Chapter: Medical Immunology: Cell-Mediated Immunity
T-Cell Activation
T-CELL ACTIVATION
A. Initial Recognition of Antigen-Derived Peptides by T-Cell Receptors
Early observations showed that T lymphocytes couldn’t respond to soluble, unmodified antigens to which B lymphocytes obviously respond with antibody synthesis. T cells rec-ognize antigen-derived peptides associated to self MHC molecules. The tertiary structure of the oligopeptides generated as a consequence of processing by anti-gen-presenting cells (APCs) has no resemblance to the tertiary structure of the native anti-gen. In other words, T and B cells respond to very different structures of one given antigen.
The antigen-derived oligopeptides that fit into the binding sites of MHC-I or MHC-II molecules interact with T-cell receptors of different T-cell subpopulations. MHC-II–pep-tide complexes expressed on the surface of APC are recognized by the T-cell receptor of CD4+ lymphocytes, while MHC-I–peptide complexes expressed by a variety of cells are recognized by the T-cell receptor of CD8+ lymphocytes. The recognition of MHC-associ-ated oligopeptides is the first step in T-cell activation. Other signals are required for efficient and complete T-cell activation. While the following discussion applies mostly to CD4+ cells, the general principles also apply to CD8+ cells, which have been less well studied.
B. Co-stimulatory Signals Essential for T-Cell Activation
Each T cell carries a unique TcR that recognizes only one MHC-antigen complex. This in-teraction delivers an antigen-specific signal, essential but not sufficient, for T-cell activa-tion. Co-stimulatory signals are delivered by the APCs with which CD4 T cells establish strong adhesion interactions (Fig. 11.1). In the absence of co-stimulatory signals, TcR oc-cupancy, by itself, induces T-cell unresponsiveness or anergy .
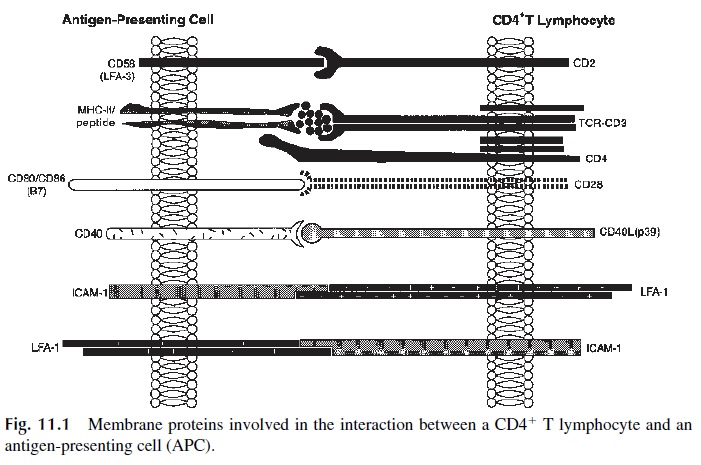
The delivery of co-stimulatory signals requires close interactions between helper and effector cells. Two sets of adhesion molecules (CD2/CD58 and LFA-1/ICAM-1) play a pri-mary role in T lymphocyte–APC adhesion, allowing the proper delivery of co-stimulatory signals.
CD2 molecules expressed by essentially all T lymphocytes bind to CD58 (LFA-3) molecules expressed by most nucleated cells as well as by erythrocytes. The initial inter-action between the MHC-peptide complex and the TcR causes a conformational change on the CD2 molecule that increases the affinity of CD2 for the CD58 molecule expressed by the APC. In addition to its role in stabilizing cell-cell contact, the interaction between CD2 and CD58 delivers an activating signal to the T lymphocyte.
The leukocyte function antigen-1 (LFA-1) molecule that interacts with the intercel-lular adhesion molecule 1 (ICAM-1) also undergoes conformational changes in the early stages of T-cell activation. Both APC and T cells express LFA-1 and ICAM-1, and the het-erotypic interaction between pairs of these molecules results in the formation of strong in-tercellular bonds.
The CD4 molecule on the lymphocyte membrane interacts with nonpolymorphic ar-eas of the class II MHC molecules on the APC. This interaction may not only help stabi-lize the contact between MHC-II and TcR but is also involved in signal transduction in the early stages of T-lymphocyte activation.
The establishment of APC–T-lymphocyte interactions brings the membranes of the two cells into close proximity. Such close contact is critical for the delivery of additional activating signals to the T cell and allows for high local concentrations and maximal effects of the interleukins and cytokines released by APC and T lymphocytes. Some of the most significant costimulatory signals result from cell-cell interactions mediated by membrane proteins, such as those involving the interactions between CD28 and B7 family members and between CD40L and CD40. Others involve cytokines, such as interleukin-1 (IL-1), in-terleukin-6 (IL-6), interleukin-12, and tumor necrosis factor- α(TNF-α ), released by APCs activated as a consequence of ingestion and processing of immunogenic substances. These interleukins provide essential signals necessary for the proliferation of antigen-primed T lymphocytes, acting as cofactors in early T-cell activation.
C. Intracellular Events Associated with the Activation of CD41 T Lymphocytes
When the adequate complement of activating signals is delivered to a CD4+T lymphocyte, a cascade of intracellular signals is triggered. The activation sequence can be divided into two distinct phases.
1. From TcR Occupancy to Interleukin-2 Gene Expression
The TcR heterodimer itself has no recognizable kinase activity. The associated CD3 complex, however, has at least 10 intracytoplasmic motifs (ITAMs) that are believed to play a key role in the activation sequence, particularly the - chain–associated ITAMs. Several tyrosine and serine/threonine kinases have been pro-posed to play a role in T-lymphocyte activation (Fig. 11.2).
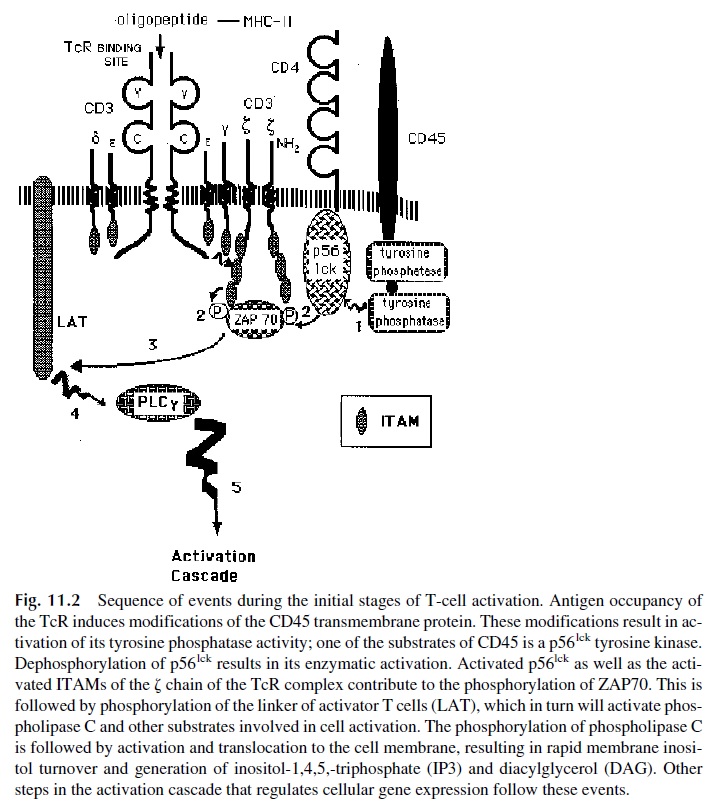
· The src-related tyrosine kinase p56lck, which is noncovalently associated with the cy-toplasmic domains of CD4 and of CD8, is apparently activated by dephospho-rylation mediated by CD45, a tyrosine phosphatase constitutively expressed on all hematopoietic cells. Although the molecular details are not well understood, it is known that CD45 is linked to the TcR and any signal leading to TcR clus-tering after interaction with several MHC-peptide complexes activates CD45 phosphatase activity.
· In addition to p56lck activation, a second src-related tyrosine kinase, p59fyn, is acti-vated by CD45. p59fyn is associated with the chain of the CD3 molecule af-ter TcR occupancy.
· The activation of p56lck and of p59fyn is followed by phosphorylation of tyrosine residues in the ITAMs of the CD3 ζ chains. The phosporylation of CD3 ζ ITAMs is followed by recruitment and activation of a third tyrosine kinase, the ZAP70 kinase.
The activation of ZAP70 leads to subsequent phosphorylation and activation of ad-ditional substrates, including MAP kinase and phospholipase Cγ (PLC-γ ). Apparently as a consequence of PLC-γ activation, membrane phosphoinositols are hydrolyzed yielding two potential intracellular second messengers. One of these messengers is inositol-1,4,5-triphosphate (IP3), responsible for a sharp rise in intracellular ionized calcium concen-tration by mobilizing Ca2+ from intracytoplasmic stores. The rise in Ca2+ allows activa-tion of the calcium- and calmodulin-dependent serine/threonine phosphatase calcineurin. The second is diacylglycerol (DAG), responsible for the activation of protein kinase C (PKC).
Other pathways of phosphoinositol hydrolysis resulting from the engagement of co-stimulatory molecules are being intensively studied. One that has received considerable at-tention involves the activation of a PI3 kinase, triggered as a consequence of the engage-ment of the CD28 molecule on the T-cell membrane by its ligand (CD80), constitutively expressed on the membrane of APCs.
Once these kinases are activated, transcription factors need to be activated in order to induce the expression of a large number of molecules that control T-cell proliferation. These molecules are not expressed by resting T cells, and their genes must be transcribed de novo. Over 70 genes are expressed in a characterizable sequence starting minutes after activation of a helper cell and continuing for the next several days. Those genes encode crit-ical transcription factors, enzymes, and substrates involved in proliferation, differentiation, and effector function.
If the signaling cascade is interrupted or the synthesis of new genes is halted, the im-mune response will be perturbed. If no IL-2 is produced, T cells cannot proliferate and the immune response fails to develop. In some instances the lack of IL-2 synthesis results in T-lymphocyte anergy, i.e., inability to mount an active immune response after proper stimu-lation. In other cases, improperly activated T lymphocytes undergo apoptotic cell death consequent to the expression of alternate sets of apoptosis-inducing genes.
A major determinant of the ultimate fate of an activated T cell is believed to be the level of expression of anti-apoptotic genes, such as Bcl-2. When the cells are productively activated, these genes are upregulated and the cells proliferate. If, however, Bcl-2 and re-lated genes remain downregulated, apoptotic stimuli will proceed unchecked.
Among the well-characterized transcription factors involved in T-cell activation, the following deserve special mention:
· Nuclear factor kB (NF-kB) is a DNA-binding protein found complexed with a spe-cific inhibitor (IkB) in the cytoplasm of resting T lymphocytes. After phos-phorylation by a IkB kinase, IkB is destroyed and the untethered NF-kB moves into the nucleus, where it binds to the promoters of many genes, including the IL-2 receptor gene and the c-myc gene, mediating their transcriptional activa-tion (Fig. 11.3). Additionally, NF-kB activation is essential for the prevention of apoptotic cell death.
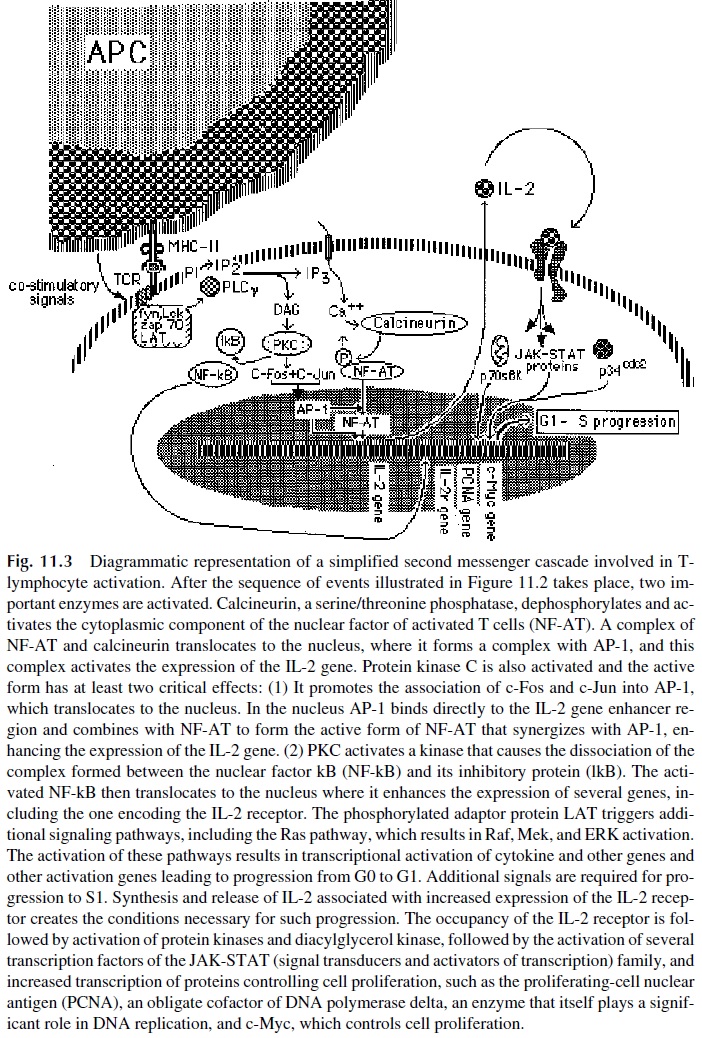
· Nuclear factor of activated T cells (NF-AT1) is almost exclusively found in hematopoietic cells. NF-AT1 binds to regions of specific DNA sequences, found in the 5 promoter region of most cytokine genes. Two components are required to form a complex capable of transcriptional activation: NFAT-1 and AP-1.
· An inactive phosphorylated form of NF-AT1 is located in the cytoplasm of resting T cells. This form is activated by dephosphorylation catalyzed by a calcium-dependent serine/threonine phosphatase known as calcineurin. Upon activation, NFAT-1 translocates into the nucleus, where it becomes associated with AP-1.
· AP-1 is a nuclear protein induced when T cells are stimulated via the TcR plus an-cillary signals mediated by IL-1 or by CD28 cross-linking. It is formed by the association of two proto-oncogene products, c-Fos and c-Jun, whose synthesis is activated by PKC. The cytoplasmic form of c-Jun is activated by a specific kinase (c-Jun N terminal kinase). c-Fos is activated by substrates of the MAP kinase pathway, including Elk. c-Fos and c-Jun form a complex known as the AP-1 transcription factor, which diffuses into the nucleus, where it binds to many DNA sequences including those close to the NF-AT-1–binding site. The full expression of cytokine and cytokine receptor genes is controlled by pro-moter sequences recognized by NF-kB and/or by the complex of NFAT-1 and AP-1. Their occupancy by these transcription factors is essential for control of their expression during T-cell activation.
2. IL-2 Synthesis and T-Lymphocyte Proliferation
Two critical early events in T-cell activation are the appearance of mRNAs for interleukin-2 (IL-2) and for the IL-2 receptor (IL-2R) in the cytoplasm. The upregulation of the IL-2 gene, an essential step for T-cell proliferation at the onset of the immune response, requires occupancy of the promoter region by NF-AT/AP-1/NF-kB complexes. The expression of the IL-2 receptor, on the other hand, seems to be primarily controlled by NF-kB. The re-lease of IL-2 into the cellular environment of activated T cells expressing IL-2 receptors has significant biological consequences.
Autocrine and Paracrine Effects of IL-2.IL-2 stimulates lymphocyte prolifera-tion in both autocrine and paracrine loops. Autocrine stimulation involves release of IL-2 from an activated T cell and binding and activation of IL-2 receptors expressed by the same T cell. Paracrine stimulation is likely a consequence of IL-2 overproduction after persistent stimulation of CD4+ T cells. The released IL-2 exceeds the binding capacity of the IL-2 re-ceptors expressed by the producing cell and can stimulate other nearby cells expressing those receptors. The targets of the paracrine effects of IL-2 are helper T lymphocytes, cy-totoxic T lymphocytes, B lymphocytes, and NK cells, all of which express IL-2 receptors of varying affinities.
The IL-2 Receptor.The IL-2R expressed by T and B lymphocytes is composed ofthree different polypeptide chains.
· CD25, a 55 kDa polypeptide chain (IL-2R a chain), is expressed at very low lev-els in about 30% of the circulating (nonactivated) T cells but is sharply upregu-lated a few hours after activation. CD25 binds IL-2 with low affinity and has a short intracytoplasmic domain unable to transduce growth signals upon binding to its ligand.
· The IL2R b chain is a 70–75 kDa polypeptide chain expressed by resting T lym-phocytes. The IL-2R a/b heterodimer has an increased affinity for IL-2. The b chain appears capable of signal transduction, as its intracytoplasmic domain is 286 amino acids long. In addition, engagement of a/b dimers results in the re-cruitment of g chains, forming the high-affinity IL-2R.
· The IL2R g chain, or gc, is the third polypeptide chain that constitutes the trimeric IL-2 receptor and is expressed after T-cell activation. This chain has also a long intracytoplasmic segment and is therefore structurally suited to transmit activation signals. The gc chain is shared by a number of cytokine receptors, in-cluding those for IL-2, IL-4, IL-7, IL-9, and IL-15.
Consequences of the Binding of IL-2 to the IL-2 Receptor on an Activated T Cell. The binding of IL-2 to the trimeric high-affinity IL-2 receptor induces the associa-tion of the IL-2R to the src-related p56lck tyrosine kinase and to other specific tyrosine ki-nases of the JAK family. The end result is the phosphorylation of transcription factors of the STAT family, which dimerize after phosphorylation and then translocate to the nucleus.
In the nucleus, activated STAT factors upregulate the expression of genes coding for cy-tokine receptors and induce the appearance of cyclins. As a consequence, the cell enters into S phase (see Fig. 11.3).
Progression to Mitosis: Requirement for Co-stimulatory Signals. IL-2 synthe-sis and IL-2R expression are sustained for about 48 hours and subsequently downregulated. Persistent IL-2 gene expression, however, is required to sustain the proliferation drive. This requires additional signals delivered by other molecules, particularly those resulting from CD86-CD28 interactions. Twelve hours after effective T-cell stimulation by APC, CD86 (B7-2)—a molecule related to CD80 (B7-1)—becomes expressed on the APCs, probably as a consequence of autocrine cytokine stimulation. CD86 activates T cells through inter-action with a membrane protein known as CD28. The interaction between CD86 and CD28 results in the synthesis of a set of DNA-binding proteins that appear to block the rapid de-cay of the IL-2 mRNA and induce members of the anti-apoptotic Bcl-2 gene family. If the CD80/86-CD28 interaction is inhibited, T cells will become anergic, i.e., unresponsive to subsequent stimulation.
T lymphocyte Proliferation and Differentiation.Even when co-stimulatory sig-nals are delivered, IL-2 synthesis and IL-2R expression are sustained for about 48 hours and are subsequently downregulated. However, the T cell is committed to cell division within 8 hours of IL-2 interacting with IL-2R. After commitment, even if all triggering sig-nals are withdrawn, the T cell will divide. Clonal expansion of antigen-specific CD4+ T lymphocytes takes place as a consequence of sustained activation through the IL-2 receptor.
It is estimated that the initial helper T-lymphocyte population is capable of expand-ing 100-fold in 6 days, reaching more than 1000-fold its starting number by day 10, the time that it takes to elicit a detectable primary immune response to most infectious agents. This expansion results in the differentiation not only of the short-lived effector cells needed for the ongoing immune response but also of long-lived memory cells. Following such rapid and robust T-cell expansion, many of the activated effector cells will undergo apoptosis. However, when the immune response subsides, an expanded population of memory T cells will remain and will be able to assist the onset of subsequent responses to the same antigen with greater efficiency and speed, which is characteristic of an anamnestic (memory) re-sponse.
D. Cytokines and Chemokines
One of the most remarkable consequences of the activation of CD4+T cells is the synthe-sis of cytokines, soluble molecules that deliver activating or inhibitory signals to other leukocyte populations. A special subset of cytokines is the interleukins, of which 23 dif-ferent ones have been characterized. Although activated CD8+ cells may produce some cy-tokines, and APCs themselves produce others, CD4+ T cells are their major source. The bi-ological effects of cytokines and chemokines are extremely diverse: they influence not only the immune response but also inflammatory processes and hematopoiesis (Table 11.1).


1. Pro-inflammatory Cytokines
The group of soluble factors that influence inflammatory reactions includes interleukin-1 (IL-1), tumor necrosis factor (TNF, also known as cachectin), interferon-γ , interleukin-6 and -8, and migration inhibition factor (MIF). IL-1 and TNF have membrane-associated and secreted forms and seem to be directly or indirectly responsible for the systemic metabolic abnormalities and circulatory collapse characteristic of shock associated with se-vere infections. The main biological functions of these two cytokines are listed in Table 11.2.

IL-1 exists in two molecular forms, IL-1α and IL-1β , encoded by two separate genes and displaying only 20% homology to one another. In spite of this structural difference, both forms of IL-1 bind to the same receptor and share identical biological properties. IL-1α tends to remain associated to cell membranes, while IL-1β , synthesized as an inactive precursor, is released from the cell after being processed posttranslationally by a cysteine-asparagin protease (caspase-1 or interleukin-converting enzyme, ICE).
IL-1 and TNF (Table 11.2) have effects at three different levels:
1.Metabolic effects: IL-1 and TNF induce the synthesis of many proteins such as α1-antitrypsin, fibrinogen, and C-reactive protein in the liver. These proteins are known generically as acute phase reactants, because of their increase in situa-tions associated with inflammatory reactions. In cases of prolonged and severe infections, protracted TNF production may result in negative protein balance, loss of muscle mass, and progressive wasting (cachexia).
Vascular effects: IL-1β and TNF cause the upregulation of cell adhesionmolecules, particularly P-selectin and E-selectin, in vascular endothelial cells. This upregulation promotes the adherence of inflammatory cells, which eventu-ally egress to the extravascular space, where they form tissue inflammatory in-filtrates. One of the possible consequences of the adherence of activated en-dothelial cells and inflammatory cells is endothelial cell damage. This is a major component of gram-negative septic shock and of toxic shock syndrome, both dramatic examples of the adverse effects of massive stimulation of cells capable of releasing excessive amounts of interleukins .
Central nervous system effects: IL-1 does not cross the blood-brain barrier butacts on the periventricular organs where the blood-brain barrier is interrupted. It interacts with a group of nuclei in the anterior hypothalamus, causing fever (sec-ondarily to stimulation of prostaglandin synthesis) and sleep, and increases the production of ACTH.
Interleukin-6 (IL-6), synthesized primarily by monocytes, macrophages, and other antigen-presenting cells, has pro-inflammatory and hematopoietic activities. Its role as a pro-inflammatory cytokine is similar to those of IL-1 and TNF, particularly in the induc-tion of the synthesis of acute phase response proteins by the liver.
2. Cytokines with Predominant Immunoregulatory Functions
Interferon-γ (IFN-γ ), produced by CD4+ cells and NK cells, has a wide range of effects. In concert with IL-1β , it induces an increase in the expression of ICAM-1 on the cytoplas-mic membrane of endothelial cells, enhancing T-lymphocyte adherence to the vascular en-dothelium, an essential first step for T-lymphocyte egress from the vascular bed. Large numbers of T lymphocytes will thus exit the vascular bed in areas near the tissues where activated T cells are releasing interleukins and will form perivenular infiltrates character-istic of delayed hypersensitivity reactions.
The most important effect of IFN-γ seems to be the activation of monocytes. After exposure to IFN-γ , monocytes undergo a series of changes typical of their differentiation into phagocytic effector cells:
· The cell membrane becomes ruffled; the number of cytoplasmic microvilli in-creases by a factor of 10. This change reflects a considerable increase in phago-cytic capacities.
· The expression of MHC class II antigens and Fcγ receptors increases. The increase in the expression of MHC-II enhances the efficiency of monocytes as anti-gen-presenting cells, and the increased expression of Fc receptors further en-hances their efficiency as phagocytic cells.
· The production of cytokines (TNF, IL-12) and of several antimicrobial proteins and compounds [including defensins, cathepsins, collagenases, superoxide radi-cals, and inducible nitric oxide synthase (iNOS)] is upregulated. As a conse-quence, engulfed organisms are rapidly killed and ingested proteins are effi-ciently digested in the phagolysosomes. It must be noted that excessive and protracted production of IFN-γ may have adverse effects. Hyperstimulated monocytes may become exceedingly cytotoxic and may mediate tissue damage in inflammatory reactions and autoimmune diseases.
Interleukin 12 is produced by APCs after endocytosis of particulate materials or mi-crobes and plays a crucial role in linking innate and adaptive immunity. IL-12 induces the production of IFN-γ by CD4+ T cells and NK cells at the early stages of the immune re-sponse, before the differentiation of effector T cells.
Interleukin 18, a recently discovered interleukin, was initially named interferon—γ inducing factor, reflecting its major biological role. In many respects it is similar to IL-1 and IL-12. IL-18 is produced and released by APCs, and its main targets are TH 0/TH1 CD4+ T cells and NK cells.
3. Chemokines
This designation is given to a group of cytokines with chemotactic properties. They are di-vided into two major groups, α and , β depending on their tertiary structure. In the α chemokines one amino acid separates the first two cysteine residues (Cys-X amino acid-Cys) and for that reason are also known as CXC chemokines. In β chemokines the first two cysteine residues are adjacent to each other and for that reason are also known as CC chemokines.
Interleukin-8 (Neutrophil-Activating Factor) is the most important of the α chemokines. It is released by T lymphocytes and monocytes stimulated with TNF or IL-1. It functions as a chemotactic and activating factor for granulocytes, the cell population with the highest level of IL-8 receptor expression. IL-8 recruits granulocytes to areas of inflam-mation and increasing their phagocytic and pro-inflammatory abilities. It has also been demonstrated to be chemotactic for T lymphocytes.
β Chemokines include four major cytokines, which act predominantly on mononu-clear cells, the cells that predominantly express the receptors for this group of cytokines:
1. RANTES (regulated on activation, normal T-cell expressed and secreted), re-leased by T cells, attracts T cells with memory phenotype, NK cells, eosinophils, and mast cells.
2. Macrophage inflammatory proteins (MIP), released by monocytes and macrophages, attract eosinophils, lymphocytes, and NK and LAK cells.
3. Macrophage chemotactic proteins (MCP), produced by monocytes, macrophages, and related cells, attract monocytes, eosinophils, and NK and LAK cells.
4. Eotaxin, a chemokine induced by IL-4 that recruits eosinophils and TH2 CD41 T cells to the sites of allergic inflammation.
Other cytokines and peptides with chemotactic activity, but structurally different from classical chemokines, have also been characterized. Migration-inhibition factor (MIF), the structure of which is not fully known, is released by T cells, monocytes, and macrophages and keeps macrophages in the area where the reaction is taking place and contributes to their activation, promoting the release of TNF. Recently it has been found that endotoxin stimu-lates the release of MIF by the pituitary gland, and its release seems associated with in-creased mortality in the post-acute phase of septic shock. β-Defensins, released primarily by granulocytes, have also been characterized as chemotactic for T lymphocytes.
4. Interleukins and Hematopoiesis
Several different interleukins and cytokines have significant hematopoietic effects, pro-moting the proliferation and differentiation of various cell types.
Interleukin-1 (IL-1), acting as a growth factor on the bone marrow, is a major factor leading to the general mobilization of leukocytes, objectively reflected by peripheral blood leukocytosis, characteristic of bacterial infections.
Granulocyte-monocyte colony-stimulating factor (GM-CSF), released by activated T lymphocytes, stimulates granulocyte and monocyte production and release from the bone marrow. It also stimulates monocyte cytotoxic activity and expression of MHC-II molecules, dendritic cell proliferation and differentiation, and B-cell proliferation and dif-ferentiation (at least in vitro).
Interleukin-3 (IL-3) and stem cell factor, both released principally by activated T lymphocytes, are important growth factors for early bone marrow progenitors, inducing production of a wide variety of leukocytes.
Interleukin-6 (IL-6) promotes the differentiation of B-lymphocyte precursors as well as the differentiation of T-lymphocyte precursors into cytotoxic T lymphocytes.
Interleukin-7 (IL-7) is produced in the bone marrow, thymus, and spleen. Its main role seems to be to induce the proliferation of B- and T-lymphocyte precursors. In the thy-mus, IL-7 drives pre–T-cell differentiation into double positive CD4/CD8+ T-cell precur-sors. In the periphery, IL-7 seems to play a key role promoting the expansion of antigen-stimulated T cells. The molecular mechanisms for this effect on T-cell expansion seems linked to the upregulation of the expression of bcl-2 and related genes, whose products pro-tect the cell against apoptotic death.
5. Cytokine Genes and Structure
The precise role of individual cytokines in the immune and inflammatory responses is dif-ficult to determine because the same cells produce so many cytokines, and each cytokine can exert different and overlapping effects on multiple targets. Shared biological effects cannot be explained by structural similarities between molecules. However, many cytokine receptors activate different but overlapping sets of protein kinases and other signaling molecules. In addition, several interleukin genes appear to be regulated by the same set of nuclear binding proteins, and several interleukin receptors are structurally similar. These may be keys to understanding the plurality of interleukin responses and apparent redun-dancy of effects among structurally different interleukins.
While interleukin genes differ from each other in their coding sequence, they have >70% homology in their untranslated 5’ region, suggesting the use of common transcrip-tion factors, such as NF-AT. In addition, the mRNA transcripts have many AUUUA re-peats near the 3’ end that, in part, determine the rapid rate of mRNA decay. The expression of interleukin genes, therefore, is generally very transient but may be more prolonged de- pending on the activating signals received by the cell. Increased stability of interleukin mR-NAs is observed after calcineurin phosphorylation and after CD28 engagement.
In spite of the diversity of interleukin genes, all monomeric interleukins have similar tertiary structure: at least four α helices arranged in pairs of parallel symmetry, each pair being anti-parallel relative to the other. The α helices are joined by connecting loops that contain β helical sheets of variable length. The helix located near the aminoterminal end is the one that interacts with the cytokine receptor.
6. Cytokine Receptors
Cytokine receptors can be grouped into several families depending on structural character-istics (Fig. 11.4). Upregulation of a given subunit of these receptors is often a consequence of cellular activation and usually results in the expression of a high-affinity receptor able to transduce activation signals. The activation pathways triggered after receptor occupancy tend to be similar for receptors of the same family but differ for receptors of different fam-ilies. The fact that several cytokines may share a given receptor explains why some bio-logical properties are common to several interleukins.
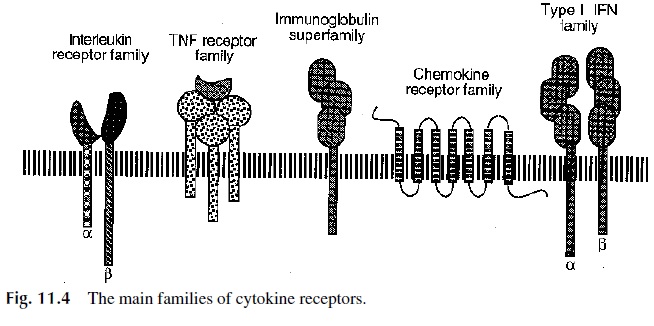
The most common type of receptor is the lymphokine receptor family, formerly known as the hemapoietin-receptor family, because it was initially characterized as the ery-thropoietin receptor. The receptors of this family are heterodimers or heterotrimers and al-ways include an α and a β chain, the latter with a longer intracytoplasmic segment and sig-naling functions. The receptors for IL-2, IL-3, IL-4, IL-5, IL-6, IL-7, IL-9, IL-15, and GM-CSF are included in this family. Some of them share subunits:
1. Receptors for IL-2, IL-4, IL-7, IL-9, and IL-15 share a third chain ( γc), which plays a significant role in signal transduction. One of the forms of severe com-bined immunodeficiency is secondary to abnormalities in the γ-chain gene .
2. Receptors for IL-3, IL-5, and GM-CSF share a common β chain. A different β chain is shared by the receptors for IL-6 and IL-11.
Other receptor families include:
1. The tumor necrosis factor receptor family, including receptors for TNF, lym- photoxin-α (LT-α , TNFβ ), CD40, tumor necrosis factor–related activation-in-duced cytokine (TRANCE), and Fas. There are two types of TNF receptor: TNFR-1 and TNFR-2. TNFR-1 and CD40 both have “death domains” associated with the induction of apoptosis in their intracytoplasmic tails. En-gagement of TNFR-1, however, does not necessarily trigger cell death, because an anti-apoptotic pathway leading to NF- kB synthesis is simultaneously trig-gered; the balance between pro-apoptotic and anti-apoptotic signals dictates the choice between proliferation and apoptosis. In contrast, the engagement of TNFR-2 usually results in cell activation.
2. The immunoglobulin superfamily receptors, which include the IL-1 ( α and β) and IL-18 receptors, a receptor for colony-stimulating factor 1, and, as a special subfamily, the interferon receptors.
The interferon receptors are constituted by two different chains. In the rest-ing state the two chains are loosely associated with kinases, including members of the JAK family. After occupancy with interferon, one or two members of the STAT family of transcription factors undergo phosphorylation and translocation into the nucleus.
The JAK-SAT system is polymorphic. There are three known JAK kinases and seven STAT transcription factors. The STAT-1 transcription factor is predominantly involved in signaling after occupancy of interferon re-ceptors. Its biological significance is underlined by the results of inactivating the encoding gene in mice. The STAT-1–deficient mice do not respond to interfer-ons and are highly susceptible to bacterial and viral infections.
3. The chemokine receptor family, whose common feature is seven transmembrane domains and include receptors for IL-8, platelet factor-4 (PF-4), RANTES, and macrophage chemotactic and activating proteins.
Related Topics