Chapter: Medical Microbiology: An Introduction to Infectious Diseases: Pneumocystis carinii
Pneumocystosis
PNEUMOCYSTOSIS
Pneumocystis pneumonia is insidious, beginning with mild fever or malaise in individuals whose immune system is compromised. Signs referable to the lung come later with nonproductive cough and shortness of breath. Radiographs reveal symmetrical alveolar pulmonary infiltrates, which spread from the hili. Progressive cyanosis, hypoxia, and asphyxia can lead to death in a 3- to 4-week period.
EPIDEMIOLOGY
Pulmonary infection with P. carinii occurs worldwide in humans and a broad spectrum of animal life. Exposure must be common; specific antibodies are present in nearly all chil-dren by the age of 4. The reservoir and mode of transmission remain unknown, but the view that the majority of Pneumocystis carinii pneumonia (PCP) cases represent reactiva-tion of latent infection is no longer held. P. carinii is not found in the respiratory tract of asymptomatic persons, even among HIV-infected individuals, and the strains involved in second and third episodes are frequently antigenically different. Animal studies have shown that airborne transmission is possible, and the circumstances of hospital outbreaks point to active cases as a probable source.
Before the acquired immunodeficiency syndrome (AIDS) pandemic, PCP occurred sporadically among infants with congenital immunodeficiencies and in older children and adults as a complication of immunosuppressive therapy. Now AIDS has become the most common predisposing condition in the United States, and PCP is often the presenting manifestation of AIDS. In fact, prior to the development of effective chemoprophylactic regimens (see Treatment and Prevention), it was present in approximately half of all AIDS patients at the time of initial diagnosis. Eventually, most AIDS patients develop one or more bouts of PCP, often in conjunction with another opportunistic infection.
PATHOGENESIS
P. carinii is an organism of low virulence that seldom produces disease in a host with nor-mal T-lymphocyte function. In experimental animals, progressive infection can be initi-ated with starvation or corticosteroid administration, and in AIDS patients the risk of developing pneumocystosis increases dramatically once the CD4+ T lymphocyte count has fallen to 200 cells/mm3 or below. Concurrent viral, bacterial, fungal, and protozoan infections are found frequently in human cases, suggesting that P. cariniimay require the presence of another microbial agent for its multiplication.
Little is known about the early stages of disease. A major surface glycoprotein (MSG) abundant on the surface of P. carinii may act as an attachment ligand to several host proteins, including fibronectin, vitronectin and surfactant proteins.
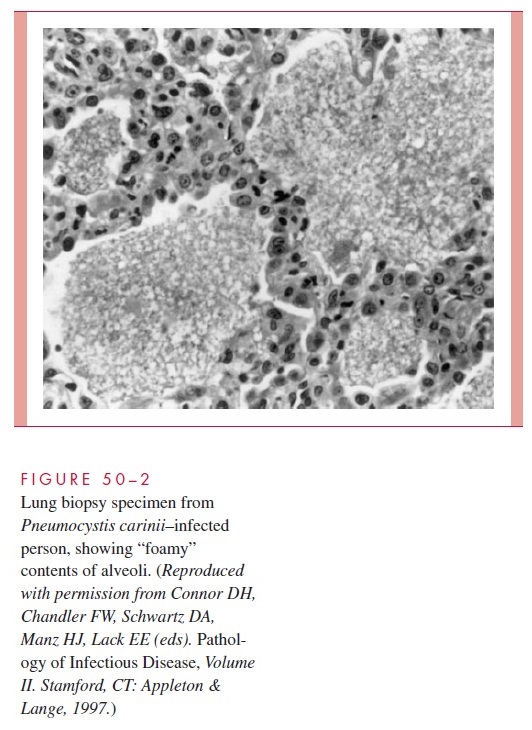
MSG undergoes antigenic variation, which could aid in its persistence in human hosts. Histologically, PCP is characterized by alveoli filled with desquamated alveolar cells, monocytes, organisms, and fluid, producing a distinctive foamy, honeycombed appearance (Fig 50–2); hyaline membranes may be present, and round cell infiltrates may be visible in the septa. It has been suggested that P. carinii maintains an extracellular existence within alveoli obtain-ing essential nutrients from the alveolar fluid and lining cells.
IMMUNITY
The nature of the immunodeficiencies in patients with pneumocystosis points to the pri-macy of cell-mediated immunity (CMI) in resolution of infection with P. carinii.Alveolar macrophages are the first line of defense, with activated macrophages and CD4 lym-phocytes playing essential roles in the resolution of the infection. Activated macrophages release several cytotoxic factors, including O2-derived radicals, reactive nitrogen interme-diates, and cytokines (tumor necrosis factor- , interleukin-2).
Specific antibody responses to the MSG and other antigens appear in the course of pneumocystosis. A significant role for humoral immunity is suggested by the ability of MSG antibody to protect against experimental PCP in animals.
Related Topics