Chapter: Microbiology and Immunology: Antimicrobial Agents Therapy and Resistance
Mechanisms of Action of Antimicrobial Drugs
Mechanisms of Action of Antimicrobial
Drugs
Antimicrobial drugs may be bactericidal or bacteriostatic. A
bactericidal drug kills bacteria, whereas a bacteriostatic drug inhibits the
growth of bacteria, but does not kill them.
·
Bactericidal drugs are very much useful in (a) life- threatening situations, (b) endocarditis, (c)
patients with low polymor-phonuclear count (below 500/ L), and (d) conditions in which bacteriostatic
drugs do not cause a cure.
·
The bacteriostatic drugs depend on the host defense mecha-nisms,
such as phagocytes to kill the bacteria. Hence, these drugs are not used when
the patient has too few neutrophils.
Antibiotics act against bacteria by the following mechanisms:
a)
Inhibition of cell wall synthesis
b)
Inhibition of protein synthesis
c)
Inhibition of nucleic acid synthesis
d)
Alteration of cell membrane function
Inhibition of Cell Wall Synthesis
Penicillins, cephalosporins, and vancomycin are the antibiot-ics
that act against bacteria by interfering with their cell wall synthesis.
Penicillins and cephalosporins are called -lactam antibiotics because they
possess an intact -lactam ring essen-tial for antimicrobial activity.
◗ Penicillins
The penicillins are called -lactam antibiotics
because they have a common chemical nucleus (6-aminopenicillanic acid) that
contains a -lactam ring. They are bactericidal antibiotics that act by
inhibiting bacterial cell wall synthesis. They act primarily against
Gram-positive organisms. Other penicillins, such as ampicillin, amoxicillin,
carbenicillin, ticarcillin, piperacillin, etc., act against both Gram-positive
and Gram-negative organisms. Penicillins kill bacteria by the following
mechanisms:
·
Penicillins kill bacteria during their growing stage and are more
active against replicating bacteria during the log phase than the lag phase of
the bacterial growth curve.
·
The intact peptidoglycan in the cell wall of the bacteria has
chains of N-acetyl muramic acid (NAM)
and N-acetyl glucosamine (NAG)
glycans cross-linked by peptide bridges. Penicillins and cephalosporins act by
inhibiting penicillin-binding proteins (PBPs), also known as transpeptidases,
that link the cross-bridges between NAMs, thereby, greatly weakening the cell
wall meshwork. Streptococcus pneumoniae
is an example of bacteria that show resistance against penicillins due to
mutations that occur in the genes encoding PBPs.
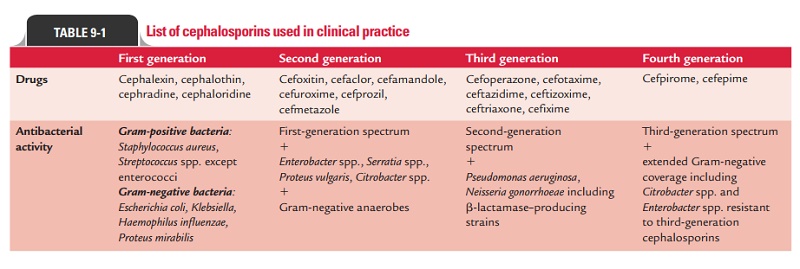
◗ Cephalosporins
Cephalosporins like penicillins are -lactam antibiotics. However,
they differ from penicillins in having 7-amino-cephalosporanic acid instead of
6-aminopenicillanic acid in their structure. They exhibit bactericidal activity
similar to that of penicillin. They are effective against a wide variety of
bacterial pathogens.
·
First-generation cephalosporins are active mainly against
Gram-positive cocci.
·
Second-, third-, and fourth-generation cephalosporins are active
primarily against Gram-negative bacilli.
The important cephalosporins are listed in Table 9-1.
Carbapenems (such as imipenem) and monobactams (such as aztreonam)
are other examples of –lactam antibiotics, but these are structurally different
from penicillins and cephalosporins.
◗ Vancomycin
Vancomycin is a glycopeptide, but its mode of action is very
similar to that of -lactam antibiotics, such as penicillins and cephalosporins.
It kills bacteria by inhibiting their cell wall syn-thesis. Vancomycin is a
bactericidal antibiotic and is used:
·
Most widely against Clostridium
spp. and Staphylococcus spp. infections;
·
Orally for treatment of antibiotics-associated colitis; and
·
Also for treatment of methicillin-resistant Staphylococcusaureus (MRSA) infections.
◗ Teicoplanin
Teicoplanin is a glycopeptide antibiotic extracted from Actinoplanes teichomyceticus, with a
similar spectrum of activityto vancomycin. Its mechanism of action is to
inhibit bacterial cell wall synthesis. Teicoplanin is used in:
·
Prophylaxis and treatment of serious infections caused by
Gram-positive bacteria, including MRSA and Enterococcusfaecalis.
·
Treatment of pseudomembranous colitis and Clostridium dif-ficile-associated diarrhea, with comparable
efficacy with van-comycin. Its strength is considered to be due to the length
of the hydrocarbon chain.
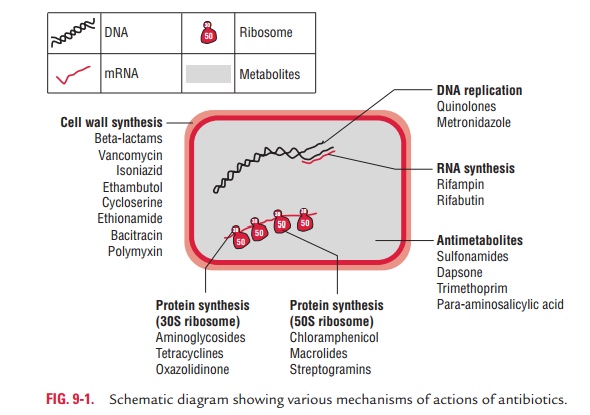
Inhibition of Protein Synthesis
Bacteria have 30S and 50S ribosomal units, whereas mam-malian cells
have 80S ribosomes. The subunits of each type of ribosome, their chemical
composition, and their functional specificities are sufficiently different,
which explains why these antimicrobial drugs can inhibit protein synthesis in
bacte-rial ribosomes without having a major effect on mammalian ribosomes.
Aminoglycosides and tetracyclines act at the level of 30S ribosomal
subunits, whereas erythromycins, chloramphenicol, and clindamycins act at the
level of 50S ribosomal subunits (Fig. 9-1).
◗ Aminoglycosides
Aminoglycosides are a family of drugs that include streptomy-cin,
gentamicin, tobramycin, amikacin, and neomycin, which are increasingly used in
different clinical conditions. They kill bacteria by inhibiting protein
synthesis. They do so by binding to the 30S subunit ribosome, which blocks the
initiation com-plex, leading to no formation of peptide bonds or polysomes.
They act mostly against Gram-negative bacteria.
◗ Tetracyclines
Tetracyclines are a family of drugs that include dimethylchlo-rotetracyclines,
doxycycline, and rolitetracycline. They are bacteriostatic drugs. They act by
inhibiting protein synthesis of the bacteria by blocking the binding of
aminoacyl t-RNA to the 30S ribosomal subunits. Therefore, they prevent
introduction of new amino acids to the nascent peptide chain. The action is
usually inhibitory and reversible upon withdrawal of the drug. They are
variably effective in infections caused by both cocci and bacilli. They are
also highly effective against rickettsial and chlamydial infections.
Doxycycline, dimethylchlorotetra-cycline, and rolitetracycline are the examples
of semisynthetic tetracyclines.
◗ Macrolides
Macrolides (such as erythromycin, azithromycin, clarithromy-cin,
and roxithromycin) have a macrolide ring to which sug-ars are attached. They
are bacteriostatic antibiotics. They act by inhibiting protein synthesis of the
bacteria by blocking the release of the t-RNA after it has transferred its
amino acids to the growing polypeptide. Erythromycin is effective against
Gram-positive cocci including MRSA, Neisseria
spp., and Haemophilusinfluenzae.
◗ Chloramphenicol
Chloramphenicol is mainly a bacteriostatic agent, and growth of
bacteria resumes when the drug is withdrawn. It acts by bind-ing to the 50S
subunit of the ribosome and blocking peptidyl transferase, the enzyme that
delivers the amino acid to the grow-ing polypeptide, resulting in the
inhibition of bacterial protein synthesis. Chloramphenicol is used less
frequently nowadays.
◗ Clindamycin
The mode of action of clindamycin is similar to that of
eryth-romycin. It inhibits bacterial protein synthesis by blocking the release
of t-RNA after it has transferred its amino acids to the growing polypeptide.
Inhibition of Nucleic Acid Synthesis
Sulfonamides, trimethoprim, quinolones, and
rifampin are examples of drugs that act by inhibition of nucleic acid
synthesis.
◗ Sulfonamides and trimethoprim
Sulfonamides and trimethoprim inhibit nucleic acid synthesis by
inhibiting nucleotide synthesis. Sulfonamides are structural analogs of
para-aminobenzoic acid (PABA). Due to structural similarity to PABA,
sulfonamide competes with the latter during bacterial metabolism. Sulfonamides
enter into the reaction in place of PABA and compete for the active center of
the enzyme. As a result, nonfunctional analogs of folic acid are formed,
preventing further growth of the bacterial cell. The inhibitory action of
sulfonamides on bacterial growth can be counteracted by an excess of PABA in
the environment (competitive inhibi-tion). Many other bacteria, however,
synthesize folic acid, as mentioned above, and consequently are susceptible to
action by sulfonamides. Sulfonamides are bacteriostatic drugs effective against
a variety of Gram-negative and Gram-positive bacteria.
Trimethoprim
(3,4,5-trimethoxybenzylpyrimidine) is a bac-teriostatic drug active against
both Gram-positive and Gram-negative organisms. The compound inhibits the
enzyme dihydrofolic acid reductase 50,000 times more efficiently in bacteria
than in mammalian cells. This enzyme reduces dihydrofolic to tetrahydrofolic
acid, leading to decreased syn-thesis of purines and ultimately of DNA.
Sulfonamides and trimethoprim inhibit the synthesis of
tet-rahydrofolic acid, the main donor of the methyl groups that are essential
to synthesize adenine, guanine, and cytosine.
◗ Quinolones
Quinolones are synthetic analogs of nalidixic
acid. They are a fam-ily of drugs that include ciprofloxacin, ofloxacin, and
levofloxacin. They are bactericidal and act by inhibiting bacterial DNA
synthesis by blocking DNA gyrase. DNA
gyrase is the enzyme that unwinds DNA strands, so they can be replicated.
Quinolones are effective against both Gram-positive and Gram-negative
organisms.
◗ Rifampin
Rifampin inhibits bacterial growth by binding
strongly to the DNA-dependent RNA polymerase of bacteria. Thus, it inhibits
bacterial RNA synthesis. The mechanism of rifampin action on viruses is
different. It blocks a late stage in the assembly of poxviruses.
Alteration of Cell Membrane Function
The cytoplasm of all living cells is surrounded by the cytoplas-mic
membrane, which serves as a selective permeability bar-rier. The cytoplasmic
membrane carries out active transport functions, and thus controls the internal
composition of the cell. If the functional integrity of the cytoplasmic
membrane is disrupted, macromolecules and ions escape from the cell, and cell
damage or death ensues. The cytoplasmic membrane of bacteria and fungi has a
structure different from that of animal cells and can be more readily disrupted
by certain agents. Consequently, selective chemotherapy is possible.
Antifungal drugs act by altering the cell membrane function of the fungi. They show selective toxicity because cell membrane of the fungi contains ergosterol, while human cell membrane has cholesterol. Bacteria with the exception of Mycoplasma do not have sterols in their cell membranes, hence are resistant to action of these drugs.
Polymyxins bind to the cell membrane and alter its structure,
making it more permeable. The resulting water uptake leads to cell death. They
are cationic, basic proteins that act like detergents (surfactants).
Side-effects include neurotoxicity and acute renal tubular necrosis. It is
commonly used in the topical first-aid preparation.
Cellular target sites of antimicrobial drugs are listed in Table
9-2.

Related Topics