Chapter: Modern Pharmacology with Clinical Applications: Local Anesthetics
Properties of Local Anesthetics
The first clinical uses of a
local anesthetic agent oc-curred in 1884, when cocaine was employed as a topical
agent for eye surgery and to produce a nerve block. These events inaugurated a
new era, that of regional anesthesia. New applications were developed,
including spinal, epidural, and caudal anesthesia. The search for a better
local anesthetic led to chemical synthesis of a number of other compounds that
have more selective local anesthetic properties and few systemic side effects.
PROPERTIES OF
LOCAL ANESTHETICS
An important property of the
ideal local anesthetic is low systemic toxicity at an effective concentration.
Onset of action should be quick, and duration of action should be sufficient to
allow time for the surgical proce-dure. The local anesthetic should be soluble
in water and stable in solution. It should not deteriorate by the heat of sterilization,
and it should be effective both when in-jected into tissue and when applied
topically to mucous membranes. Its effects should be completely reversible.
Although the characteristics
of an ideal local anes-thetic are easily identifiable, synthesis of a compound
possessing all these properties has not been accom-plished. The compounds
discussed in the following sec-tions fall short of the ideal in at least one
aspect. However, the judicious choice of a particular agent for a particular
need will permit the practitioner to employ local anesthesia effectively and
safely.
Chemistry
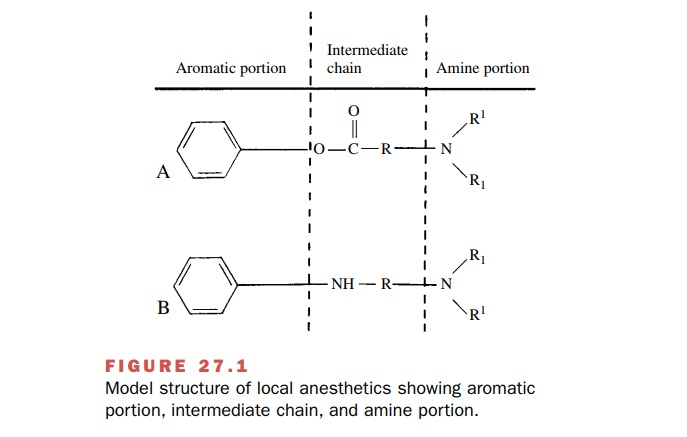
The basic components in the
structure of local anes-thetics are the lipophilic aromatic portion (a benzene
ring), an intermediate chain, and the hydrophilic amine portion (Fig. 27.1).
The intermediate chain has either an ester linkage from the combination of an
aromatic acid and an amino alcohol or an amide linkage from the combination of
an aromatic amine and an amino acid. The commonly used local anesthetics can be
classified as esters or amides based on the structure of this inter-mediate
chain.
Mechanism of Action
The application of a local
anesthetic to a nerve that is actively conducting impulses will inhibit the
inward mi-gration of NA+ . This elevates the threshold for
electrical excitation, reduces the rate of rise of the action poten-tial, slows
the propagation of the impulse, and if the drug concentration is sufficiently
high, completely blocks conduction. The
local anesthetics interfere with the process
fundamental to the generation of the action potential, namely, the large,
transient voltage-dependent rise in the permeability of the membrane to NA+
.
While the physiological basis
for the local anesthetic action is known, the precise molecular nature of the
process is not completely clear. Almost all local anes-thetics can exist as
either the uncharged base or as a cation. The uncharged base is important for
adequate penetration to the site of action, and the charged form of the
molecule is required at the site of action. The cation forms of local
anesthetics appear to be required for binding to specific sites in or near the NA+
channels. The presence of the local anesthetic at these sites inter-feres with
the normal passage of NA+ through the cell membrane by stopping a
conformational change in the subunits of the voltage-gated NA+ channel.
Studies suggest that the
receptor for the local anes-thetic is near the inner (axoplasmic) surface of
the cell membrane, because quaternary analogues of local anes-thetics are quite
effective when applied to the inside of the axonal membrane but are inactive
when placed on the outside of the membrane. These permanently charged molecules
cannot penetrate to the receptor sites.
Nerves that are rapidly
depolarizing are inherently particularly susceptible to the effects of local
anesthet-ics. This is termed frequency-dependent
blockade and is thought to occur because the local anesthetics get totheir
receptor sites only when the NA+ channel is open (depolarizing).
Differential Blockade
Peripheral nerve functions
are not affected equally by local anesthetics. Loss of sympathetic function
usually is followed by loss of temperature sensation; sensation to pinprick,
touch, and deep pressure; and last, motor function. This phenomenon is called differential block-ade. Differential
blockade is the result of a number of factors,
including the size of the nerve, the presence and amount of myelin, and the
location of particular fibers within a nerve bundle. For conduction to be ef-fectively
blocked, the local anesthetic must exert its ef-fects over the distance between
several nodes of Ranvier. Since the smallest nerves (C fibers) have no myelin,
they can be most easily blocked; thus, sympa-thetic functions often are blocked
soon after a local anesthetic is applied to a particular nerve bundle. Small
myelinated nerves have correspondingly short distances between nodes of Ranvier
and therefore are often blocked next. These nerves subserve tempera-ture and
sharp pain sensation. Larger nerves then be-come blocked, accounting for the
loss of function up to and including motor innervation.
Pharmacokinetic Properties
Absorption and Distribution
The rate of absorption of a
local anesthetic into the bloodstream is affected by the dose administered, the
vascularity at the site of injection, and the specific physicochemical
properties of the drug itself. Local anesthetics gain entrance into the
bloodstream by ab-sorption from the injection site, direct intravenous
in-jection, or absorption across the mucous membranes af-ter topical
application. Direct intravascular injection occurs accidentally when the needle
used for infiltration of the local anesthetic lies within a blood vessel, or it
oc-curs intentionally when lidocaine is used for the control of cardiac
arrhythmias.
All tissues will be exposed
to local anesthetics after absorption, but concentrations will vary among the
or-gans. Although the highest concentrations appear to oc-cur in the more
highly perfused organs (i.e., brain, kid-ney, and lung), factors such as degree
of protein binding and lipid solubility also affect drug distribution. The lung
can absorb as much as 90% of a local anesthetic during the first pass.
Consequently, the lungs can act as a buffer to prevent higher and therefore
more toxic concentrations.
Placental transfer of local
anesthetics is known to occur rapidly, fetal blood concentrations generally
re-flecting those found in the mother. However, the quan-tity of drug crossing
to the fetus is also related to the time of exposure, that is, from the time of
injection to delivery. Subtle neurobehavioral changes in the neonate are
detectable for as long as 8 hours after mepivacaine administration to the
mother but are absent following the use of bupivacaine, lidocaine, and
chloroprocaine. In general, minimal amounts of chloroprocaine reach the fetus
because of its rapid hydrolysis by serum cholinesterase; this feature is its
principal advantage in obstetrics.
Metabolism
The metabolic degradation of
local anesthetics depends on whether the compound has an ester or an amide
link-age. Esters are extensively and rapidly metabolized in plasma by
pseudocholinesterase, whereas the amide link-age is resistant to hydrolysis.
The rate of local anesthetic hydrolysis is important, since slow
biotransformation may lead to drug accumulation and toxicity. In patients with
atypical plasma cholinesterase, the use of ester-linked compounds, such as
chloroprocaine, procaine and tetracaine, has an increased potential for
toxicity. The hy-drolysis of all ester-linked local anesthetics leads to the
formation of paraaminobenzoic acid (PABA), which is known to be allergenic.
Therefore, some people have al-lergic reactions to the ester class of local
anesthetics.
Local anesthetics with an
amide linkage (and one ester-lined anesthetic, cocaine) are almost completely
metabolized by the liver before excretion. However, the total dose administered
and the degree of drug accu-mulation resulting from the initial and subsequent
doses are still a concern.
Clinical Uses
Local anesthetics are
extremely useful in a wide range of procedures, varying from intravenous
catheter inser-tion to extensive surgery under regional block. For mi-nor
surgery, the patients can remain awake; this is an advantage in emergency
surgery, because protective air-way reflexes remain intact. Many operative
procedures in the oral cavity are facilitated by regional block of specific
nerves. If surgery permits, the patient can return home, because he or she is
less sedated than would be the case after general anesthesia.
Topical Anesthesia
Local anesthetics are used
extensively on the mucous membranes in the nose, mouth, tracheobronchial tree,
and urethra. The vasoconstriction produced by some lo-cal anesthetics, cocaine
especially, adds a very important advantage to their use in the nose by
preventing bleed-ing and inducing tissue shrinkage. Topical anesthesia permits
many diagnostic procedures in the awake pa-tient, and when it is combined with
infiltration tech-niques, excellent anesthesia may be obtained for many surgical
procedures in the eye and nose. The practi-tioner should be cautious when
higher volumes are re-quired, since overdosage may cause systemic reactions.
Additionally, when the tracheobronchial tree and lar-ynx are anesthetized,
normal protective reflexes, which prevent pulmonary aspiration of oral or
gastric fluids and contents, are lost.
Infiltration
Infiltration (i.e., the
injection of local anesthetics under the skin) of the surgical site provides
adequate anesthe-sia if contiguous structures are not stimulated. Since the
onset of local anesthesia is rapid, the surgical proce-dures can proceed with
little delay. Minimally effective concentrations should be used, especially in
extensive procedures, to avoid toxicity from overdosage.
Regional Block
Regional block, a form of
anesthesia that includes spinal and epidural anesthesia, involves injection
near a nerve or nerve plexus proximal to the surgical site. It provides
excellent anesthesia for a variety of proce-dures. Brachial plexus block is
commonly used for the upper extremity. Individual blocks of the sciatic,
femoral, and obturator nerves can be used for the lower extremity. An amount
that is close to the maximally tol-erated dose is required to produce blockade
of a major extremity.
Spinal Anesthesia
Spinal anesthesia
(subarachnoid block) produces exten-sive and profound anesthesia with a minimum
amount of drug. The local anesthetic solution is introduced di-rectly into the
spinal fluid, where the nerves are not pro-tected by a perineurium. This
produces, in effect, a tem-porary cord transection such that no impulses are
transmitted beyond the level that is anesthetized. The onset is rapid, and with
proper drug selection, the anes-thesia may last 1 to 4 hours. With careful
technique, neu-rological complications are extremely rare. Procedures as high
as upper-abdominal surgery can be performed under spinal anesthesia. Arterial
hypotension produced by the local anesthetic is proportional to the degree of
interruption of sympathetic tone, and it can produce pooling of blood in the
lower extremities, which leads to decreased cardiac filling pressures. Knowing
this allows blood pressure to be easily controlled, and hypotension is not
usually a deterrent to spinal anesthesia. The sites of action of spinal
anesthesia are the spinal nerve roots, spinal ganglia, and (perhaps) the spinal
cord.
Lumbar Epidural Anesthesia
Lumbar epidural anesthesia
affects the same area of the body as does spinal anesthesia. As the name
implies, the drug is deposited outside the dura. In contrast to spinal
anesthesia, this method requires a much larger amount of drug. This procedure
makes segmental anesthesia possi-ble, whereby the anesthetized area is bordered
caudally and cephalad by unaffected dermatomes and myotomes.
The concentration and volume
of the local anes-thetic solution will affect the extent of the cephalad and
caudad spread of the block. The anesthesia can be made continuous by
maintaining a small catheter in the epidural space; prolonged effects are
obtained by peri-odically injecting supplemental doses through the catheter or
by attaching it to a computer pump. The site of anesthetic action is on the
nerves as they leave the in-tervertebral foramina. However, effective drug
concen-trations may be found in the spinal fluid, probably gain-ing entrance
through the arachnoid villi. Arterial hypotension occurs by the same mechanism
and is man-aged as in spinal anesthesia.
Epidural anesthesia is
especially useful in obstetrics. Excellent analgesia occurs and the patient
remains awake. Analgesia by the epidural route can be provided for labor and
delivery or for cesarean section. Bupivacaine in lower concentrations has the
advantage of providing excellent analgesia while minimally reduc-ing motor
strength.
Caudal Anesthesia
In the caudal form of
extradural anesthesia, the agent is introduced through the sacral hiatus above
the coccyx. It is particularly applicable to perineal and rectal proce-dures.
Anesthetization of higher anatomical levels is not easily obtained, because the
required injection volume can be excessive. Although caudal anesthesia has been
used extensively in obstetrics, lumbar epidural blockade is now more commonly
used because of the lower dose of drug required; in addition, the sacral
segments are spared until their anesthesia is required for the delivery.
Intravenous Extremity Block
Excellent and rapid
anesthetization of an extremity can be obtained easily. Following insertion of
an intra-venous catheter in the limb of interest, a rubber band-age is used to
force blood out of the limb, and a tourni-quet is applied to prevent the blood
from reentering; a dilute solution of local anesthetic, most commonly
lido-caine, is then injected intravenously. This technique fills the limb’s
vasculature and carries the anesthetic solu-tion to the nerve by means of the
blood supply. Because of the pain produced by a tourniquet after some time,
this procedure usually is limited to less than 1 hour. The systemic blood
levels of drug achieved after tourniquet release generally remain below toxic
levels.
Although it is more easily
and therefore more com-monly used on the upper extremity, intravenous
ex-tremity anesthesia can be used on the leg and thigh.
Sympathetic Block
Blockade of the sympathetic
nervous system can be more selectively accomplished than that which occurs
during spinal or epidural anesthesia. Cell bodies for pre-ganglionic
sympathetic nerves originate in the interme-diolateral cell column of the
spinal cord, from the first thoracic to the second lumbar segments. The
myelinated axons of these cells travel as white communicating rami before
joining the sympathetic chain and synapsing in the ganglia. The best location
for a sympathetic block is at the sympathetic ganglia, since a block at this
level will affect only the sympathetic nerves. For example, local anesthetic
blockade of the stellate ganglion (which in-cludes T1) blocks sympathetic
innervation to all of the upper extremity and head on the injected side. A
block of the sympathetic chain at L2 affects all of the lower ex-tremity. This
form of local anesthesia is particularly use-ful during treatment of a variety
of vasospastic diseases of the extremities and for some pain syndromes.
Control of Cardiac Arrhythmias
Procainamide and lidocaine
are two of the primary drugs for treating cardiac arrhythmias. Since lidocaine
has a short duration of action, it is common to adminis-ter it by continuous
infusion. Procainamide, because of its amide linkage, has longer action than
does its pre-cursor, procaine. Orally active analogues of local anes-thetics
(e.g., mexiletine) also are used as antiarrhyth-mics .
Use of Vasoconstrictors
The most commonly used
vasoconstrictors, the sympa-thomimetic drugs, are often added to local
anesthetics to delay absorption of the anesthetic from its injection site. By
slowing absorption, these drugs reduce the anes-thetic’s systemic toxicity and
keep it in contact with nerve fibers longer, thereby increasing the drug’s
dura-tion of action. Administration of lidocaine 1% with ep-inephrine results
in the same degree of blockade as that produced by lidocaine 2% without the
vasoconstrictor.
Many vasoconstrictors are
available, but epineph-rine is by far the most commonly employed. Because
epinephrine can have systemic α- and β-adrenergic ef-fects, precaution is needed when local anesthetics
containing this amine are given to a patient with hyper-tension or an irritable
myocardium. Sensitivity to epi-nephrine may be incorrectly diagnosed as an
allergy to local anesthetics. Epinephrine-containing solutions should be used
cautiously in persons taking tricyclic an-tidepressants or monoamine oxidase
(MAO) inhibitors, since those drugs may enhance the systemic pressor ef-fects
of sympathomimetic amines.
Levonordefrin (Neo-Cobefrin) is an active optical
isomer of nordefrin that has α1-adrenergic activity and possesses little or no -agonist
properties. It is used ex-clusively in some dental anesthetic cartridges as a
vaso-constrictor. Its theoretical advantage is that it causes less hypertension
and tachycardia than does epinephrine.
Phenylephrine hydrochloride (Neo-Synephrine) is a pure -agonist that
is occasionally used for subarach-noid block and is marketed with procaine for
use in dentistry. It has little direct cardiac effect.
Adverse Effects
The central nervous and
cardiopulmonary systems are most commonly affected by high plasma levels of
local anesthetics. Local anesthetics given in initially high doses produce
central nervous system (CNS) stim-ulation characterized by restlessness,
disorientation, tremors, and at times clonic convulsions. Continued ex-posure
to high concentrations results in general CNS depression; death occurs from
respiratory failure sec-ondary to medullary depression. Treatment requires
ventilatory assistance and drugs to control the seizures. The
ultra–short-acting barbiturates and the benzodi-azepine derivatives, such as
diazepam, are effective in controlling these seizures. Respiratory stimulants
are not effective. CNS manifestations generally occur be-fore cardiopulmonary
collapse.
Cardiac toxicity is generally the result of drug-induced depression
of cardiac conduction (e.g., atrioven-tricular block, intraventricular conduction block)
and systemic vasodilation. These effects may progress to se-vere hypotension
and cardiac arrest.
Allergic reactions, such as
red and itchy eczematoid dermatitis or vesiculation, are a concern with the ester-type
local anesthetics. True allergic manifestations have been reported with
procaine. The amides are essentially free of allergic properties, but
suspected allergic phenom-ena may be caused by methylparaben, a
parahydroxyben-zoic acid derivative used as an antibacterial preservative in
multiple-dose vials and some dental cartridges. Esters probably should be
avoided in favor of an amide when the patient has a history of allergy to a
PABA-containing preparation such as certain cosmetics or sunscreens.
Related Topics