Chapter: Medical Microbiology: An Introduction to Infectious Diseases: Herpesviruses
Herpes Simplex : Clinical Aspects
HERPES SIMPLEX : CLINICAL ASPECTS
MANIFESTATIONS
Herpes Simplex Type 1
Infection with HSV-1 is usually “above the waist.” It consists characteristically of grouped or single vesicular lesions that become pustular and coalesce to form single or multiple ulcers. On dry surfaces, these ulcers scab before healing; on mucosal surfaces, they reepithelialize directly. Herpes simplex virus can be isolated from almost all ulcera-tive lesions, but the titer of virus decreases as the lesions evolve. Infections generally involve ectoderm (skin, mouth, conjunctiva, nervous system).
Primary infection with HSV-1 is often asymptomatic. When symptomatic, typically in children, it appears most frequently as gingivostomatitis, with fever and ulcerative lesions involving the buccal mucosa, tongue, gums, and pharynx. The lesions are quite painful, and the acute illness usually lasts 5 to 12 days. After this initial infection, HSV may be-come latent within sensory nerve root ganglia of the trigeminal nerve.
Lesions usually recur on a specific area of the lip and the immediate adjacent skin; these lesions are referred to as mucocutaneous and are commonly called “cold sores” or “fever blisters.” Because reactivation is usually from a single latent source, these lesions are typically unilateral. Their recurrence may be signaled by premonitory tingling or burn-ing in the area. Systemic complaints are unusual, and the episode generally lasts approxi-mately 7 days. It should be noted that HSV may be reactivated and excreted into the saliva with no apparent mucosal lesions present. Herpes simplex virus has been isolated from saliva in 5 to 8% of children and 1 to 2% of adults who were asymptomatic at the time.
Herpes simplex virus sometimes infects the finger or nail area. This infection, termed herpetic whitlow, usually results from the inoculation of infected secretions through asmall cut in the skin. Painful vesicular lesions of the finger develop and pustulate; they are often mistaken for bacterial infection and mistreated accordingly.
Herpes simplex virus infection of the eye is one of the most common causes of corneal damage and blindness in the developed world. Infections usually involve the conjunctiva and cornea, and characteristic dendritic ulcerations are produced. With recurrence of disease, there may be deeper involvement with corneal scarring. Occasionally, there may be extension into deeper structures of the eye, especially if topical steroids are used .
Encephalitis may rarely result from HSV-1 infection. Most cases occur in adults with high levels of anti–HSV-1 antibody, suggesting reactivation of latent virus in the trigeminal nerve root ganglion and extension of productive (lytic) infection into the temporoparietal area of the brain. Primary HSV infection with neurotropic spread of the virus from periph-eral sites up the olfactory bulb into the brain may also result in parenchymal brain infection.
Classically, HSV encephalitis affects one temporal lobe, leading to focal neurologic signs and cerebral edema. If untreated, mortality is 70%. Clinically, the disease can re-semble brain abscess, tumor, or intracerebral hemorrhage. Rapid diagnosis by the poly-merase chain reaction (PCR) has replaced brain biopsy as the diagnostic test. Intravenous acyclovir can reduce the morbidity and mortality of the disease, especially if treatment is initiated early.
Herpes Simplex Type 2
Genital herpes is an important sexually transmitted disease. Both HSV-1 and HSV-2 can cause genital disease, and the symptoms and signs of acute infection are similar for both viruses. Seventy percent of first episodes of genital HSV infection in the United States are caused by HSV-2, and genital HSV-2 disease is also more likely to recur than genital HSV-1 infection. Ninety percent of HSV-2 antibody–positive patients have never had a clinically evident genital HSV episode. In many instances, the first clinical episode is years after primary infection.
Primary Genital Herpes Infection
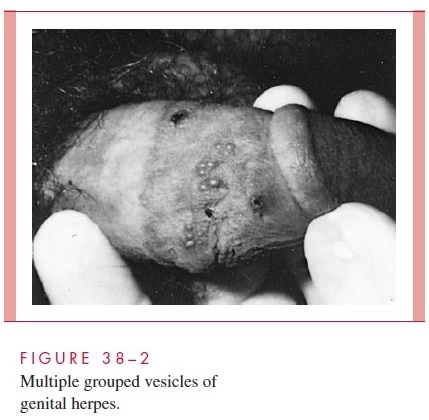
For the relatively few individuals who develop clinically evident primary genital HSV disease, the mean incubation period from sexual contact to onset of lesions is 5 days. Lesions begin as small erythematous papules that soon form vesicles and then pustules (Fig 38–2). Within 3 to 5 days, the vesiculopustular lesions break to form painful coalesced ulcers that subsequently dry; some form crusts and heal without scarring. With primary disease, the genital lesions are usually multiple (mean number, 20), bilateral, and extensive. The urethra and cervix are also infected frequently, with discrete or coalesced ulcers on the exocervix. Bilateral enlarged tender inguinal lymph nodes are usually present and may persist for weeks to months. About one third of patients show systemic symptoms such as fever, malaise, and myalgia, and approximately 1% develop aseptic meningitis with neck rigidity and severe headache. First episodes of disease last an average of 12 days.
Recurrent Genital Herpes Infection
In contrast to primary infection, recurrent genital herpes is a disease of shorter duration,
usually localized in the genital region, and without systemic symptoms. A common symptom is prodromal paresthesias in the perineum, genitalia, or buttocks that occur 12 to 24 hours before the appearance of lesions. Recurrent genital herpes usually presents with grouped vesicular lesions in the external genital region. Local symptoms such as pain and itching are mild, lasting 4 to 5 days, and lesions usually last 2 to 5 days.
At least 80% of patients with primary genital HSV-2 infection develop recurrent episodes of genital herpes within 12 months. In patients whose lesions recur, the median number of recurrences is four or five per year. They are not evenly spaced, and some patients experience a succession of monthly attacks followed by a period of qui-escence. Over time, the number of recurrences decrease by a median of one-half to one recurrence per year. Most recurrences result from reactivation of virus from dorsal root ganglia. Rarely, recurrent infections may be due to reinfection with a different strain of HSV-2. Recurrent viral shedding from the genital tract may occur without clinically ev-ident disease.
Neonatal Herpes
Neonatal herpes usually results from transmission of virus during delivery through infected genital secretions from the mother. In utero infection, although possible, is uncommon. In most cases, severe neonatal herpes is associated with primary infection of a seronegative woman at or near the time of delivery. This results in an intense viral exposure of a seronegative infant during the birth process. The incidence rate of neona-tal herpes simplex infection varies greatly among populations, but is estimated at ap-proximately 1 per 2500 live births in the United States. Because a normal immune re-sponse is absent in the neonate born to a mother with recent primary infection, neonatal HSV infection is an extremely severe disease with an overall mortality of approxi-mately 60%, and neurologic sequelae are high in those who survive. Manifestations vary. Some infants show disseminated vesicular lesions with a widespread internal or-gan involvement and necrosis of the liver and adrenal glands, and others have involve-ment of the central nervous system only, with listlessness and seizures.
DIAGNOSIS
Herpes simplex viruses are best cultured by isolation in a variety of other cell lines inocu-lated with infected secretions or lesions. The cytopathic effects of HSV can usually be demonstrated 24 to 48 hours after inoculation of the culture. Isolates of HSV-1 and HSV-2 can be differentiated by staining virus-infected cells with type-specific monoclonal anti-bodies to the two types. A direct smear prepared from the base of a suspected lesion and stained by either the Giemsa or Papanicolaou method may show intranuclear inclusions or multinucleated giant cells typical of herpes (Tzanck test), but this is less sensitive than viral culture and not specific; similar changes can be seen in cells infected with VZV. En-zyme immunoassays and immunofluorescence are rapid and relatively sensitive assays for direct detection of herpes antigen in lesions. Although early versions of these noncultural tests lacked sensitivity, more recent procedures have correlations with culture that ap-proach 90%. Serology should not be used to diagnose active HSV infections, such as those affecting the genital or central nervous systems; frequently there is no change in an-tibody titer when reactivation occurs. Serology can be useful in detecting those with asymptomatic HSV-2 infection. PCR on cerebrospinal fluid (CSF) is the best test to diag-nose HSV encephalitis. Restriction endonuclease digests can also be used to define epi-demiologic relationships; that is, strains acquired between sexual partners or through mother–infant transmission.
TREATMENT
Several antiviral drugs that inhibit HSV have been developed. The most effective and commonly used is the nucleoside analog acyclovir, which is converted by a viral en-zyme (thymidine kinase) to a monophosphate and then by cellular enzymes to the triphosphate form, which is a potent inhibitor of the viral DNA polymerase. Acyclovir significantly decreases the duration of primary infection and has a lesser but definite effect on recurrent mucocutaneous HSV infections. If taken daily, it can also suppress recurrences of genital and oral–labial HSV. In its intravenous form, it is effective in re-ducing mortality of HSV encephalitis and neonatal herpes. Acyclovir-resistant HSV has been recovered from immunocompromised patients with persistent lesions, especially those with acquired immunodeficiency syndrome (AIDS). Foscarnet is active against acyclovir-resistant HSV. In 1996, the U.S. Food and Drug Administration approved both valacyclovir and famciclovir for the treatment of recurrent genital HSV. Valacyclovir is a prodrug of acyclovir with better bioavailability (54% as opposed to 15–20%). It is rapidly converted to acyclovir and, in every characteristic except absorption, it is identi-cal to the parent compound. Valacyclovir is not more effective than acyclovir but can be given in lower dose and less frequently (500 mg twice daily). Famciclovir is the prodrug of another guanosine nucleoside analog, penciclovir. The bioavailability of penciclovir is also high (77%). After conversion, penciclovir must be phosphorylated, just like acy-clovir. Penciclovir has a much longer tissue half-life than acyclovir and can be given as 125 mg twice daily for treatment of recurrent genital HSV. Valacyclovir and famciclovir are now also approved for chronic suppression of recurrent genital HSV. No antiviral agents have been developed that decrease the long-term risk of subsequent reactivation of disease.
PREVENTION
Avoiding contact with individuals with lesions reduces the risk of spread; however, virus may be shed asymptomatically and transmitted from the saliva, urethra, and cervix by individuals with no evident lesions. Safe sex practices should reduce transmission. Al-though acyclovir has never been shown to reduce asymptomatic shedding from the geni-tal tract, studies are in progress to determine whether oral antivirals can actually dimin-ish transmission. Because of the high morbidity and mortality of neonatal infection, special attention must be paid to preventing transmission during delivery. Where active HSV lesions are present on maternal tissues, caesarean section may be used to minimize contact of the infant with infected maternal genital secretions, but caesarean section may not be effective if rupture of the membranes precedes delivery by more than several hours.
Related Topics