Chapter: Essentials of Psychiatry: Stimulants and Related Compounds
Psychostimulants: Efficacy and Utility
Psychostimulants:
Efficacy and Utility
Two
groups of stimulants are currently approved by the FDA for treatment of ADHD in
children, and are available in both brand and generic. These include: the
amphetamines (Adderall, Dextrostat, Dexedrine) and the methylphenidates
(Concerta, Metadate-ER, Metadate-CD, methylphenidate, Methylin, Ritalin,
Ritalin-SR, Ritalin-LA and Focalin). Characteristics of these stimulants can be
found in Table 82.1. Dextroamphetamine (DEX) andmethylphenidate (MPH) are
structurally related to the catecholamines (DA and NE) (McCracken 1991). Signs
of CNS stimulation, including blood pressure and heart rate increases, are more
pronounced in adults. The term psychostimulant
used for these compounds refers to their ability to increase CNS activity in
some but not all brain regions. Stimulants ameliorate disruptive ADHD behaviors
cross-situationally (classroom, lunchroom, playground and home) when repeatedly
administered throughout the day.

Mechanism of Action
The term
psychostimulant refers to the ability of these compounds to increase CNS
activity in some but not all brain regions. For example, while increasing the
activity of striatum and connections between orbitofrontal and limbic regions,
stimulants seem to have an inhibitory effect on the neocortex. The CNS
psychostimulant effects of DEX and MPH may result in part from their lack of
benzene ring substituents. Prominent central effects include activation of the
medullary respiratory center and a lessening of central depression from
barbiturates.
Psychostimulants have been described as noncatecholamine sympathomimetics. Sympathomimetics show potent agonist effects at alpha- and beta-adrenergic receptors. Presynaptically, they cause a stoichiometric displacement of norepinephrine and dopamine from storage sites in the presynaptic terminal. Stimulants also block the reuptake of dopamine by the dopamine transporter (DAT). Postsynaptically, they function as direct agonists at the adrenergic receptor. They also block the action of a degradative enzyme, catechol-o-methyltransferase (COMT).
The
putative action of psychostimulants in ADHD has been attributed to the release
of DA and their ability to block its reuptake by the DAT at the presynaptic
nerve terminal. Radioli-gand binding studies have demonstrated the direct
action of psy-chostimulants, particularly MPH, on striatal DAT. The dopamine
action of psychostimulants also explains the appearance of be-havioral
stereotypies, seen at high doses. Tritiated MPH binding in rat brain is highest
in striatum and is highly dependent on so-dium concentration, suggesting that
MPH binding is associated with a neurotransmitter transport system.
Yet, no
single theory explains the psychostimulant mecha-nism of action on the CNS
which ameliorates ADHD symptoms.
Most
hypotheses regarding the neurochemical basis of ADHD have focused on the
catecholamines norepinephrine and dopamine. These hypotheses, which posit a
dysregulation in the norepinephrine or dopamine neurotransmitter systems, or
both, are based primarily on the success of these dopaminergic medications in
treating ADHD. All stimulant medications used for treating ADHD have their
primary effects on these two neurotransmitter systems. Furthermore, the more
selective norepinephrine-acting tricyclic medications, such as desipramine and
imipramine, as well as the alpha-2-adrenergic agonist clonidine, have all been
found to reduce symptoms of ADHD in children. Similarly, although much less
commonly used, dopamine-blocking antipsychotic medications also have been found
to be effective for treating children with ADHD (Zametkin and Rapoport, 1987).
Pharmacokinetics: Absorption and Metabolism
Psychostimulants
are rapidly absorbed from the gut and thus act quickly, often within the first
30 minutes after ingestion. Food enhances absorption. Due, in part, to MPH’s
low plasma binding rate (15%), it is highly available to cross the blood–brain
barrier. Effects on behavior appear during absorption, beginning 30 minutes
after ingestion and lasting 3 to 4 hours. Half-lives range between 3 hours for
MPH and 6 to 9 hours for d-amphetamine.
The concentration-enhancing and activity-reducing effects of MPH can disappear
well before the medication leaves the plasma, a phenomenon termed clockwise hysteresis.
MPH’s
metabolism is rapid and complete because it is not highly bound to plasma
protein and does not disappear into fat stores. MPH peaks in plasma in 2 to 2.5
hours and falls to half the peak (half-life) after 3 hours.
Because
MPH’s short half-life prevents it from reaching steady state in the plasma, the
standard tablet must be given several times a day to maintain behavioral
improvement through-out the school day. Although the Physicians’ Desk Reference
(PDR) suggests giving MPH before meals, standard administra-tion times are
after breakfast (8:00 AM), after lunch (noon), and before homework (about 3:30
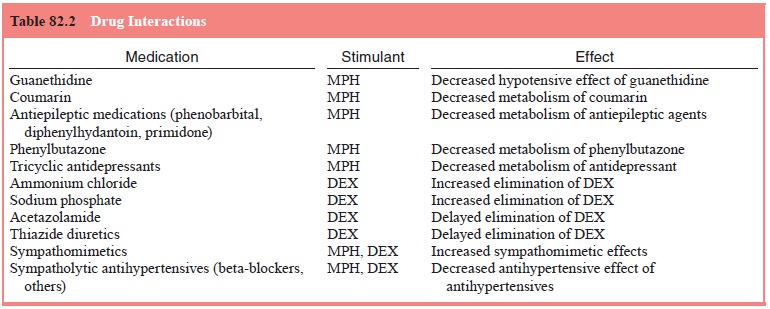
pm). See Table 82.2 for review of some of the pharmacokinetics of the
psychostimulants used to treat children with ADHD.
Effects in Adults
Studies
in adults taking d-amphetamine have
shown a prolonga-tion of performance at repetitive tasks before the onset of
fatigue, a decreased sense of fatigue, mood elevation, euphoria, and increased
speech rate and initiative (Rapoport et
al., 1980). The psychostimulants increase CNS alertness, as shown on tasks
re-quiring vigilance, both in laboratory tasks, such as the CPT, and on the
job, such as maintaining the ability to notice new events on a radar screen for
periods of hours. These changes have been described as the drug’s ability to
“increase capacity”, although this phrase has been misinterpreted to mean an
increase over the person’s innate ability.
Substance Abuse of Stimulants
Psychostimulants
have been abused by adults, but the risk for addiction by children with ADHD is
low. Klein (1980) found that the psychostimulants differ in their ability to
induce euphoria, with “dextroamphetamine the most euphorigenic,
methylphenidate, less so, and magnesium pemoline, hardly at all.” Adolescents
and young adults with ADHD do not list the psychostimulants among medications
used recreationally, whether or not they had received treatment with
psychostimu-lants. Adolescents previously diagnosed as having ADHD during their
school-age years are at greater risk for substance abuse than control subjects,
but those who do abuse medications tend not to pick stimulants.
Even
though the evidence from the literature suggests that the abuse potential of
psychostimulants by children with ADHD is low, DEX and MPH have been classified
by the US Food and Drug Administration as potentially drugs of abuse (schedule
II) and have warnings concerning abuse in the PDR. MPH was re-classified as a
schedule II medication in 1971 because of a con-cern that the order by the
Bureau of Narcotics and Dangerous Drugs to schedule methamphetamine and DEX
would direct po-tential drug abusers to MPH. As a schedule II medication, the
annual amount of stimulant medication manufactured must be approved or
allocated.
Related Topics