Chapter: Medical Immunology: Phagocytic Cells
Physiology of the Polymorphonuclear Leukocytes
PHYSIOLOGY OF THE POLYMORPHONUCLEAR LEUKOCYTES
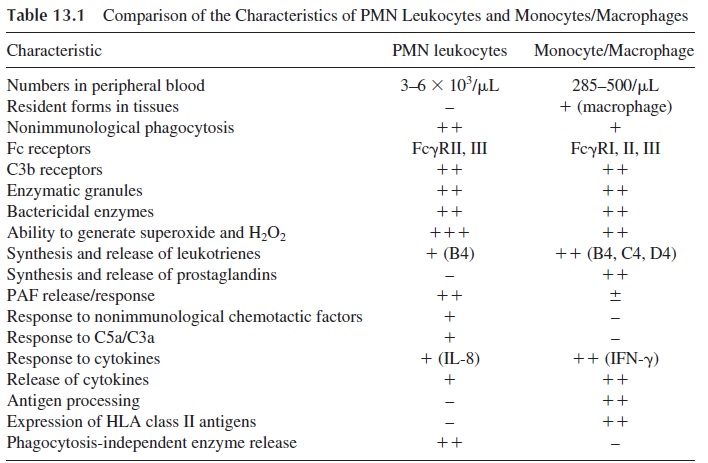
Neutrophils and other polymorphonuclear (PMN) leukocytes are “wandering” cells, constantly circulating around the vascular network, able to recognize foreign matter by a wide variety of immunological and nonimmunological mechanisms. Their main biological char-acteristics are summarized in Table 13.1. Their effective participation in an anti-infectious response depends on the ability to respond to chemotactic signals, ingest the pathogenic agent, and kill the ingested microbes.
A. Chemotaxis and Migration to the Extravascular Compartment
In normal conditions, the interaction between leukocytes and endothelial cells is rather loose and involves a family of molecules known as selectins, which are constitutively expressed on endothelial cells and glycoproteins expressed on the leukocyte cell membrane.
There interactions cause the slowing down (“rolling”) of leukocytes along the vessel wall but do not lead to firm adhesion of leukocytes to endothelial cells.
A variety of chemotactic stimuli can be involved in the recruitment of leukocytes to the extravascular space. In most cases those chemotactic factors are of bacterial origin, but they can also be released as a consequence of tissue necrosis, as a result of monocyte and lymphocyte activation, or as a by-product of complement activation. Among bacterial products, formyl-methionyl peptides, such as f-methionine-leucine-phenylalanine (f-met-leu-phe), are extremely potent chemotactic agents.
Complement-derived chemotactic factors can be generated in several ways. Tissue damage may result in the activation of the plasmin system, which may in turn initiate complement activation with generation of C5a. After the inflammatory process has been established, proteases released by activated neutrophils and macrophages can also split C5, and the same cells may release leukotriene B4, another potent chemotactic factor, at-tracting more neutrophils to the site. On the other hand, many microorganisms can gen-erate C5a by activation of the complement system through the alternative pathway. Fi-nally, activated T cells and monocytes can also release chemokines such as IL-8, monocyte chemotactic protein-1, and RANTES, which have neutrophils and/or mono-cytes as targets.
After receiving a chemotactic stimulus, the neutrophil undergoes changes in the cell membrane, which is smooth in the resting cell, and becomes “ruffled” after the cell receives the chemotactic signal. he activated PMN leukocyte has a marked increase in cell adhe-siveness, associated with increased expression of adherence molecules, namely integrins of the CD11/CD18 complex, which include:
1. CD11a [the chain of LFA (leukocyte function antigen)-1]
2. CD11b (the C3bi receptor or CR3, also known as Mac-1)
3. CD11c (also known as protein p150,95)
4. CD18 [the chain of LFA (leukocyte function antigen)-1]
These cell adhesion molecules (CAM) are common to the majority of leukocytes, but their individual density and frequency may vary in the two main groups of phagocytic cells. While CD11a and CD18 are expressed virtually by all monocytes and granulocytes, CD11b is more prevalent among granulocytes and CD11c is more frequent among monocytes.
The expression of these CAM mediates a variety of cell-cell interactions, such as those that lead to neutrophil aggregation and, most importantly, those that mediate firm ad-hesion of neutrophils to endothelial cells. For example, CD11a (LFA-1) and CD11b inter-act with molecules of the immunoglobulin gene family, such as ICAM-1, ICAM-2, and VCAM-1, expressed on the endothelial cell membrane. The expression of VCAM-1 and, to a lesser degree, of ICAM-1 and -2 is also upregulated by cytokines released by activated monocytes and lymphocytes, such as IL-1 and TNF. Consequently, the adhesion of leuko-cytes to endothelial cells is further enhanced.
After adhering to endothelial cells, leukocytes migrate to the extravascular compart-ment. The transmigration involves interaction with a fourth member of the immunoglobu-lin gene family—platelet endothelial cell adhesion molecule 1 (PECAM-1)—which is ex-pressed at the intercellular junctions between endothelial cells. The interaction of leukocytes with PECAM-1 mediates the process of diapedesis, by which leukocytes squeeze through the endothelial cell junctions into the extravascular compartment.
The diapedesis process involves the locomotor apparatus of the neutrophils, a con-tractile actin-myosin system stabilized by polymerized microtubules. Its activation is es-sential for the neutrophil to move into the extravascular space, and an intact CD11b protein seems essential for the proper modulation of microtubule assembly, which will not take place in CD11b-deficient patients.
B. Phagocytosis
At the area of infection, PMN leukocytes recognize the infectious agents, which are in-gested and killed intracellularly. The sequence of events leading to opsonization and intra-cellular killing is summarized in Figure 13.1.
Several recognition systems appear to be involved in the phagocytosis step. The most important recognition systems are those that mediate the ingestion of opsonized particles. Two major types of receptors expressed by phagocytic cells are involved in this process:
1. Fcg receptors are predominantly involved in promoting ingestion of antibody-coated particles. Neutrophils express two types of Fcγ receptors, FcγRII and FcγRIII, both of which are involved in phagocytosis. In experimental conditions, Fca receptors may also be involved in phagocytosis, but their efficiency seems to be much lower than that of Fcγ receptors.
2. The CR1 (C3b) receptor is also able to mediate phagocytosis with high efficiency. This receptor is expressed by all phagocytic cells, including polymor-phonuclear leukocytes, monocytes, and macrophages. The binding and ingestion of microorganisms through this receptor has been well established.
Opsonization with both IgG antibodies and C3b seems associated with maximal ef-ficiency in ingestion.
Opsonization is not an absolute requirement for ingestion by neutrophils. A variety of receptors may be involved in nonimmune phagocytosis.
These nonimmune mechanisms are particularly effective in promoting the in-gestion of microorganisms with polysaccharide-rich outer layers. In addition, neutrophils are also able to ingest a variety of particulate matter, such as latex beads, silicone, asbestos fibers, etc., in the absence of opsonizing antibodies or complement.
C. Intracellular Killing
No matter the nature of the receptors that may mediate it, ingestion is achieved through for-mation of pseudopodia that surround the particle or bacteria, eventually fusing at the distal pole to form a phagosome. The cytoplasmic granules of the neutrophil (lysosomes) then fuse with the phagosomes, and their contents empty inside the phagosomes (degranulation). This degranulation process is very rapid and delivers a variety of antimicrobial substances to the phagosome:
· The azurophilic or primary granules contain, among other substances, myeloperoxi-dase, lysozyme, acid hydrolases (such as β-glucuronidase), cationic proteins, defensins, metalloproteinases (including proteases and collagenases), elastase, and cathepsin C2.
· The secondary granules or lysosomes contain lysozyme and lactoferrin.
Killing of ingested organisms depends on the effects of cationic proteins from the pri-mary granules, lysosomal enzymes, such as lysozyme and lactoferrin, defensins, nitric ox-ide, and by-products of the respiratory burst, activated as a consequence of phagocytosis.
Cationic proteins bind to negatively charged cell surfaces (such as the bacterial outer membrane) and interfere with microbial growth. Lactoferrin has antimicrobial activity by chelating iron and preventing its use by bacteria that need it as an essential nutrient.
Lysozyme splits the β-1,4 linkage between the N-acetylmuramic acid peptide and N-acetylglucosamine on the bacterial peptidoglycan. Some bacteria are exquisitely sensitive to the effects of this enxyme, which causes almost immediate lysis. However, the impor-tance of this enzyme as a primary killing mechanism has been questioned due to the rela-tive inaccessibility of the peptidoglycan layer in many microorganisms, which may be sur-rounded by capsules or by the lipopolysaccharide-rich outer membrane (gram-negative bacteria).
Defensins are antimicrobial peptides released by almost all eukaryotic species, in-cluding plants, invertebrate animals, and vertebrate animals. Structurally, defensins are cationic molecules with spatially separated hydrophobic and charges regions, which insert themselves into phospholipid membranes, causing their disruption. In mammals, defensins are produced by specialized mucosal cells (i.e., the Paneth cells in the gut) and by phago-cytic cells. The mucosal defensins are believed to play an important role protecting mu-cosal cells from pathogenic bacteria. The neutrophil defensins are packaged on the azurophilic granules and are delivered to the phagosomes and also spilled into the extra-cellular environment.
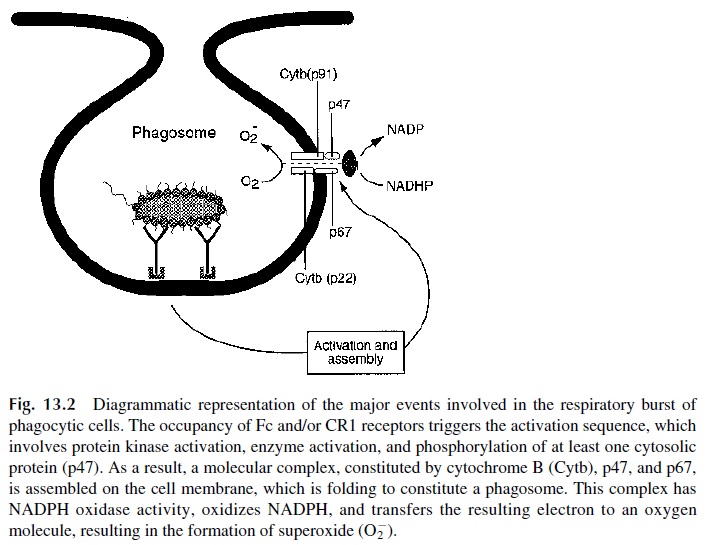
From a bactericidal point of view, the activation of the superoxide generating system (respiratory burst) appears considerably more significant. This system is activated primar-ily by opsonization, but also by a variety of PMN leukocyte–activating stimuli, ranging from f-met-leu-phe to C5a. the activating stimuli are responsible for the induction of a key enzymatic activity (NADPH oxidase), a molecular complex located on the cell membrane responsible for the transfer of a single electron from NADPH to oxygen, generating super-oxide (O2- ).
The molecules responsible for NADPH oxidase activity are:
· Cytochrome B244, which is a heterodimer formed by two polypeptide chains (91 and 22 kDa, respectively), believed to play the key role in the reduction of oxygen to superoxide, possibly by being the terminal electron donor
· Two cytosolic proteins—p47 and p67—one of which (p47) is a substrate for protein kinase C p21rac, an ubiquitous ras-related GTPase
· In a resting cell the complex is inactive and its components are not associated. After the cell is activated, p47 is phosphorylated and becomes associated with p67 and with p21rac. The phosphorylated complex binds to cytochrome B244 in the lysosomal mem-branes, forming what is considered to be the active oxidase.
The electron transfer from NAPDH to oxygen is believed to involve at least three steps:
1. Reduction of a flavin adenine dinucleotide (FAD), bound to the high molecular weight subunit of cytochrome B
2. Transfer of an electron from FADH2 to ferric iron in a heme molecule associated to the low molecular weight subunit of cytochrome B
3. Transfer of an electron from reduced iron to oxygen, generating superoxide
The formation of the active molecular complex with oxidase activity coincides with phagolysosome fusion. Thus, most of the active oxygen radicals generated by this system are delivered to the phagolysosome (Fig. 13.2).
The respiratory burst generates two toxic compounds essential for intracellular killing of bacteria: superoxide and H2O2. Through myeloperoxidase, H2O2 can be peroxi-dated and led to form hypochlorite and other halide ion derivatives, which are also potent bactericidal agents. These compounds are also toxic to the cell, particularly superoxide, which can diffuse into the cytoplasm. The cell has several detoxifying systems, including superoxide dismutase, which converts superoxide into H2O2, and in turn, H2O2 is detoxi-fied by catalase and by the oxidation of reduced glutathione, which requires activation of the hexose monophosphate shunt.
Activated phagocytic cells also express an inducible form of nitric oxide synthase (iNOS), which generates nitric oxide (NO) from arginine and molecular oxygen, using a variety of cofactors that include NADPH. Nitric oxide is a short-lived, highly cytotic free radical gas, which is believed to contribute significantly to intracellular killing. It can also participate in the induction of inflammatory reactions when spilled into the extracellular space.
Related Topics