Chapter: Medical Physiology: Dominant Role of the Kidney in Long-Term Regulation of Arterial Pressure and in Hypertension: The Integrated System for Pressure Control
Quantitation of Pressure Diuresis as a Basis for Arterial Pressure Control
Quantitation of Pressure Diuresis as a Basis for Arterial Pressure Control
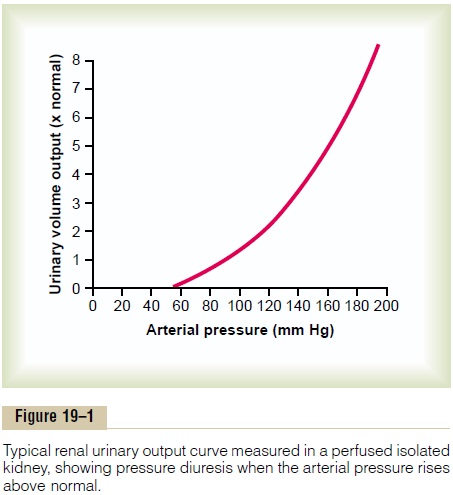
Figure 19–1 shows the approximate average effect of different arterial pressure levels on urinary volume output by an isolated kidney, demonstrating markedly increased output of volume as the pressure rises. This increased urinary output is the phenomenon of pres-sure diuresis. The curve in this figure is called a renal urinary output curve or a renal function curve. In thehuman being, at an arterial pressure of 50 mm Hg, the urine output is essentially zero. At 100 mm Hg it is normal, and at 200 mm Hg it is about six to eight times normal. Furthermore, not only does increasing the arterial pressure increase urine volume output, but it causes approximately equal increase in sodium output, which is the phenomenon of pressure natriuresis.
An Experiment Demonstrating the Renal–Body Fluid System for Arterial Pressure Control. Figure 19–2 shows the resultsof a research experiment in dogs in which all the nervous reflex mechanisms for blood pressure control were first blocked. Then the arterial pressure was sud-denly elevated by infusing about 400 milliliters of blood intravenously. Note the instantaneous increase
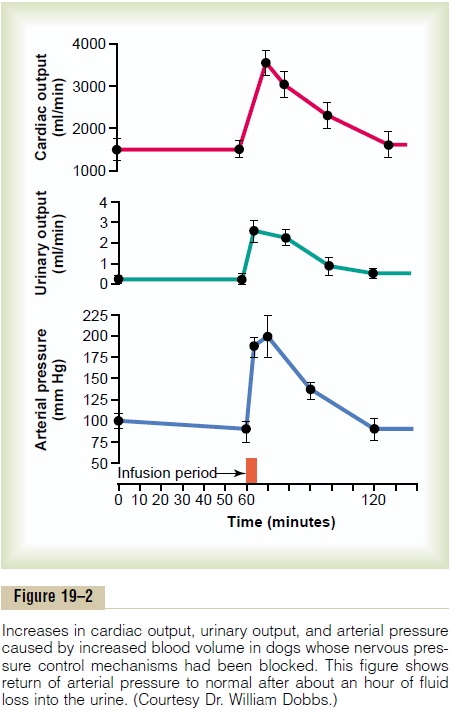
in cardiac output to about double normal and increase in mean arterial pressure to 205 mm Hg, 115 mm Hg above its resting level. Shown by the middle curve is the effect of this increased arterial pressure on urine output, which increased 12-fold. Along with this tremendous loss of fluid in the urine, both the cardiac output and the arterial pressure returned to normal during the subsequent hour. Thus, one sees an extreme capability of the kidneys to eliminate fluid volume from the body in response to high arterial pressure and in so doing to return the arterial pressure back to normal.
Graphical Analysis of Pressure Control by the Renal–Body Fluid Mechanism, Demonstrating an “Infinite Feedback Gain” Feature. Figure 19–3 shows a graphical method thatcan be used for analyzing arterial pressure control by the renal–body fluid system. This analysis is based on two separate curves that intersect each other: (1) the renal output curve for water and salt in response to rising arterial pressure, which is the same renal output curve as that shown in Figure 19–1, and (2) the curve (or line) that represents the net water and salt intake.
Over a long period, the water and salt output must equal the intake. Furthermore, the only place on the graph in Figure 19–3 at which output equals intake is where the two curves intersect, which is called the equilibrium point.
Now, let us see what happens if thearterial pressure becomes some value that is different from that at the equilibrium point.
First, assume that the arterial pressure rises to 150 mm Hg. At this level, the graph shows that renal output of water and salt is about three times as great as the intake. Therefore, the body loses fluid, the blood volume decreases, and the arterial pressure decreases. Furthermore, this “negative balance” of fluid will not cease until the pressure falls all the way back exactly to the equilibrium level. Indeed, even when the arte-rial pressure is only 1 mm Hg greater than the equi-librium level, there still is slightly more loss of water and salt than intake, so that the pressure continues to fall that last 1 mm Hg until the pressure eventuallyreturns exactly to the equilibrium point.
If the arterial pressure falls below the equilibrium point, the intake of water and salt is greater than the output. Therefore, body fluid volume increases, blood volume increases, and the arterial pressure rises until once again it returns exactly to the equilibrium point. This return of the arterial pressure always exactlyback to the equilibrium point is the infinite feedback gain principle for control of arterial pressure by therenal–body fluid mechanism.
Two Determinants of the Long-Term Arterial Pressure Level. InFigure 19–3, one can also see that two basic long-term factors determine the long-term arterial pressure level. This can be explained as follows.
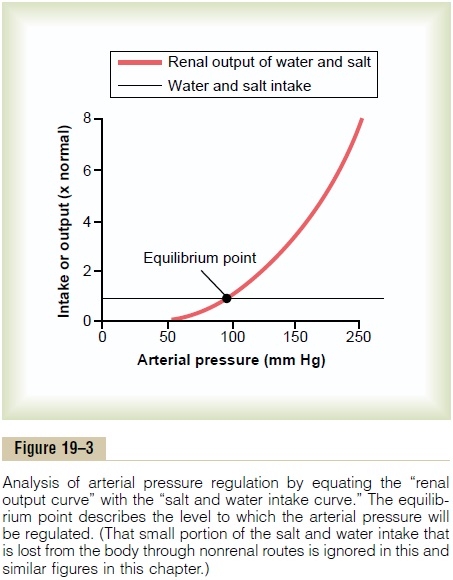
As long as the two curves representing (1) renal output of salt and water and (2) intake of salt and water remain exactly as they are shown in Figure 19–3, the long-term mean arterial pressure level will always readjust exactly to 100 mm Hg, which is the pressure level depicted by the equilibrium point of this figure.
Furthermore, there are only two ways in which the pressure of this equilibrium point can be changed from the 100 mm Hg level. One of these is by shifting the pressure level of the renal output curve for salt and water; and the other is by changing the level of the water and salt intake line. Therefore, expressed simply, the two primary determinants of the long-term arte-rial pressure level are as follows:
1.The degree of pressure shift of the renal output curve for water and salt
2.The level of the water and salt intake line
Operation of these two determinants in the control of arterial pressure is demonstrated in Figure 19–4. In Figure 19–4A, some abnormality of the kidneys has caused the renal output curve to shift 50 mm Hg in the high-pressure direction (to the right). Note that the equilibrium point has also shifted to 50 mm Hg higher than normal. Therefore, one can state that if the renal output curve shifts to a new pressure level, so will the arterial pressure follow to this new pressure level within a few days.
Figure 19–4B shows how a change in the level of salt and water intake also can change the arterial pressure. In this case, the intake level has increased fourfold and the equilibrium point has shifted to a pressure level of 160 mm Hg, 60 mm Hg above the normal level. Con-versely, a decrease in the intake level would reduce the arterial pressure.
Thus, it is impossible to change the long-term meanarterial pressure level to a new value without changingone or both of the two basic determinants of long-term arterial pressure—either (1) the level of salt and water intake or (2) the degree of shift of the renal function curve along the pressure axis. However, if either of these is changed, one finds the arterial pressure there-after to be regulated at a new pressure level, at the pressure level at which the two new curves intersect.
Failure of Increased Total Peripheral Resistance to Elevate the Long-Term Level of Arterial Pressure if Fluid Intake and Renal Function Do Not Change
Now is the chance for the reader to see whether he or she really understands the renal–body fluid mecha-nism for arterial pressure control. Recalling the basic equation for arterial pressure—arterial pressure equals cardiac output times total peripheral resistance—it isclear that an increase in total peripheral resistance should elevate the arterial pressure. Indeed, when thetotal peripheral resistance is acutely increased, the arte-rial pressure does rise immediately. Yet if the kidneys continue to function normally, the acute rise in arterial pressure usually is not maintained. Instead, the arte-rial pressure returns all the way to normal within a day or so. Why?
The answer to this is the following: Increasing resist-ance in the blood vessels everywhere else in the body besides in the kidneys does not change the equilibriumpoint for blood pressure control as dictated by the kidneys (see again Figures 19–3 and 19–4). Instead, the kidneys immediately begin to respond to the high arte-rial pressure, causing pressure diuresis and pressure natriuresis. Within hours, large amounts of salt and water are lost from the body, and this continues until the arterial pressure returns exactly to the pressure level of the equilibrium point.
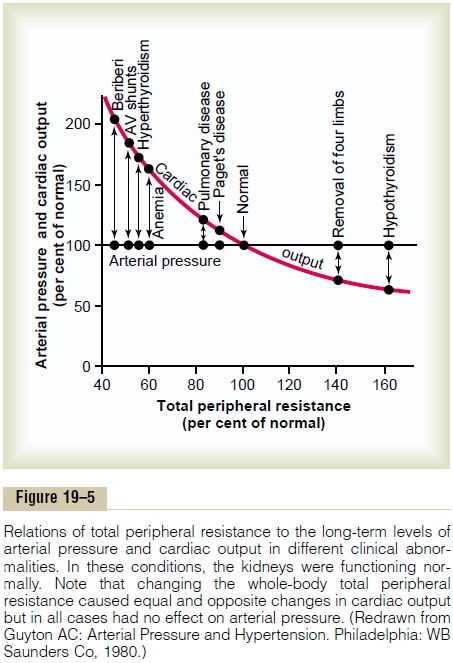
As proof of this principle that changes in total peripheral resistance do not affect the long-term level of arterial pressure if function of the kidneys is still normal, carefully study Figure 19–5. This figure shows the approximate cardiac outputs and the arterial pres-sures in different clinical conditions in which the long-term total peripheral resistance is either much less thanor much greater than normal, but kidney excretion of salt and water is normal. Note in all these different clinical conditions that the arterial pressure is also exactly normal.
(A word of caution! Many times when the total peripheral resistance increases, this increases theintrarenal vascular resistance at the same time, whichalters the function of the kidney and can cause
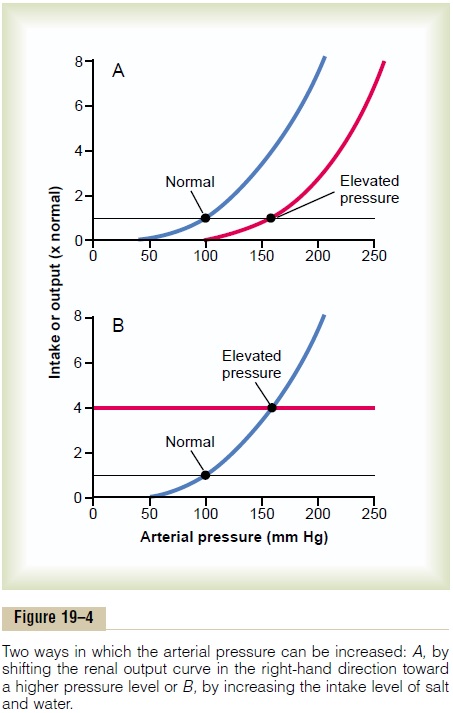
hypertension by shifting the renal function curve to a higher pressure level, in the manner shown in Figure 19–4A. We see an example of this later when we discuss hypertension caused by vasoconstric-tor mechanisms. But it is the increase in renal resistance that is the culprit, not the increased total peripheralresistance—an important distinction!)
Increased Fluid Volume Can Elevate Arterial Pressure by Increasing Cardiac Output or Total Peripheral Resistance
The overall mechanism by which increased extracellu-lar fluid volume elevates arterial pressure is given in the schema of Figure 19–6. The sequential events are (1) increased extracellular fluid volume (2) increases the blood volume, which (3) increases the mean cir-culatory filling pressure, which (4) increases venous return of blood to the heart, which (5) increases cardiac output, which (6) increases arterial pressure.
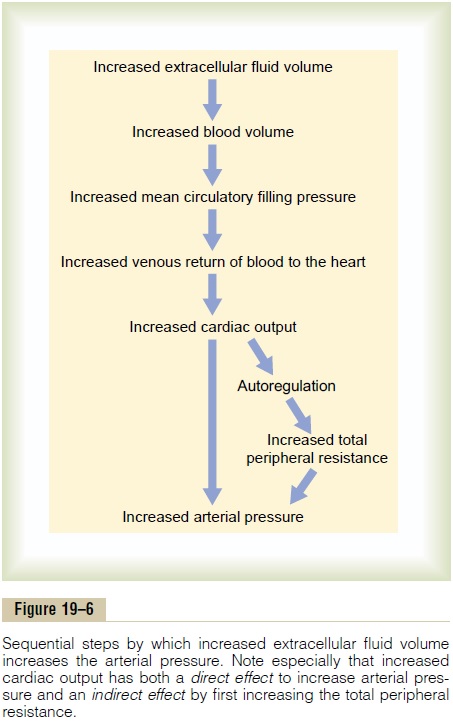
Note especially in this schema the two ways in which an increase in cardiac output can increase the arterial pressure. One of these is the direct effect of increased cardiac output to increase the pressure, and the other is an indirect effect to raise total peripheral vascular resistance through autoregulation of blood flow. The second effect can be explained as follows.
Referring back, let us recall that whenever an excess amount of blood flows through a tissue, the local tissue vasculature constricts and decreases the blood flow back toward normal. This phenomenon is called “autoregulation,” which means simply regulation of blood flow by the tissue itself. When increased blood volume increases the cardiac output, the blood flow increases in all tissues of the body, so that this autoregulation mechanism constricts blood vessels all over the body. This in turn increases the total peripheral resistance.
Finally, because arterial pressure is equal to cardiacoutput times total peripheral resistance, the secondaryincrease in total peripheral resistance that results from the autoregulation mechanism helps greatly in increas-ing the arterial pressure. For instance, only a 5 to 10 per cent increase in cardiac output can increase the arterial pressure from the normal mean arterial pres-sure of 100 mm Hg up to 150 mm Hg. In fact, the slight increase in cardiac output is often unmeasurable.
Importance of Salt (NaCl) in the Renal–Body Fluid Schema for Arterial Pressure Regulation
Although the discussions thus far have emphasized the importance of volume in regulation of arterial pres-sure, experimental studies have shown that an increase in salt intake is far more likely to elevate the arterial pressure than is an increase in water intake.The reason for this is that pure water is normally excreted by the kidneys almost as rapidly as it is ingested, but salt is not excreted so easily. As salt accumulates in the body, it also indirectly increases the extracellular fluid volume for two basic reasons:
1. When there is excess salt in the extracellular fluid, the osmolality of the fluid increases, and this in turn stimulates the thirst center in the brain, making the person drink extra amounts of water to return the extracellular salt concentration to normal. This increases the extracellular fluid volume.
2. The increase in osmolality caused by the excess salt in the extracellular fluid also stimulates the hypothalamic-posterior pituitary gland secretory mechanism to secrete increased quantities of antidiuretic hormone. The antidiuretic hormone then causes the kidneys to reabsorb greatly increased quantities of water from the renal tubular fluid, thereby diminishing the excreted volume of urine but increasing the extracellular fluid volume.
Thus, for these important reasons, the amount of salt that accumulates in the body is the main determinant of the extracellular fluid volume. Because only small increases in extracellular fluid and blood volume can often increase the arterial pressure greatly, accumula-tion of even a small amount of extra salt in the body can lead to considerable elevation of arterial pressure.
Related Topics