Chapter: Clinical Anesthesiology: Clinical Pharmacology: Local Anesthetics
Local Anesthetic: Structure Activity Relationships
STRUCTURE ACTIVITY RELATIONSHIPS
Local anesthetics consist of a
lipophilic group (usu-ally an aromatic benzene ring) separated from a
hydrophilic group (usually a tertiary amine) by an intermediate chain that
includes an ester or amide linkage. Articaine, the most popular local
anes-thetic for dentistry in several European countries, is an amide but it
contains a thiophene ring rather than a benzene ring. Local anesthetics are
weak bases that usually carry a positive charge at the ter-tiary amine group at
physiological pH. The nature of the intermediate chain is the basis of the
classifi-cation of local anesthetics as either esters or amides (Table 16–2).
Physicochemical properties of local anesthetics depend on the substitutions in
the aro-matic ring, the type of linkage in the intermediate
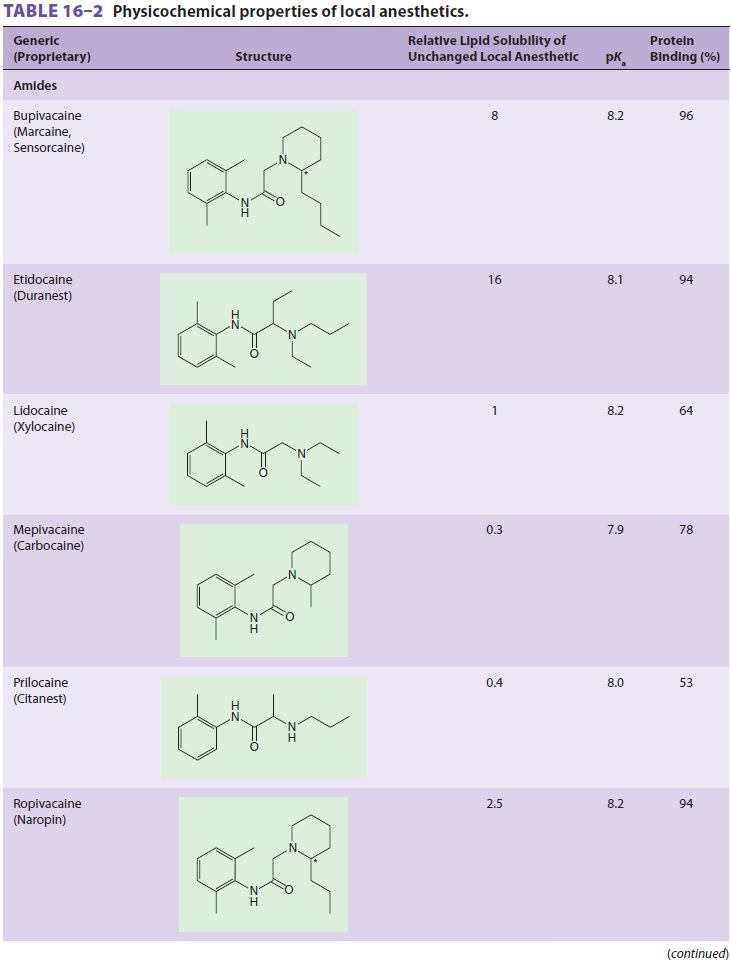
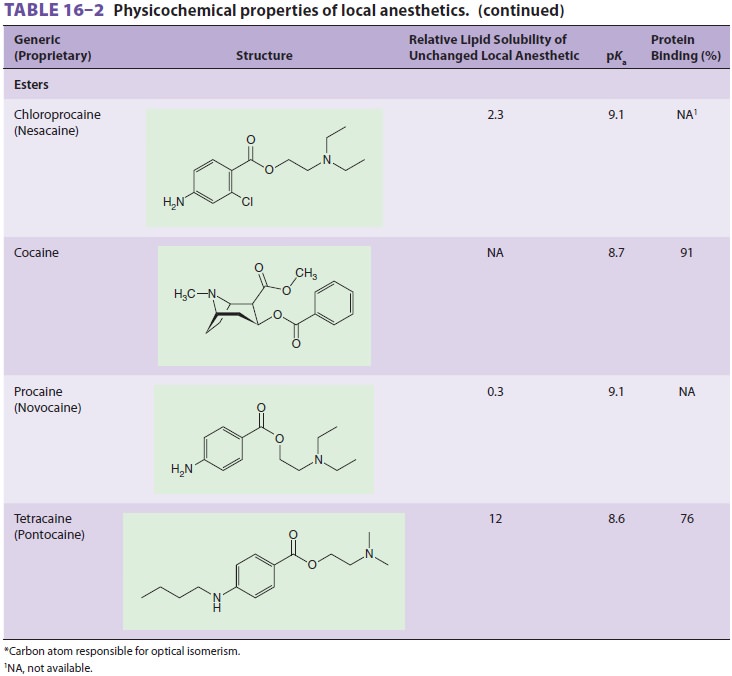
chain, and the alkyl groups attached to
the amine nitrogen. Potency correlates with octanol solubility, which in turn
reflects the ability of the local anesthetic molecule to permeate lipid
membranes. Potency is increased by adding large alkyl groups to a parent
molecule (compare tetracaine to procaine or bupivacaine to mepivacaine). There
is no measure-ment of local anesthetic potency that is analogous to the minimum
alveolar concentration (MAC) of inha-lation anesthetics. The minimum
concentration of local anesthetic that will block nerve impulse conduction is
affected by several factors, including fiber size, type, and myelination; pH
(acidic pH antagonizes block); frequency of nerve stimulation; and electrolyte
concentrations (hypokalemia and hypercalcemia antagonize blockade).Onset of
local anesthetic action depends on many factors, including lipid solubility and
the relative concentration of the nonionized lipid-soluble form (B) and the
ionized water-soluble form (BH+), expressed by the pKa. The pKa is the pH at which the fraction of
ionized and nonionized drug is equal. Less potent, less lipid-soluble agents
generallyhave a faster onset than more potent, more
lipid-soluble agents.
Local anesthetics with a pKa
closest to physi-ological pH will have (at physiological pH) a greater fraction
of nonionized base that more read-ily permeates the nerve cell membrane,
generally facilitating a more rapid onset of action. It is the lipid-soluble
form that more readily diffuses across the neural sheath (epineurium) and
passes through the nerve membrane. Curiously, once the local anes-thetic
molecule gains access to the cytoplasmic side of the Na channel, it is the
charged cation (rather than the nonionized base) that more avidly binds the Na
channel. For instance, the pKa of lidocaine exceeds physiological pH. Thus, at
physiological pH (7.40) more than half the lidocaine will exist as the charged
cation form (BH+).
It is often stated that the onset of
action of local anesthetics directly correlates with pKa. This asser-tion is not supported by
actual data; in fact, the agent of fastest onset (2-chloroprocaine) has the
greatest pKa of all clinically used agents. Other factors, such
as ease of diffusion through connective tissue, can affect the onset of action
in vivo. Moreover, not all local anesthetics exist in a charged form (eg,
benzocaine).
The importance of the ionized and
nonion-ized forms has many clinical implications, at least for those agents
that exist in both forms. Local anesthetic solutions are prepared commercially
as water-soluble hydrochloride salts (pH 6–7). Because epinephrine is unstable
in alkaline envi-ronments, commercially formulated, epinephrine-containing,
local anesthetic solutions are generally more acidic (pH 4–5) than the
comparable “plain” solutions lacking epinephrine. As a direct conse-quence,
these commercially formulated, epineph-rine-containing preparations may have a
lower concentration of free base and a slower onset than when the epinephrine
is added by the clinician at the time of use. Similarly, the extracellular
base-to-cation ratio is decreased and onset is delayed when local anesthetics
are injected into acidic (eg, infected) tissues. Tachyphylaxis—the decreased
efficacy of repeated doses—could be partly explained by the eventual
consumption of the local extracellular buffering capacity by repeat injections
of the acidic local anesthetic solution, but data are lacking. Some researchers
have found that alkalini-zation of local anesthetic solutions (particularly
commercially prepared, epinephrine-containing ones) by the addition of sodium
bicarbonate (eg, 1 mL 8.4% sodium bicarbonate per 10 mL local anesthetic)
speeds the onset and improves the quality of the block by increasing the amount
of free base available. Interestingly, alkalinization also decreases pain
during subcutaneous infiltration.Duration of action correlates with potency and
lipid solubility. Highly lipid-soluble localanesthetics have a longer duration
of action, pre-sumably because they more slowly diffuse from a lipid-rich
environment to the aqueous bloodstream. Lipid solubility of local anesthetics
is correlated with plasma protein binding. Local anesthetics are mostly bound
by α1-acid
glycoprotein and to a lesser extent to albumin. Sustained-release systems using
liposomal encapsulation or microspheres for deliv-ery of local anesthetics can
significantly prolong their duration of action, but these approaches are not
yet being used for prolonged anesthesia in the way that extended-duration
epidural morphine is being used for single-shot, prolonged epidural analgesia.
Differential block of sensory rather
than motor function would be desirable. Unfortunately, only bupivacaine and
ropivacaine display some selectively
(mostly during onset and offset of block) for sen-sory nerves; however, the
concentrations required for surgical anesthesia almost always result in some
motor blockade.
Related Topics