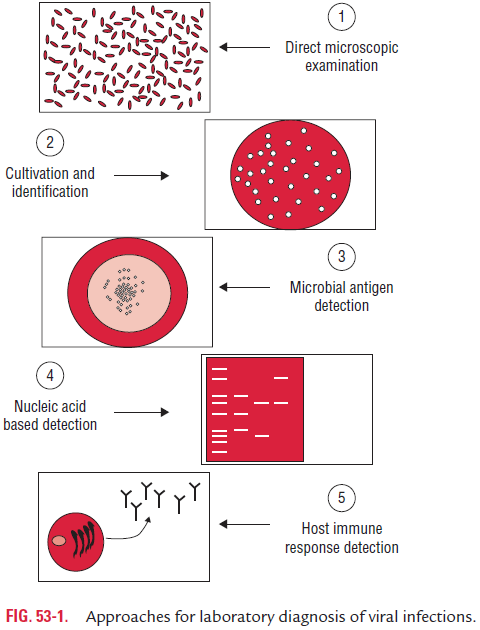
Laboratory diagnosis of viral infections can be carried out by many methods. These methods include
(a) demonstration of virus-induced cytopathic effects (CPEs) in the cells,
(b) direct detection of viruses,
(c) virus isolation and viral assays,
(d) detection of viral proteins and other enzymes,
(e) detection of viral genome, and
(f) viral serology (Fig. 53-1).
Viral Assays
The virus content of a specimen can be assessed either by estimating total virus particles count or by assay of infectivity of viruses.
◗ Total virus particles count
Electron microscopy and hemagglutination are the two meth-ods used for estimation of total virus particles.
Electron microscopy: EM is useful to count virus particlesdirectly in a negatively stained viral suspension. In this method, the virus suspension is mixed with a known concentration of latex particles, and the number of virus particles in the suspen-sion is estimated by a ratio between the virus and latex particles demonstrated by EM.
Hemagglutination: Quantitation of hemagglutinating virusesis carried out by determination of hemagglutination titers. Although it is not a sensitive method, it is used as a convenient method of virus assay. For example, approximately, 107 influ-enza virions are essential to produce microscopic agglutination in cultured cells.
◗ Assay of infectivity of viruses
Quantitative and quantal assays are the two types of assays, which are carried out to determine assay of infectivity of viruses.
Quantitative assay of infectivity: Quantitative assay is usedto estimate the presence of actual number of viable infectious viral particles in the inoculum. Two methods are available for the purpose, which include plaque assay in monolayer cell culture and pock assay on chick embryo CAM.
· Plaque assay: It was introduced by Dulbecco in 1952 as amodification of bacteriophage plaque assay. This is based on the principle that each infectious viral particle gives rise to a localized focus of infected cells that can be visual-ized by the naked eye. Such foci are called plaques, and each plaque indicates an infectious virus. The test is performed by adding a viral inoculum to a monolayer of culture cells in a bottle or Petri dish. After sometime, this allows adsorp-tion of viruses. The liquid medium is removed and replaced with a solid agar gel to ensure that the spread of progeny virus is confined to the immediate vicinity of infected cells.
· Pock assay: Viruses that form pocks on CAM can beassayed by counting the number of pocks formed on the inoculated CAM. Each pock on CAM arises from a single virus particle. This is known as pock assay. Vaccinia and variola viruses can be assayed by pock assay.
Quantal assays of infectivity: Quantal assays of infectivitycan be carried out to quantitate a virus by quantitating the virus particles in animals, in embryonated eggs, or in tissue culture. This method of assay of infectivity only indicates the presence or absence of infectious viruses, but it does not indi-cate actual number of viruses. The endpoints used for infec-tivity titrations are estimated by the (a) development of CPE in cell cultures, (b) production of hemagglutination in allan-toic fluid of embryonated egg, or (c) death of experimentally infected animals. The titer of virus is usually expressed as the “50% infectious dose (ID50)” per mL, which indicates the high-est dilution of the inoculum that initiates detectable symp-toms, antibodies, or other responses in 50% of inoculated test animals, eggs, or cell cultures.
Related Topics