Chapter: Modern Medical Toxicology: Neurotoxic Poisons: Somniferous Drugs
Opium: Diagnosis, Treatment, Autopsy Features, Forensic Issues - Somniferous Drugs(Narcotics) Neurotoxic Poisons
Diagnosis
·
Needle marks, dermal scars
(suggestive of addiction).
·
Evidence of hypoglycaemia, hypoxia,
and hypothermia.
·
Most opiates can be detected in
urine or blood by RIA, GC, GC-MS, or HPLC.
·
Empirical administration of
naloxone, (can precipitate reaction in
addicts).
Treatment
Acute Poisoning:
a.
Supportive measures—
–– Maintenance of patent airway.
–– Endotracheal intubation, assisted
ventilation: Maintain adequate ventilation and oxygenation with frequent
monitoring of arterial blood gases and/or pulse oximetry. If a high FIO2
is required to main- tain adequate oxygenation, mechanical ventilation and
positive-end-expiratory pressure (PEEP) may be required; ventilation with small
tidal volumes (6 ml/kg) is preferred if ARDS develops. Crystalloid solutions
must be administered judiciously. Pulmonary artery monitoring may help. In
general the pulmonary artery wedge pressure should be kept relatively low while
still maintaining adequate cardiac output, blood pressure and urine output.
–– Ipecac-induced emesis is not recommended
because of the potential for CNS depression and seizures.
–– Consider prehospital administration of
activated charcoal as an aqueous slurry in patients with a potentially toxic
ingestion who are awake and able to protect their airway. Activated charcoal is
most effec- tive when administered within one hour of ingestion.
b. Naloxone is the antidote of choice for opiate
poisoning,.
c. The use of physostigmine salicylate (0.04
mg/kg IV)has been suggested for reversing respiratory depression if the regular
opiate antidotes are not available, since it increases the acetylcholine
content of the reticular formation of the brainstem which is suppressed by
opiates. However, there is controversy regarding such a measure since
physostigmine (unlike regular opiate antagonists) is a dangerous antidote with
serious adverse effects.
Antidotes for opiates
Naloxone—
It is effective against all opiates including pentazocine, but is not
very effective against buprenorphine. Dramatic reversal of the following
features is achieved: miosis, respiratory depression, hypotension, coma.
Dose: The usual initial dose is 1.2 mg for an adult and 0.4 mg for a
child. The best route of administration is IV, but if venous access is
difficult, the drug can be injected sublingually or intramuscularly, or even
instilled down an endotracheal tube. Since the effect of a single bolus dose of
naloxone is usually short-lived, repeated doses are required. Repeat doses of 2
mg may be given to achieve a clinical effect. Generally, if no response is
observed after 10 mg has been administered, the diagnosis of opiate-induced
toxicity should be questioned. Very large doses of naloxone (10 mg or more) may
be required to reverse the effects of buprenorphine overdose. Some
investigators state that a naloxone infusion is better than repeated
injections. The following steps are suggested:
·
Determine the maintenance fluid requirement for
24 hours.
·
To determine the amount of naloxone (in mg) to
add to the maintenance fluid for a 24-hour period, take the bolus dose required
for initial response (in mg) and multiply by 2/3 and 24 (hrs).
·
To determine the desired rate of infusion
(ml/hr), take the maintenance fluid, and divide by 24 (hrs)
This method is said to reduce the risk of possible fluid overload and
pulmonary oedema. A continuous infusion of naloxone is espe-cially useful in
circumstances of opiate overdose with long acting opiates. Naloxone can be
diluted in normal saline or 5% dextrose, but should not be added in alkaline
solutions. Any prepared solution of naloxone should be used up in 24 hours.
Caution should be exercised in reversing opiate toxicity in addicts because of
the risk of precipitating withdrawal reaction. Observe patients for evidence of
CNS or respiratory depression for at least 2 hours after discontinuing the
naloxone infusion.
The American Academy of Pediatrics recommends a neonatal dose of 0.1
mg/kg intravenously or intratracheally from birth until age 5 years or 20 kg
body weight.
Naltrexone—
Naltrexone is a long-acting opiate antagonist which can be administered
orally. It is usually used for treating opiate addiction. However, it must not
be given to an opiate-dependant patient who has not been detoxified. A
challenge dose of naloxone to confirm the lack of opiate dependence is
recommended before beginning naltrexone.
Dose: 50 mg/day orally, which may have to be continued for several weeks
or months.
Nalmefene—
Nalmefene is a naltrexone derivative with pure opiate antagonistic
effects, and has a longer duration of effect than naloxone in acute opiate
poisoning. It is usually given intravenously beginning with 0.1 mg, and if
withdrawal reaction does not occur, 0.5 mg is admin-istered, followed by 1 mg
in 2 to 5 minutes (if necessary). Nalmefene can also be given intramuscularly
or subcutaneously
d. The use of drugs such as levorphanol and amiphena-zole is
no more recommended today.
e. Convulsions may be treated with benzodiazepines in the
usual manner (5 to 10 mg initially, repeat every 5 to 10 minutes as needed),
though this is frequently not necessary if naloxone is available. Monitor for
respira-tory depression, hypotension, arrhythmias, and the need for
endotracheal intubation. Evaluate for hypoxia, electrolyte disturbances, and
hypoglycaemia (or treat with intravenous dextrose 50 ml IV in an adult, or 2
ml/ kg in 25% dextrose for a child).
f.
For hypotension: Infuse 10 to 20 ml/kg of
isotonicfluid and place in Trendelenburg position. If hypoten-sion persists,
administer dopamine (5 mcg per kg per min, progressing in 5 mcg per kg per min
increments as needed), or noradrenaline (0.5 to 1 mcg per min, and titrate to
maintain adequate blood pressure). Consider central venous pressure monitoring
to guide further fluid therapy.
g. Prevention
of rhabdomyolysis: Early aggressivefluid replacement is
the mainstay of therapy and may help prevent renal insufficiency. Diuretics
such as mannitol or furosemide may be needed to maintain urine output. Urinary
alkalinisation is not routinely recommended.
Chronic Poisoning:
·
Gradual withdrawal of the opiate.
·
Substitution therapy with methadone
begun at 30 to 40 mg/day and then gradually tapered off.
·
A beta adrenergic blocker like
propranolol (80 mg) is said to be quite effective in relieving the anxiety and
craving associated with opiate addiction, but has no effect on physical
symptoms. Alternatively, drugs such as clonidine can be used. Buprenorphine or
naltrexone can also be used. Recent reports suggest favourable outcome with
gabapentin combined with clonidine and naltrexone. The regimen suggested is
clonidine 0.1 mg thrice a day for 7 days, followed by naltrexone 50 mg twice a
day for 14 days, along with gabapentin 600 mg twice a day on all the 21 days.
·
Antispamodics for abdominal cramps
associated with vomiting and diarrhoea.
·
Tranquillisers or bed time sedation
if necessary.
·
Psychiatric counselling.
Autopsy Features
·
Injection marks, dermal abscesses,
scarring. Look for injec-tion marks in the antecubital fossae, forearms, back
of the hands, neck, groin, and ankles.
·
Tattooing, (a common feature of the
drug sub-culture).
· Emaciation, unkempt appearance.
· Gross pulmonary oedema with froth
exuding out of mouth and nostrils, especially in sudden heroin-related death.
·
Another frequent autopsy finding in
heroin fatalities is undiagnosed pneumonia.
·
Autopsy findings of heroin-induced
spongiform leukoen-cephalopathy include spongiform degeneration of white
matter, vacuolisation and fluid accumulation in myelin sheaths.
·
Cerebral oedema.
·
Congestion of liver with enlargement
of hepatic lymph nodes. Chemical analysis of lymph nodes may reveal pres-ence
of morphine.*
·
Myocardial damage, with focal
lesions formed by small mono-nuclear inflammatory cells and with degenerated,
necrotic myocardial fibres and congestion, has been shown to occur as a result
of prolonged hypoxic coma following opiate intoxica-tion.
·
Samples of blood, urine, brain,
liver, and bile must always be preserved for chemical analysis.
·
It is important to remember that
infections such as serum hepa-titis and AIDS are common among intravenous drug
abusers, and hence autopsies conducted in drug-related deaths must be done
cautiously with necessary precautions.
Forensic Issues
· Opiates are among the commonest of
the drugs abused today in India. Heroin (brown
sugar) is popular among all classes of addicts in metropolitan cities such
as Mumbai, Delhi, Bangalore, etc. Opiates used for therapeutic purposes, e.g.
morphine, pethidine, and pentazocine are more commonly abused by medical and
paramedical personnel. Buprenorphine is emerging as a major drug of abuse even
among non-medical personnel in recent times. Codeine which is easily available
over the counter in the form of antitussive preparations is being increasingly
abused especially by college-going youth.
· Accidental deaths are not infrequent
from overdose, particu-larly among intravenous abusers of heroin (“death on the
needle”). Wound botulism, which has been associated with subcutaneous or
intramuscular black tar heroin injections, has caused potentially lethal,
descending, flaccid paralysis when Clostridium
botulinum spores germinated in wounds, releasing neurotoxins.
· “Cotton fever” is common among IV
abusers, which is a febrile reaction that develops because these drugs are
often filtered through cotton balls prior to being injected.
·
Chronic parenteral opiate abuse can result in abscesses,
acute transverse myelitis, anaphylaxis, AIDS, arrhythmias, wound botulism,
cellulitis, endocarditis, faecal impaction, glomerulonephritis, hyper or
hypoglycaemia, osteomyelitis, post-anoxic encephalopathy, tetanus,
thrombophlebitis, nephropathy, hoarseness, hepatitis, pneumothorax, para-plegia,
mycotic aneurysms, or leucoencephalopathy.
· Suicidal deaths are also reported
from time to time since opiates are reputed to cause a painless death.
Homicides are rare, but a few cases have been documented in medical literature.
· Body Packer Syndrome—Although this is more
commonlyassociated with smuggling of cocaine, it has also been reported in the
case of other drugs, especially heroin.
o
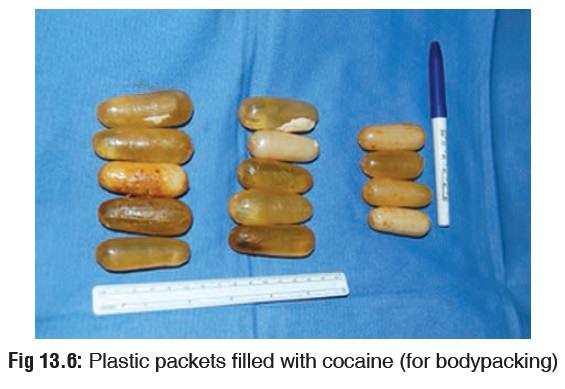
A “bodypacker” or “mule” is an individual who attempts to
transport illicit drugs from one country to another by ingesting wrapped
packages, or condoms (Fig 13.6), or
balloons containing concentrated cocaine or heroin. After arrival at the
destination, cathartics are self-administered and the packets are defaecated
out. Sometimes rectal suppositories or disposable enemas are used.
o
Although generally asymptomatic, in a few cases serious
toxicity may result due to rupture of packets. Death due to intestinal
obstruction and perforation has been reported in heroin body packers.
o
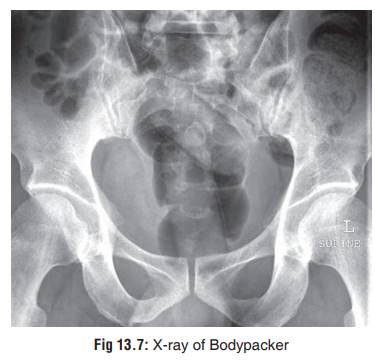
Diagnosis of an asymptomatic body packer can be accomplished
with the help of an abdominal X-ray (Fig13.7),
ultrasound, or CT scan. The last two methodsshould be resorted to only if X-ray
is unclear. In some cases, faecal examination over a period of a few days may
be necessary. Magnetic resonance does not visualise packets because of the lack
of protons.
o Asymptomatic patients should be treated by whole bowel irrigation with polythylene glycol solution. However some investigators do not approve of this method since the polythylene glycol can dissolve the heroin from a package, rupturing it and increasing absorption of heroin. Instead, a period of waiting is recommended so that all packages pass into the colon from the stomach, and then low-volume phosphosoda enemas or high-volume saline enemas are administered. Food ingestion must not be permitted until all packages have moved into the colon. Metoclopramide 10 mg, 8th hourly, may be administered to encourage gastric emptying. Bowel obstruction must be ruled out. It may be advisable to empty the rectum first by a bisacodyl suppository.
·
Symptomatic patients must be managed with
contin-uous-infusion naloxone, activated charcoal, and whole bowel irrigation.
Intestinal perforation or obstruction by packets requires surgical
intervention.
Related Topics