Chapter: Medical Microbiology: An Introduction to Infectious Diseases: Pseudomonas and Other Opportunistic Gram-negative Bacilli
Pseudomonas aeruginosa Disease
P.aerugInosa DISEASE
P. aeruginosa producesinfection at awiderangeof pulmonary,urinary, andsofttissue sites, much like the opportunistic Enterobacteriaceae. The clinical manifes-tations of these infections reflect the organ system involved and are not unique for Pseudomonas. However, once established,infections areparticularly virulent anddifficult to treat. Affected patients almost always have some form of debilitation or compromise of immune defenses.
EPIDEMIOLOGY
The primary habitat of P. aeruginosa is the environment. It is found in water, soil, and various types of vegetation throughout the world. P. aeruginosa has been isolated from the throat and stool of 2 to 10% of healthy persons. Colonization rates may be higher in hospitalized patients. Infection with P. aeruginosa, rare in previously healthy persons, is one of the most important causes of invasive infection in hospitalized patients with seri-ous underlying disease, such as leukemia, cystic fibrosis (CF), and extensive burns.
The ability of P. aeruginosa to survive and proliferate in water with minimal nutrients can lead to heavy contamination of any nonsterile fluid, such as that in the humidifiers of respirators. Inhalation of aerosols from such sources can bypass the normal respiratory defense mechanisms and initiate pulmonary infection. Infections have resulted from the growth of Pseudomonas in medications, contact lens solutions, and even in some disinfectants. Sinks and faucet aerators may be heavily contaminated and serve as the en-vironmental source for contamination of other items. The presence of P. aeruginosa in drinking water or food is not a cause for alarm. The risk lies in the proximity between items susceptible to contamination and patients uniquely predisposed to infection.
P. aeruginosa is nowthemost commonbacterialpathogen to complicatethemanage-ment of patients with CF, an inherited defect in chloride ion transport that leads to a buildup of thick mucus in ducts and the tracheobronchial tree. In a high proportion of cases, the respiratory tract becomes colonized with P. aeruginosa, which, once estab-lished, becomes almost impossible to eradicate. This infection is a leading cause of mor-bidity and eventual death of these patients.
PATHOGENESIS
Although P. aeruginosa is an opportunistic pathogen, it is one of particular virulence. The or-ganism usually requires a significant break in first-line defenses (such as a wound) or a route past them (such as a contaminated solution or intratracheal tube) to initiate infection. Attach-ment to epithelial cells is the first step in infection and is likely mediated by pili, flagella, and the extracellular polysaccharide slime. The receptors include sialic acid and N-acetylglu-cosamine borne by cell surface glycolipids. There is evidence that attachment is favored by loss of surface fibronectin, which explains in part the propensity for debilitated persons.
Once established, the virulence of P. aeruginosa involves multiple factors, particu-larly exotoxin A, exotoxin S, and elastase, which are directly injected into host cells by a specialized contact secretion system. The importance of exotoxin A is supported by stud-ies in human and animals, which correlate its presence with a fatal outcome and antibody against it with survival. No diphtheria-like systemic effect of exotoxin has been demon-strated, but its cytotoxic action correlates with the primarily invasive and locally destruc-tive lesions seen in P. aeruginosa infections.
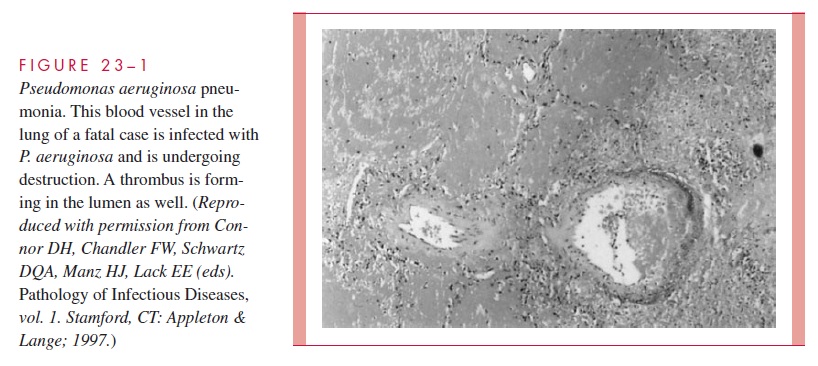
Exoenzyme S is associated with dissemination from burn wounds and with actions destructive to cells, including its action on the cytoskeleton. The many biologically im-portant substrates of elastase argue for its importance, particularly its namesake, elastin. Elastin is found at some sites P. aeruginosa preferentially attacks, such as the lung and blood vessels. Hemorrhagic destruction, including the walls of blood vessels (Fig 23 – 1), is the histologic hallmark of Pseudomonas infection.
P. aeruginosa and Cystic Fibrosis (CF)
P. aeruginosa is the most persistent of the infectious agents that complicate the course ofCF. Initial colonization may be aided by the fact that cells from CF patients are less highly sialylated than normal epithelial cells, providing increased receptors for P. aerugi-nosa attachment. Defects in the epithelia of CF patients may also retard their clearing bydesquamation. Once the bronchi are colonized, the organisms remain, forming a biofilm containing microcolonies of bacteria, which together are called aglycocalyx. The most striking feature of this association is the unique presence of strains with multiple muta-tions in regulatory genes that cause overproduction of the alginate polymer. These genes are activated by the high osmolarity of the thick CF secretions. The selective advantages of this biofilm include adhesion; inaccessibility of the immune system (complement, anti-body, phagocytes); and interference with the access and action of antimicrobial agents.
Virulence Regulation
The multiple virulence factors of P. aeruginosa are controlled by several regulatory pathways, some of which respond to environmental stimuli. In addition, some of the extracellular prod-ucts, including exotoxin A and elastase, are regulated in an interactive way by cell-to-cell signaling. These signaling systems are able to monitor bacterial population density in a way that only initiates transcription of a virulence factor when certain population thresholds are reached. This could be valuable either as an economy measure or as a mechanism to with-hold the onslaught of injury-producing molecules until the host has little time to respond.
IMMUNITY
Human immunity to Pseudomonas infection is not well understood. Inferences from ani- mal studies and clinical observations suggest that both humoral and cell mediated immu- nity are important. The strong propensity of P. aeruginosa to infect those with defective cell-mediated immunity indicates that these responses are particularly important.
Related Topics