Chapter: Obstetrics and Gynecology: Reproductive Cycles
Hypothalamic-Pituitary-Ovarian Axis
Reproductive Cycles
In the female reproductive cycle,
ovulation is followed by menstrual bleeding in a
cyclic, predictable sequence. This recurring process is established dur-ing
puberty (average age of menarche is 12.43 years) and continues until the
years prior to menopause (average age 51.4 years). Regular ovulatory cycles are
usually established by the third year after menarche, and continue until the
perimenopause. Therefore, between 15 and 45 years of age, a woman has
approximately 30 years of ovulatory reproduc-tive cycles. The reproductive
cycles may be interrupted by conditions including pregnancy, lactation,
illness, gyneco-logic disorders and endocrine disorders, and exogenous factors
such as hormone-based contraceptives and various other medications.
The
duration of an adult reproductive cycle, from the begin-ning of one menses to
the beginning of the next menses, averages approximately 28 days, with a range
of 23 to 35 days, and com-prises three distinct phases. Thefollicular phasebegins withthe onset of
menses (the first day of the menstrual cycle) and ends on the day of the luteinizing hormone (LH) surge. Ovulation occurs within 30–36 hours of
the LH surge.The luteal phase begins
on the day of the LH surge and ends with the onset of menses. The follicular
and luteal phases each last approximately 14 days in reproductive-age women;
however, variability in cycle length is more fre-quent at the extremes of the
reproductive age. The duration of the luteal phase remains relatively constant,
while the duration of the follicular phase can vary.
HYPOTHALAMIC-PITUITARY-OVARIAN AXIS
Hypothalamic-pituitary-ovarian
axis refers to
the complexinteractions between the hypothalamus, pituitary, and ovaries that
regulate the reproductive cycle. These interactions arebased on
the interplay of the hormones released by these structures:
gonadotropin-releasing hormone (GnRH), the gonadotropins follicle-stimulating hormone (FSH) and LH, and the ovarian sex
steroid hormones, estrogen and progesterone. Through stimulatory and inhibitory
actions, these hormones directly and indirectly stimulate oocyte development
and ovulation, endometrial development to facilitate embryo implantation, and
menstruation. Feedback loops between the hypothalamus, pituitary, and ovaries
are presented in Figure 33.1.
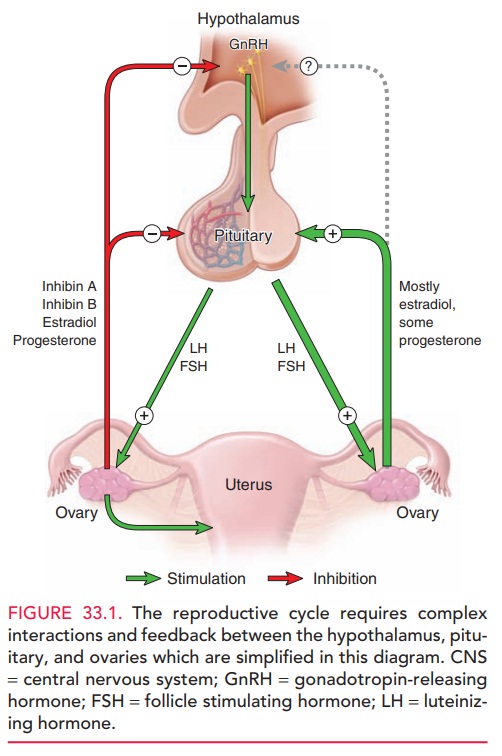
Disruption
of any of these communication and feedback loops results in alterations of
hormone levels, which can lead to dis-orders of the reproductive cycle; ultimately,
ovulation, repro-duction, and menstruation can be affected.
Hypothalamic GnRH Secretion
The gonadotropin-releasing hormone is secreted in a pulsatile fashion
from the arcuate nucleus of the hypothal-amus. GnRH reaches the anterior
pituitary through the hypothalamic–pituitary portal vascular system. The
pul-satile secretion of GnRH stimulates and modulates pitu-itary gonadotropin
secretion. Due to its remote location and a half-life of 2 to 4 minutes, GnRH
cannot be directly measured, thus measurements of LH pulses are used to
indicate GnRH pulsatile secretion. Ovarian function requires the pulsatile
secretion of GnRH in a specific pattern that ranges from 60-minute to 4-hour
intervals. Therefore, the hypothalamus
serves as the pulse generator of the reproductive cycle. Coordinated GnRH
release is stimulatedby various neurotransmitters and catecholamines as well as
by the inherent pulsatility of the GnRH neurons.
Pituitary Gonadotropin Secretion
The pituitary gonadotropins FSH and LH are glycoprotein hormones secreted by the anterior pituitary gland. FSH and LH are also secreted in pulsatile fashion in response to the pulsatile release of GnRH; the magnitude of secretion and the rates of secretion of FSH and/or LH are determined largely by the levels of ovarian steroid hormones, estrogen and progesterone, and other ovarian factors (such as inhibin, activin, and follistatin).
When a woman is in a state of
relative estrogen defi-ciency, as in the early follicular phase, the principal
gona-dotropin secreted is FSH. The ovary responds to FSH secretion with
estradiol production, with subsequent neg-ative feedback on the pituitary
inhibiting FSH secretion and positive feedback facilitating LH secretion.
Ovarian Steroid Hormone Secretion
At
midcycle, there is a marked increase in LH secretion (the LH surge), which
triggers ovulation. With ovulation, the
ovarianfollicle is converted into a corpus luteum and begins secret-ing
progesterone.
At birth, the human ovary
contains approximately one to two million primordial follicles. Each follicle
contains an oocyte that is arrested in prophase of the first meiotic division.
A large number of these inactive primordial fol-licles undergo a degenerative
process known as atresia during childhood; thus, at menarche, 300,000 to
500,000 oocytes remain.
The immature oocyte is encircled
by a single layer of granulosa cells, followed
by a thin basement membranethat separates the follicle from the surrounding
ovarian stroma. Early follicular maturation occurs independent of
gonadotropins; the granulosa cells proliferate into multiple layers, and the
surrounding stromal cells differentiate into theca cells. Granulosa cells produce estrogens, includingestrone
and estradiol, the latter being the
more potent of the two. Theca cells produce androgens which serve as the
precursors required for granulosa cell estrogen production. Androgens
(androstenedione and testosterone) enter the granulosa cells by diffusion and
are converted to estrogen. The two-cell theory of estrogen synthesis is
diagrammed in Figure 33.2.
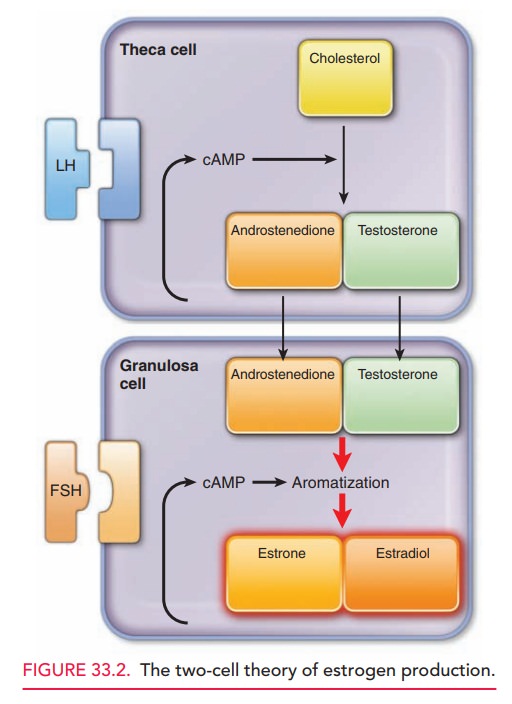
During follicular development,
FSH binds to FSH-receptors on the granulosa cells, causing cellular
prolifer-ation and increased binding of FSH and, hence, increased production of
estradiol. Estradiol stimulates the prolifera-tion of LH-receptors on theca and
granulosa cells, and LH stimulates the theca cells to produce androgens.
Greater androgen production leads to increased estradiol produc-tion. Rising
estrogen levels influence the pituitary gland through negative feedback and
results in suppression of
With
ovulation, the dominant ovarian follicle releases itsoocyte and transitions to
a progesterone-secreting ovarian cyst, the corpus
luteum.The process of follicular maturation ispresented in Figure 33.3.
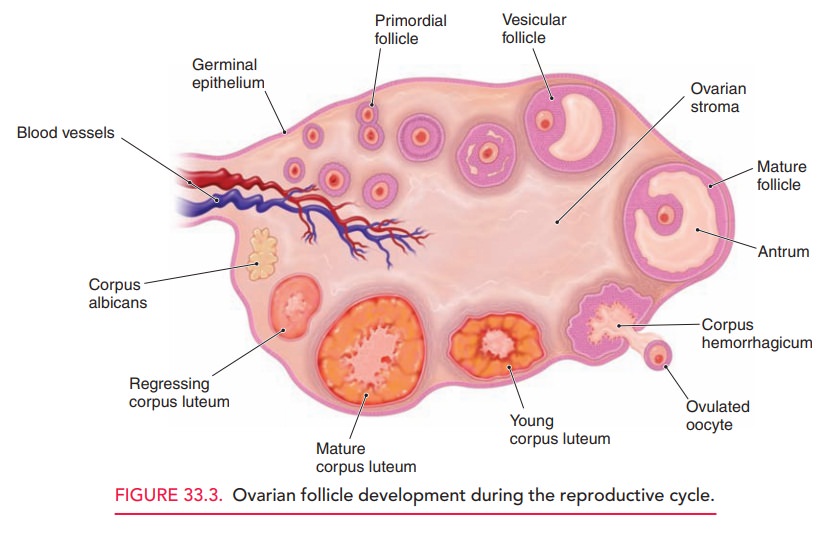
Related Topics