Chapter: Aquaculture Principles and Practices: Reproduction and Genetic Selection
Induced reproduction: Hypophysation, Gametes and fertilization - Aquaculture
Induced reproduction
As explained in the previous section, the hypothalamus regulates the reproductive functions of the pituitary gland. The correct combination of environmental factors required for matura-tion, ovulation and spawning, brings about an accelerated release of gonadotropin from the pituitary into the bloodstream. Ng and Idler (1978 a, b) and Idler and Ng (1979) have isolated two gonadotropic hormones: one with a low carbohydrate content that induces vitellogenesis and the other, rich in carbohydrates, inducing maturation and ovulation. The surge of gonadotropins that occurs brings about maturational changes culminating in the act of spawning. Environmental conditions required for the initiation of oocyte maturation, ovulation and spawning are much more complex than those for gametogenesis.
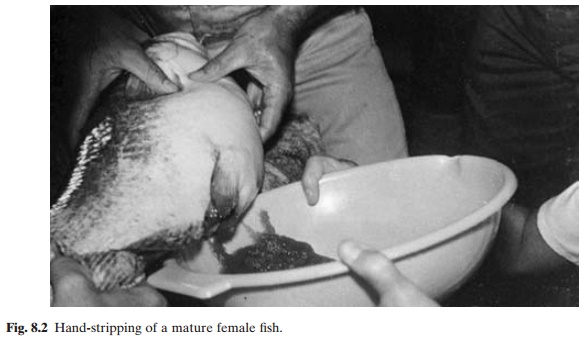
Very often under culture conditions, the required environmental conditions may not be available, or may not persist for a sufficient length of time for spontaneous maturation to occur. This has led to the development of induced reproduction or hypophysation tech-niques (Houssay, 1931;Von Ihering, 1935, 1937). By the injection of pituitary homogenates(fig. 8.1), the natural gonadotropin surge is simulated, by-passing to some extent the environmental variables of temperature, rain-fall, photoperiod, etc. Besides the advantage of regulating the time of spawning, this enables the adoption of other methods of artificial propagation, including hand-stripping (fig. 8.2), fertilization, incubation, hatching and larval
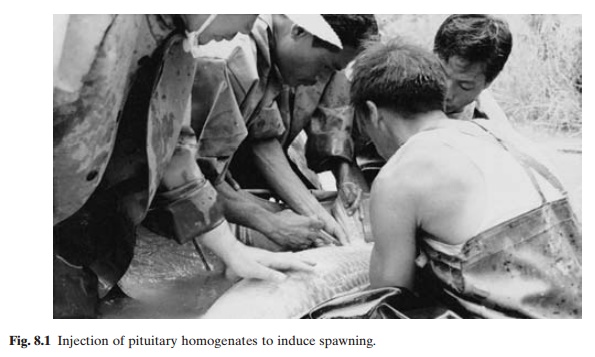
rearing. While hypophysation has been demonstrated to be effective in a large variety of fish species, its major contribution in respect of aquaculture technologies, since its first field application in Brazil in 1935, has been in the inducement of spawning in fishes that do not ordinarily breed under conditions of confinement or do so only under specific environmental conditions. It has now become a common practice in many countries and is utilized widely in the reproduction of finfish, despite the fact that the relevant mechanisms are not fully understood and little standardization of the techniques has been achieved.
Vitellogenesis in decapod crustacea, particularly Penaeid shrimps and lobsters, has been shown to be mediated by hormones. Male shrimps mature fully under captive conditions and spermatophores can be seen through the carapace. Female shrimps often do not mature fully, even though maturing eggs can be found in the ovaries. The maturation process seems to be inhibited by a gonad-inhibiting hormone (GIH) secreted by the medulla terminalis ganglionic x-organ (MTGX) and stored in the sinus gland. The y-organ, which secretes the moult hormone crustecdysone, also has an influence on maturation. The ablation (surgical removal) of eye stalks, which have the glands containing the inhibitory hormone, has been shown to accelerate vitellogenesis in many crustaceans. Besides environmental factors like temperature, photoperiod, salinity and pH, the state of nutrition of brood animals seems to be an important factor in the maturation and spawn-ing of shrimps.
Hypophysation
A more detailed description of the techniques of induced spawning, including environmental control employed for the breeding of important aquaculture species, can be found in Part II. Only some of the common features of induced spawning, with special reference to finfish, will be discussed here.
The mammalian gonadotropic hormones, LH and human chorionic gonadotropin (HCG), are effective in inducing maturation and ovulation in fishes. Although a number of species have been induced to breed by the administration of HCG or a combination of HCG and mammalian pituitary extract, there are certain refractory breeders, like the Indian and Chinese carps, where fish pituitary homogenates or extracts are needed to induce spawning. There are reports of successful breeding of even these species, by using HCG under certain circumstances. The Chinese carps, which have been bred two or three times by administration of fish pituitary extract, will respond positively to injections of HCG. Bhowmick (1979) has reported on the use of crude HCG for induced spawning of one species of Indian carp, Labeorohita. It has, however, been reported thatrepeated injections of HCG could induce a ‘drug resistance effect’ related to the production of antibodies against foreign proteins. Nevertheless, it would appear that homogenates and extracts of whole pituitary glands and partially purified fish gonadotropins are more potent in inducing maturation and ovulation in fishes than mammalian gonadotropins, and can be used extensively in commercial fish culture.
While the administration of the appropriate hormone is basic to the success of induced breeding, the condition of the brood fish and the environmental conditions are also equally important. The large number of failures in induced breeding can often be traced to poor condition of the brood fish, including their health and nutrition and stage of gonadal development, as well as to environmental conditions in spawning tanks or enclosures. Chinese farmers believe that it is more difficult to breed wild Chinese carps, as well as carp that have attained maturity for the first time. They prefer to rear spent fish in special holding ponds, fed on a special protein diet, for future breeding.
The identification of sex is another important requirement for successful induced breeding. Many species do not have distinctive and permanent sex characteristics. When there are no secondary sex characteristics, detailed morphometric characteristics will have to be used to separate sexes, particularly in the prepuberty stages. After sexual differentiation, it may be possible to distinguish the sexes through examination of the gonads. This will involve the use of endoscopy or biopsy, which is difficult to use on a large scale. Siphoning of eggs and their examination under a microscope, to determine the stage of maturity of females, have beendescribed by Chen et al. (1969) but the adoption of this method in large-scale breeding work is not always very practical. Other possible methods, such as the use of serum markers or detection of circulating vitellogenin, are also difficult to use in the field. Aquaculturists have therefore to depend largely on practical experience and field observations to distinguish the sexes and determine the stage of maturity of breeders. Brood female fish ready for spawning are identified by the well-rounded and soft abdomen and swollen genital opening. The male releases a few drops of thick milt when its abdomen is slightly pressed.
As indicated earlier, several species of fish respond to injections of HCG and other mam-malian hormones, and these hormones are commercially available to aquaculturists. Many species, which are more difficult to spawn under confinement, need injections of fish pituitary for maturation and spawning. There are differences of opinion regarding the species-specificity of the pituitary, but aquaculturists generally prefer to use the glands of the same or closely related species. It is recommended that pituitaries from phylogenetically close donors should be used, when there is a choice. However, common carp is considered a universal donor and its pituitary is being used very widely for both experimental and commercial breeding purposes for several species. Salmon pituitary is also used for breeding a number of species. Though commercially available on a limited scale, a large majority of aquaculturists have to depend on local arrangements for the collection and preservation of the glands. Glands of the recipient species or of other proven donor species are used. Fractionation and purification of teleost gonadotropins are still in experimental stages. Though potent gonadotropic preparations have been made from fish pituitaries by means of chemical/ ethanol fractionation, they have not been used widely in spawning refractory fish.
Glands extracted from catches of the selected mature donor species are preserved in alcohol or acetone or frozen for storage.
Freshly collected glands are first desiccated in absolute ethyl alcohol (changing the preservative several times) and then stored in fresh alcohol at room temperature or under refrigeration. The glands remain active for a period
of about two years. Instead of alcohol, the glands can be desiccated in acetone, changing it several times as for alcohol. The desiccated glands are dried in vacuum and stored in that condition or sealed in vials and stored in frozen condition. Acetonedried glands retain their activity for 6–10 years.The glands can also be pre-served by quick freezing, but the most common method of preservation is acetone drying.
Though a number of methods of preparing pituitary homogenates and extracts have been tried, the most commonly accepted method is extraction with distilled water or saline sollution. The glands are macerated in a small volume of water or saline solution and brought up to the desired volume. Distilled water, common salt solution (0.3–1 per cent) and physiological saline can be used, as they all seem to give equivalent results. The homogenate can be used as such for injection, or filtered or centrifuged to obtain filtrate or supernate which can be injected. Extraction with trichloracetic acid (TCA) at low concentrations of 1.25–2.5 per cent for short time-periods of three to six hours, is reported to provide more complete extraction and better results. But this practice has not received wide acceptance, probably because of the specific requirements of concentration and extraction time. It is reported that higher concentrations and/or longer extraction, can result in denaturation of glycoproteins.
As pituitary extracts are subject to rapid enzymic deterioration, they have to be prepared fresh every time fish are to be bred. This is obviously inconvenient. Methods of preserving extracts have been tried with some success. One method involves the extraction of pituitary glands in a small volume of distilled water, and refrigerating it for 24–48 hours, after which glycerine is added to make a 2:1 ratio with water. The suspension is again refrigerated for 24–48 hours, centrifuged and the supernate stored under refrigeration in airtight vials. Another method consists of grinding acetone-dried pituitaries, sieving them through 40–60 mesh/mm2 sieves and storing in sealed vials at 5°C. Both these techniques are aimed at achieving homogeneous preparations of uniform potency.
Despite its wide use, the dosage frequency and latency period of pituitary administrationremains more or less at a trial-and-error stage, and sometimes leads to poor results. This is mainly on account of the variations in the gonadotropic content of the pituitary material used and the stage of sexual maturity of the brood fish, besides the environmental conditions and the stress to which the breeders are subjected. The mode of injection (intraperitoneal or intramuscular) also appear to affect results. Development of an acceptable method of assessing gonadotropic content should greatly assist in determining practical dosages.
Though several biological units have been proposed, none seems to have gained wide acceptance.
Gametes and fertilization
Injection of pituitary homogenate or extract is usually given into the dorsal muscles above the lateral line and below the anterior part of the dorsal fin, or the dorsal part of the caudal peduncle. Injections into the body cavity are considered less efficient. The required quantity of the gland is generally administered in two to four doses (one or more preparatory injections followed by one or more final doses). As indicated earlier, suitable environmental conditions, besides pituitary injection, will be needed for ovulation to take place. Temperature, high dissolved oxygen levels and lack of stress are some of the important requirements. The process of ovulation takes some time, depending on the species and environmental conditions. Maturation of the ovum starts when its nucleus starts to migrate from the centre toward the micropyle and undergoes hydration by absorbing fluids. Ovulation starts with the disappearance of the nuclear membrane and ends with the first meiotic division. At the same time, the follicle which attaches the eggs to the wall of the ovary splits and releases the eggs into the cavity of the ovary, from where it can freely flow through the genital opening.
Many of the fish that are treated with gonadotropic hormones start to spawn in the presence of active males after normal ovulation. The eggs are fertilized by the male breeders and the fertilized eggs can be collected easily for hatching. Where such induced spawning does not occur, it becomes necessary to strip the sex products from the females and malesand artificially fertilize them. Ripe ova remaining unspawned for long periods after ovulation become over-ripe and do not develop normally. It is also not uncommon for the phenomenon of ‘plugging’ to occur in gravid females subjected to overdoses of hormone. In such cases, natural spawning cannot be accomplished, since a mass of ovarian eggs forms a plug at the urinogenital opening, preventing the free flow of eggs. Stripping will be necessary to obtain eggs from such fish. Stripping and artificial fertilization are necessary also for fish with sticky eggs like the common carp. Such eggs will have to be treated to dissolve the sticky layer, so that they can be incubated in suitable incubators. A quick washing with a weak tannin solution after the eggs have swollen will be effective in removing the stickiness of the eggs. Common salt and carbamide (urea) solution can also be used for removing the sticky layer.
The ovulated egg which has undergone the first meiotic division will have the second meiotic division when the sperm penetrates it, ending in the extrusion of the second polar body. Further embryonic development leading to the formation of the first somatic cell completes the process of fertilization. The time available for the ripe egg to become fertilized is rather limited in most fresh-water fish, as the eggs swell rapidly in water and this results in the closure of the micropyle. The time available for common and Chinese carps is about 45–60 seconds. In saline solution the eggs seem to remain fertilizable for longer periods, up to several minutes.
The sperm, which is immotile in the testis, becomes motile on contact with the medium in which fertilization takes place. The duration of the activity of spermatozoa varies with the species, but is generally not longer than a couple of minutes. In the males of most species, dense semen with highly motile spermatozoa can be obtained without hormone injection. Administration of pituitary extracts brings about thinning of the seminal plasma and would facilitate spermiation. Relatively large numbers of spermatozoa are needed to fertilize an egg. For example, the requirement of a trout egg is reported to be 10000–300000 spermato-zoa and of a carp egg 13000–30000. This is due to the fact that the spermatozoon can penetrate at only one place, i.e. the micropyle, and the distance that can be covered by a trout spermato-zoon during its life span (2mm) is often less than the circumference of the ovum which is about 15–20mm. the probability of its reaching the micropyle is therefore low, if the motility is less. The number of spermatozoa compensates for the low motility. It is necessary to take special care in regulating the quantity of water added to the sexual products during fertilization. If too much water is added, many of the sperms will not be able to reach the micropyle. On the other hand, if sufficient water is not added, the micropyle of an egg may get blocked by other eggs, due to crowding, preventing the sperm from entering it.
Related Topics