Chapter: Obstetrics and Gynecology: Puberty
Precocious Puberty
Precocious Puberty
Precocious puberty is the onset
of secondary sexual charac-teristics prior to the age 6 in black girls and age
7 in white
Precocious puberty is caused by either GnRH-dependentor GnRH-independent
sex hormone production (Box 34.1).GnRH-dependent, or true (central)
precocious puberty, develops secondary to early activation of the hypothalamic–
pituitary–gonadal axis. The most common causes are idiopathic; other causes
include infection, inflammation, or injury of the central nervous system. In
idiopathic pre-cocious puberty, the arcuate nucleus in the hypothalamus is
prematurely activated. This causes early sexual maturation with early
reproductive capability. The elevated estrogen levels affect the skeleton,
resulting in short stature in adult-hood secondary to premature closure of the
epiphyseal plates. These individuals are at risk for sexual abuse and have
psychosocial problems related to their early sexual develop-ment. Occasionally,
GnRH-dependent precocious puberty results from neoplasms of the
hypothalamic–pituitary stalk. In this situation, although sexual development
begins early, the rate of sexual development is slower than usual. Transient
inflammatory conditions of the hypothalamus may also result in GnRH-dependent
precocious puberty; however, sexual development may begin and end abruptly.
Laboratory studies show either an appropriate rise in go-nadotropins or a
steady gonadotropin level in the prepuber-tal range.

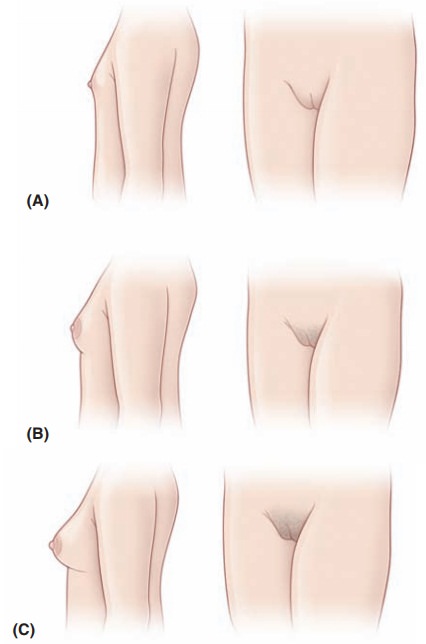
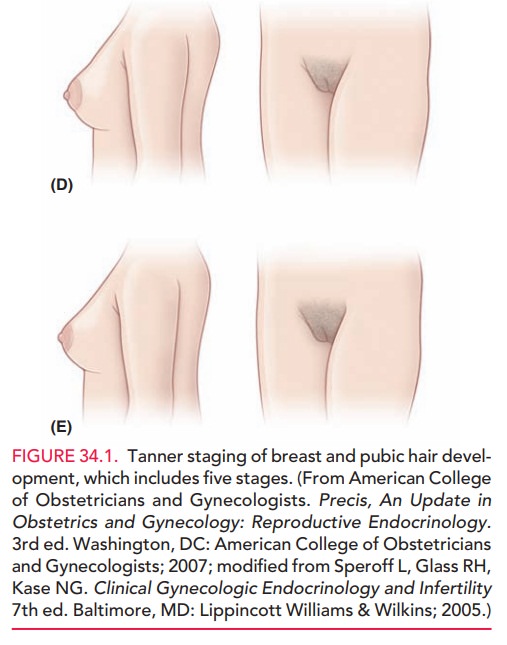
GnRH-independent
sex hormone production, or precocious pseudopuberty (peripheral), results from
sex hormone production (androgens or estrogens) independent of
hypothalamic–pituitary stimulation. This
condition can be caused by ovarian cystsor tumors, McCune–Albright syndrome,
adrenal tumors, or iatrogenic causes. Some tumors, such as granulosa cell
tumors, teratoma, or dysgerminomata, directly secrete androgen. Physical
examination usually reveals a palpable pelvic mass and leads to further
evaluation/imaging studies.
McCune–Albright
syndrome (polyostotic fibrous dysplasia) is characterized by multiple bone
fractures, café-au-lait spots, and precocious puberty. Premature
menarche can be the first sign
The syndrome is
thought to result from a defect in cellular regulation with a mutation in the
alpha subunit of the G protein that stimulates cAMP formation, which causes
affected tissues to function autonomously. This mutation causes the ovary to
produce estrogen with-out the need for FSH, resulting in sexual precocity.
Adrenal
causes of precocious puberty include adrenal tumors or enzyme-secreting
defects, such as congenital adrenal hyperpla-sia (CAH). Tumors
are very rare and must secrete estrogento cause early sexual maturation. The
most common form of CAH, 21-hydroxylase deficiency, presents at birth with the
finding of ambiguous genitalia. However, the nonclassical form, previously
known as late-onset CAH, tends to present at adolescence. In this disorder, the
adrenal glands are unable to produce adequate amounts of cortisol as a result
of a partial block in the conversion of 17-hydroxyprogesterone to
deoxycortisol. Deficiency of the 21-hydroxylase enzyme leads to a shunting away
from aldosterone and cortisol production in cholesterol biosyn-thesis toward
the production of androgens (testosterone and estradiol), which results in
precocious adrenarche. Apathognomonic
finding for 21-hydroxylase deficiency is an elevated 17-hydroxyprogesterone
level. Plasma renin is also measured todetermine the amount of
mineralocorticoid deficiency. Medical therapy is instituted as early as
possible and is aimed at steroid/mineralocorticoid replacement, depending on
the severity of the deficiency. In the nonclassical form of CAH, patients
present with premature adrenarche, anovulation, and hyperandrogenism, appearing
somewhat like patients with polycystic ovarian syndrome.
Iatrogenic
causes such as drug ingestion must be considered in all children who present
with precocious puberty.
These children may exhibit
increased pigmentation of the nipples and areola of the breast secondary to
ingestion of oral contraceptives, anabolic steroids, and hair or facial creams.
The main
goals of treatment of precocious puberty are to ar-rest and diminish sexual
maturation until a normal pubertal age, as well as to maximize adult height. Therapy
for GnRH-independent precocious puberty involves administration of a GnRH
agonist. Results occur rapidly and continue during the first year of treatment.
Treatment for GnRH-independent precocious puberty attempts to suppress go-nadal
steroidogenesis.
Related Topics