Chapter: Pharmaceutical Drug Analysis: Radioimmunoassay
Novel Applications of Radioimmunoassay(RIA)-Techniques
NOVEL APPLICATIONS OF RIA-TECHNIQUES
The radioimmunoassay technique has been gainfully
exploited in a variety of novel applications of which only the two important aspects stated below will
be discussed briefly, namely :
(i) Combined
RIA Technique-Isotope Dilution, and
(ii)
Stereospecificity.
1. COMBINED RIA-TECHNIQUES ISOTOPE DILUTION
In normal RIA-procedures the labelled drug or metabolite
not only serves as the tracer for recovery but also for RIA quantification.
However, the isotope dilution method categorically makes a clear separation of
the drug and its metabolites. Consequently, a non-specific antiserum is
employed to actually quantify the total amount of both unlabelled and labelled
substance present.
The combined RIA-technique and isotope dilution has been
successfully developed to estimate SULINDAC*
along with its two prominent metabolites, namely : its sulphone and its
sulphide present in the plasma-level
as shown in the following chemical structures X and Y.
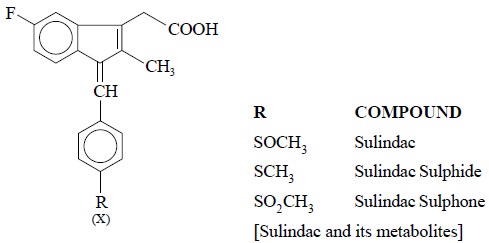
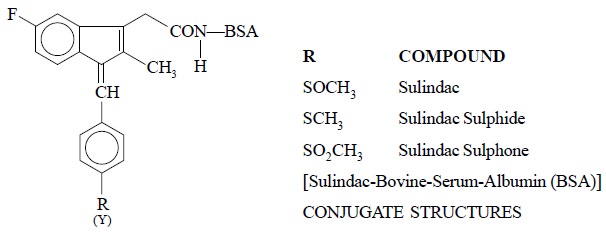
After due corrections have been incorporated with regard
to recovery, it is possible to quantify the amount of standard sulindac or
unknown ; besides standard metabolite or unknown.
Advantages : The various advantages are as
follows :
(i) The
methodology is not only very specific but also fairly sensitive, and
(ii) It serves
as a substitute to simple RIA-procedure when specific antisera are not readily
accessible.
Disadvantages : It has two main disadvantages :
(i) The method
is time-consuming, and
(ii) It
involves tiresome and meticulous process of isotope dilution.
2. STEREOSPECIFICITY
The stereospecificity
of antigen-antibody reactions has gained its due recognition more than
half-a-century ago*. However, an intensive and extensive stereospecificity
radioimmunoassay procedures have been adequately applied to a number of
pharmaceutical substances since mid-seventies, for instance : atropine, propranolol, methadone-to name a few.
Propranolol** which represents a
comparatively better conceived example shall be discussed briefly as under with regard to its stereospecificity
:
Propranolol is a recemic mixture i.e., it contains an equimolecular portion of d- and l- isomers as
given below :
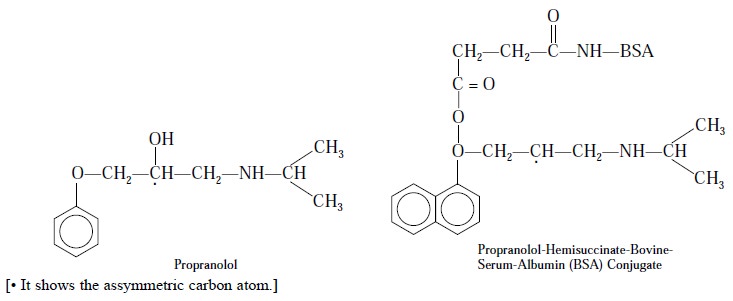
Interestingly, only the l-propranolol exhibit β-blocking activity. In actual
practice, two antisersa have been developed experimentally, namely :
(a) Antisera
against the dl-racemic mixture, and
(b) Antisera
against the l-isomer (active form
only).
The dl-propranolol
antiserum exhibits an almost equal affinity for both d- and l-isomers ; whilst
the l-propranolol shows exclusively a
marked and pronounced affinity for the l-isomer,
By the application of these two RIA-techniques, it was practically feasible to
quantify the plasma and heart concentrations of dl- and l-propranolol
individually. Thus, the concentrations of d-propranolol
could be arrived at by subtracting the con-centration of l-isomer from the dl-mixture.
It has been clearly demonstrated by Kawashima and coworkwers* that the d-propranolol undergoes distribution in vivo very sluggishly besides being
metabolized more rapidly whereas the l-isomer
gets distributed rather quickly to various tissues including the heart.
The RIA-procedure for propranolol is solely based on
antisera derived from conjugates through the asymmetric carbon (i.e., the optical carbon) as shown in
the above chemical structures. Perhaps it could be possible that the
stereospecificity of propranolol is caused due to the conformation of the
drug-hapten in rela-tion to the carrier protein to a great extent, through this
hypothesis remains to be ascertained scientifically.
Consequently, the stabilization of the optical carbon by
virtue of the conjugation to respective protein might improve upon the status
of the specificity to a considerable extent. In order to prove the validity of
this phenomenon one may carry out a definitive methodology whereby a closely
monitored and controlled study of the antisera obtained by conjugates
specifically prepared at the ‘asymmetric-carbon’ and at ‘another-site’ are both
compared simultaneously under identical experimental parameters.
Related Topics