Chapter: Medical Physiology: Hemostasis and Blood Coagulation
Initiation of Coagulation: Formation of Prothrombin Activator
Initiation of Coagulation: Formation of Prothrombin Activator
Now that we have discussed the clotting process itself, we must turn to the more complex mechanisms that initiate clotting in the first place. These mechanisms are set into play by (1) trauma to the vascular wall and adjacent tissues, (2) trauma to the blood, or (3) contact of the blood with damaged endothelial cells or with collagen and other tissue elements outside the blood vessel. In each instance, this leads to the formation of prothrombin activator, which then causes prothrombinconversion to thrombin and all the subsequent clotting steps.
Prothrombin activator is generally considered to be formed in two ways, although, in reality, the two ways interact constantly with each other: (1) by the extrin-sic pathway that begins with trauma to the vascularwall and surrounding tissues and (2) by theintrinsicpathway that begins in the blood itself.
In both the extrinsic and the intrinsic pathways, a series of different plasma proteins called blood-clotting factors play major roles. Most of these are inac-tive forms of proteolytic enzymes. When converted tothe active forms, their enzymatic actions cause the suc-cessive, cascading reactions of the clotting process.
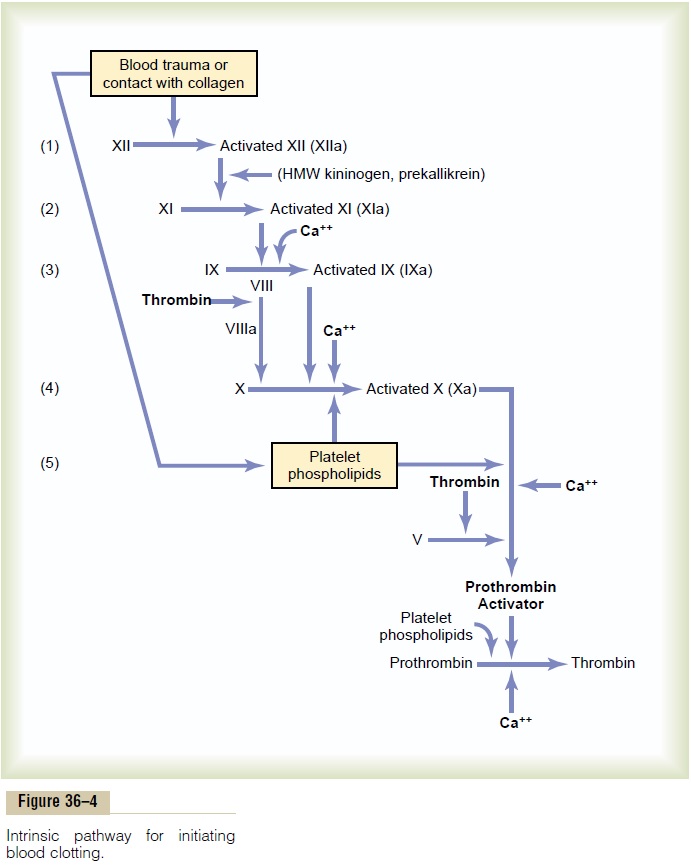
Most of the clotting factors, which are listed in Table 36–1, are designated by Roman numerals. To indicate the activated form of the factor, a small letter “a” is added after the Roman numeral, such as Factor VIIIa to indicate the activated state of Factor VIII.
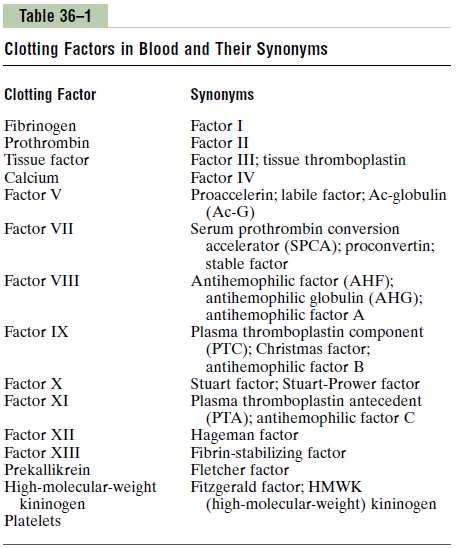
Extrinsic Pathway for Initiating Clotting
The extrinsic pathway for initiating the formation of prothrombin activator begins with a traumatized vas-cular wall or traumatized extravascular tissues that come in contact with the blood. This leads to the fol-lowing steps, as shown in Figure 36–3:
1. Release of tissue factor. Traumatized tissuereleases a complex of several factors called tissuefactor or tissue thromboplastin. This factor iscomposed especially of phospholipids from the membranes of the tissue plus a lipoproteincomplex that functions mainly as aproteolytic enzyme.
2. Activation of Factor X—role of Factor VII and tissue factor. The lipoprotein complex of tissuefactor further complexes with blood coagulation Factor VII and, in the presence of calcium ions, acts enzymatically on Factor X to form activatedFactor X (Xa).
3. Effect of activated Factor X (Xa) to form prothrombin activator—role of Factor V. Theactivated Factor X combines immediately with tissue phospholipids that are part of tissue factor or with additional phospholipids released from platelets as well as with Factor V to form the complex called prothrombin activator. Within a few seconds, in the presence of calcium ions (Ca++), this splits prothrombin to form thrombin, and the clotting process proceeds as already explained. At first, the Factor V in the prothrombin activator complex is inactive, but once clotting begins and thrombin begins to form, the proteolytic action of thrombin activates Factor V. This then becomes an additional strong accelerator of prothrombin activation. Thus, in the final prothrombin activator complex, activated Factor X is the actual protease that causes splitting of prothrombin to form thrombin; activated Factor V greatly accelerates this protease activity, and platelet phospholipids act as a vehicle that further accelerates the process. Note especially thepositive feedback effect of thrombin, acting through Factor V, to accelerate the entire process once it begins.
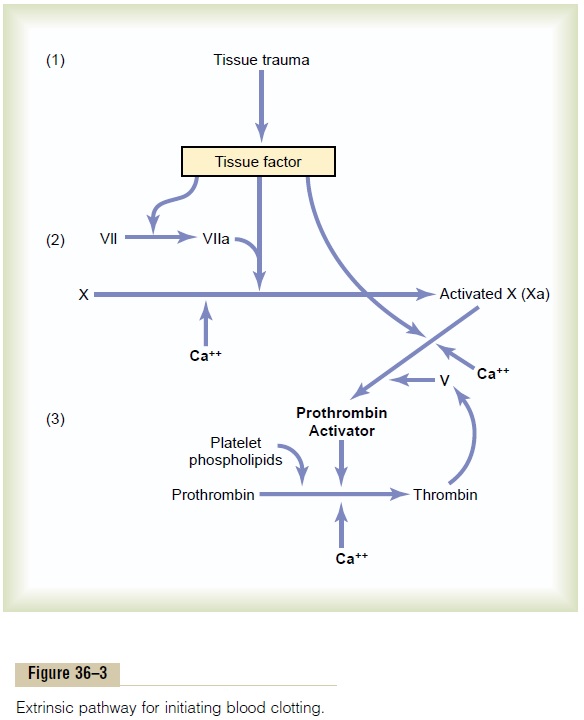
Intrinsic Pathway for Initiating Clotting
The second mechanism for initiating formation of pro-thrombin activator, and therefore for initiating clot-ting, begins with trauma to the blood itself or exposureof the blood to collagen from a traumatized bloodvessel wall. Then the process continues through the series of cascading reactions shown in Figure 36–4.
1. Blood trauma causes (1) activation of Factor XII and (2) release of platelet phospholipids. Traumato the blood or exposure of the blood to vascular
wall collagen alters two important clotting factors in the blood: Factor XII and the platelets. When Factor XII is disturbed, such as by coming into contact with collagen or with a wettable surface such as glass, it takes on a new molecular configuration that converts it into a proteolytic enzyme called “activated Factor XII.” Simultaneously, the blood trauma also damages the platelets because of adherence to either collagen or a wettable surface (or by damage in other ways), and this releases platelet phospholipids that contain the lipoprotein called platelet factor 3, which also plays a role insubsequent clotting reactions.
2. Activation of Factor XI. The activated Factor XIIacts enzymatically on Factor XI to activate this factor as well, which is the second step in the intrinsic pathway. This reaction also requires HMW (high-molecular-weight) kininogen and isaccelerated by prekallikrein.
3. Activation of Factor IX by activated Factor XI.The activated Factor XI then acts enzymatically on Factor IX to activate this factor also.
4. Activation of Factor X—role of Factor VIII.The activated Factor IX, acting in concert with activated Factor VIII and with the platelet phospholipids and factor 3 from the traumatized platelets, activates Factor X. It is clear that when either Factor VIII or platelets are in short supply, this step is deficient. Factor VIII is the factor that is missing in a person who has classic hemophilia, for which reason it is called antihemophilic factor. Platelets are the clotting factor that is lacking in the bleeding disease calledthrombocytopenia.
5. Action of activated Factor X to form prothrombin activator—role of Factor V. This step in theintrinsic pathway is the same as the last step in the extrinsic pathway. That is, activated Factor X combines with Factor V and platelet or tissue phospholipids to form the complex called prothrombin activator. The prothrombin activatorin turn initiates within seconds the cleavage of prothrombin to form thrombin, thereby setting into motion the final clotting process, as described earlier.
Role of Calcium Ions in the Intrinsic and Extrinsic Pathways
Except for the first two steps in the intrinsic pathway, calcium ions are required for promotion or accelera-tion of all the blood-clotting reactions. Therefore, in the absence of calcium ions, blood clotting by either pathway does not occur.
In the living body, the calcium ion concentration seldom falls low enough to significantly affect the kinetics of blood clotting. But, when blood is removed from a person, it can be prevented from clotting by reducing the calcium ion concentration below the threshold level for clotting, either by deionizing the calcium by causing it to react with substances such as citrate ion or by precipitating the calcium with sub-stances such as oxalate ion.
Interaction Between the Extrinsic and Intrinsic Pathways—Summary of Blood-Clotting Initiation
It is clear from the schemas of the intrinsic and extrin-sic systems that after blood vessels rupture, clotting occurs by both pathways simultaneously. Tissue factor initiates the extrinsic pathway, whereas contact of Factor XII and platelets with collagen in the vascular wall initiates the intrinsic pathway.
An especially important difference between the extrinsic and intrinsic pathways is that the extrinsicpathway can be explosive; once initiated, its speed ofcompletion to the final clot is limited only by the amount of tissue factor released from the traumatized tissues and by the quantities of Factors X, VII, and V in the blood. With severe tissue trauma, clotting can occur in as little as 15 seconds. The intrinsic pathway is much slower to proceed, usually requiring 1 to 6 minutes to cause clotting.
Related Topics