Chapter: Medical Microbiology: An Introduction to Infectious Diseases: Herpesviruses
Herpes Simplex Disease
HERPES SIMPLEX DISEASE
HSV is one of the best known of all viruses, given its frequency of infection and its propensity to cause recurrent ulcers in areas of the skin and mucous membranes. The two types differ in their predilection for causing lesions “above the waist” (HSV-1) or “below the waist” (HSV-2). As with all herpesviruses, herpes simplex persists in a latent form and reactivates to cause viral excretion and/or disease.
EPIDEMIOLOGY
Herpes simplex viruses are distributed worldwide. There are no known animal vectors, and humans appear to be the only natural reservoir. Direct contact with infected secretions isthe principal mode of spread. Seroepidemiologic studies indicate that the prevalence of antibody varies according to the age and socioeconomic status of the populationstudied. In most developing countries, 90% of the population have HSV-1 antibody by the age of 30. In the United States, HSV-1 antibody is currently found in approximately 60 to 70% of adult middle-class populations; among lower socioeconomic groups, however, the percentage is higher.
Detection of HSV-2 antibody before puberty is unusual. The virus is associated with sexual activity, and direct sexual transmission is the major mode of spread. Approxi-mately 15 to 30% of sexually active adults in Western industrialized countries have HSV-2 antibody. The virus can be isolated from the cervix and urethra of approximately 5 to 12% of adults attending sexually transmitted disease clinics; many of these patients are asymptomatic or have small, unnoticed lesions on penile or vulvar skin. Asymptomatic shedding accounts for transmission from a partner who has no active genital lesions and often no history of genital herpes. Genital herpes is not a reportable disease in the United States, but it is estimated that more than 1,000,000 new cases occur per year.
PATHOGENESIS
Acute Infections
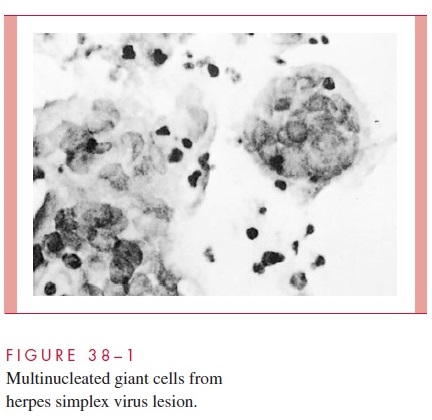
Pathologic changes during acute infections consist of development of multinucleated gi-ant cells (Fig 38–1), ballooning degeneration of epithelial cells, focal necrosis, eosinophilic intranuclear inclusion bodies, and an inflammatory response characterized by an initial polymorphonuclear neutrophil (PMN) infiltrate and a subsequent mononu-clear cell infiltrate. The virus can spread intra- or interneuronally or through supporting cellular networks of an axon or nerve, resulting in latent infection of sensory and auto-nomic nerve ganglia. Spread of virus can occur by cell-to-cell transfer and can therefore be unaffected by circulating immune globulin.
Latent Infection
In humans, latent infection by HSV-1 has been demonstrated by cocultivation techniques in trigeminal, superior cervical, and vagal nerve ganglia, and occasionally in the S2–S3 dorsal sensory nerve root ganglia. Latent HSV-2 infection has been demonstrated in the sacral (S2–S3) region. Latent infection of nervous tissue by HSV does not result in the death of the cell; however, the exact mechanism of viral genome interaction with the cell is incompletely understood. Several copies of the HSV viral genomes are in each latently infected neuronal cell. They exist in a circular form, and transcription of only a small por-tion of the viral genome occurs. Because latent infection does not appear to require syn-thesis of early or late viral polypeptides, antiviral drugs directed at the thymidine kinase enzymes or viral DNA polymerase do not eradicate the virus in its latent state.
Reactivation of virus from latently infected ganglionic cells with subsequent release of infectious virions appears to account for most recurrences of both genital and orolabial infections. The mechanisms by which latent infection is reactivated are unknown. Precipi-tating factors that are known to initiate reactivation of herpes simplex include; exposure to ultraviolet light, fever and trauma (eg, oral intubation).
IMMUNITY
Host factors have a major effect on clinical manifestations of HSV infection. Many episodes of HSV infection are either asymptomatic or mildly symptomatic. Initial symp-tomatic clinical episodes of the disease are more severe than recurrent episodes, probably because of the presence of anti-HSV antibodies and immune lymphocytes in persons with recurrent infections. Prior infection with HSV-1 may protect against or shorten the dura-tion of symptoms and lesions from subsequent infection with HSV-2 due to some degree of cross protection.
Both cellular and humoral immune responses are important in immunity to HSV. Neutralizing antibodies directed against HSV envelope glycoproteins appear to be im-portant in preventing exogenous reinfection. Antibody-dependent cellular cytotoxicity (ADCC) may be important in limiting early spread of HSV. By the second week after in-fection, cytotoxic T lymphocytes can be detected that are able to destroy HSV-infected cells prior to completion of the replication cycle. Conversely, in immunosuppressed pa-tients, especially those with depressed cell-mediated immunity, reactivation of HSV may be associated with prolonged viral excretion and persistence of lesions.
Related Topics