Chapter: Medical Physiology: Dominant Role of the Kidney in Long-Term Regulation of Arterial Pressure and in Hypertension: The Integrated System for Pressure Control
Summary of the Integrated, Multifaceted System for Arterial Pressure Regulation
Summary of the Integrated, Multifaceted System for Arterial Pressure Regulation
By now, it is clear that arterial pressure is regulated not by a single pressure controlling system but instead by several interrelated systems, each of which per- forms a specific function. For instance, when a person bleeds severely so that the pressure falls suddenly, two problems confront the pressure control system. The first is survival, that is, to return the arterial pressure immediately to a high enough level that the person can live through the acute episode. The second is to return the blood volume eventually to its normal level so that the circulatory system can re-establish full normality, including return of the arterial pressure all the way back to its normal value, not merely back to a pressure level required for survival.
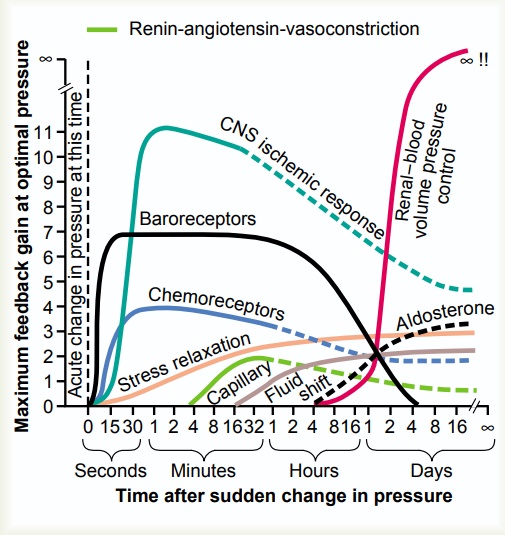
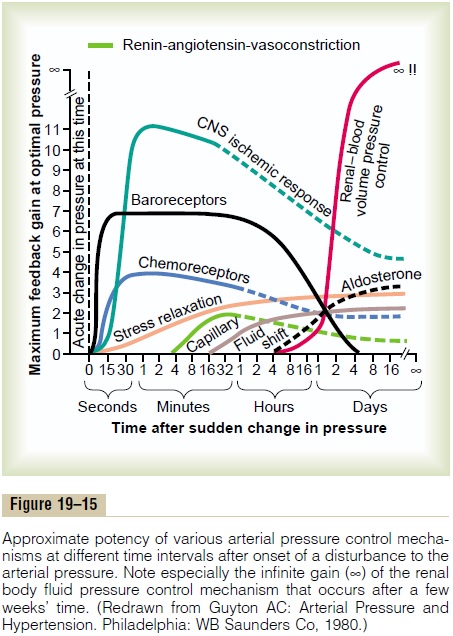
We saw that the first line of defense against acute changes in arterial pressure is the nervous control system. We have emphasized a second line of defense achieved mainly by kidney mechanisms for long-term control of arte-rial pressure. However, there are other pieces to the puzzle. Figure 19–15 helps to put these together.
Figure 19–15 shows the approximate immediate (seconds and minutes) and long-term (hours and days) control responses, expressed as feedback gain, of eight arterial pressure control mechanism. These mecha-nisms can be divided into three groups: (1) those that react rapidly, within seconds or minutes; (2) those that respond over an intermediate time period, minutes or hours; and (3) those that provide long-term arterial pressure regulation, days, months, and years. Let us see how they fit together as a total, integrated system for pressure control.
Rapidly Acting Pressure Control Mechanisms, Acting Within Seconds or Minutes. The rapidly acting pressure controlmechanisms are almost entirely acute nervous reflexes or other nervous responses. Note in Figure 19–15 the three mechanisms that show responses within seconds. They are (1) the baroreceptor feedback mechanism, (2) the central nervous system ischemic mechanism, and (3) the chemoreceptor mechanism. Not only do these mechanisms begin to react within seconds, but they are also powerful. After any acute fall in pressure, as might be caused by severe hemorrhage, the nervous mechanisms combine (1) to cause constriction of the veins and provide transfer of blood into the heart,
(2) to cause increased heart rate and contractility of the heart to provide greater pumping capacity by the heart, and (3) to cause constriction of most peripheral arterioles to impede flow of blood out of the arteries; all these effects occur almost instantly to raise the arterial pressure back into a survival range.
When the pressure suddenly rises too high, as might occur in response to rapid overadministration of a blood transfusion, the same control mechanisms operate in the reverse direction, again returning the pressure back toward normal.
Pressure Control Mechanisms That Act After Many Minutes.
Several pressure control mechanisms exhibit signifi-cant responses only after a few minutes following acute arterial pressure change. Three of these, shown in Figure 19–15, are (1) the renin-angiotensin vaso-constrictor mechanism, (2) stress-relaxation of the vasculature, and (3) shift of fluid through the tissue capillary walls in and out of the circulation to readjust the blood volume as needed.
We have already described at length the role of the renin-angiotensin vasoconstrictor system to provide a semi-acute means for increasing the arterial pressure when this is needed. The stress-relaxation mechanism is demonstrated by the following example: When the pressure in the blood vessels becomes too high, they become stretched and keep on stretching more and more for minutes or hours; as a result, the pressure in the vessels falls toward normal. This continuing stretch of the vessels, called stress-relaxation, can serve as an intermediate-term pressure “buffer.”
The capillary fluid shift mechanism means simply that any time capillary pressure falls too low, fluid is absorbed through the capillary membranes from the tissues into the circulation, thus building up the blood volume and increasing the pressure in the circulation. Conversely, when the capillary pressure rises too high, fluid is lost out of the circulation into the tissues, thus reducing the blood volume as well as virtually all the pressures throughout the circulation.
These three intermediate mechanisms become mostly activated within 30 minutes to several hours. During this time, the nervous mechanisms usually become less and less effective, which explains the importance of these non-nervous, intermediate time pressure control measures.
Long-Term Mechanisms for Arterial Pressure Regulation. To the far right in Figure 19–15 is shown the renal-blood volume pressure control mechanism (which is the same as the renal–body fluid pressure control mecha-nism), demonstrating that it takes a few hours to begin showing significant response. Yet it eventually devel-ops a feedback gain for control of arterial pressure equal to infinity. This means that this mechanism can eventually return the arterial pressure all the way back, not merely partway back, to that pressure level that provides normal output of salt and water by the kidneys.
It must also be remembered that many factors can affect the pressure-regulating level of the renal–body fluid mechanism. One of these, shown in Figure 19–15, is aldosterone. A decrease in arterial pressure leads within minutes to an increase in aldosterone secretion, and over the next hour or days, this plays an important role in modifying the pressure control characteristics of the renal–body fluid mechanism. Especially impor-tant is interaction of the renin-angiotensin system with the aldosterone and renal fluid mechanisms. For instance, a person’s salt intake varies tremendously from one day to another. We have seen that the salt intake can decrease to as little as 1/10 normal or can increase to 10 to 15 times normal and yet the regulated level of the mean arterial pressure will change only a few millimeters of mercury if the renin-angiotensin-aldosterone system is fully opera-tive. But, without a functional renin-angiotensin-aldos-terone system, blood pressure becomes very sensitive to changes in salt intake. Thus, arterial pressure control begins with the lifesaving measures of the nervous pressure controls, then continues with the sustaining characteristics of the intermediate pressure controls, and, finally, is stabilized at the long-term pressure level by the renal–body fluid mechanism. This long-term mechanism in turn has multiple interactions with the renin-angiotensin-aldosterone system, the nervous system, and several other factors that provide special blood pressure control capabilities for special pur-poses.
Related Topics