Chapter: Microbiology and Immunology: Microbial Pathogenesis
Growth and Multiplication of Bacteria at the Site of Adherence - Stages of Pathogenesis of Infections
Growth and Multiplication of
Bacteria at the Site of Adherence
Bacteria cause diseases by three main mechanisms: (a) inva-sion of tissues followed by
inflammation, (b) toxin production,
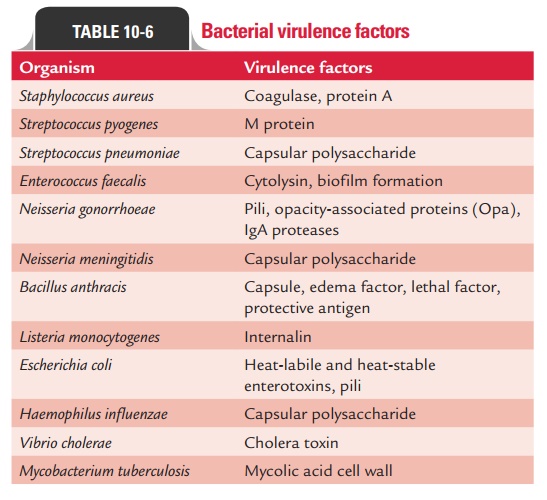
and (c) immunopathogenesis. Table
10-6 summarizes a list of bacteria with their virulence factors.
◗ Invasion of tissues followed by inflammation
Invasiveness refers to the ability of an organism to invade the host cells after establishing infection. “Invasion” is the term commonly used to describe the entry of bacteria into host cells, implying an active role for the organisms and a passive role for the host cells. For many disease-causing bacteria, invasion of the host’s epithelium is central to the infectious process. Some bacteria (e.g., Salmonella spp.) invade tissues through the intracellular junctions in the cytoplasm. Some bacteria (e.g., Shigella spp.) multiply within host cells, whereas other bacteria do not.
Shigella spp. initiate infection process by adhering to host cellsin the small
intestine. There are multiple proteins, including the invasion plasmid antigens (IpA-D), that contribute to the
process.Once inside the cells, the shigellae either are lysed or escape from
the phagocytic vesicle, where they multiply in the cytoplasm.
Other
bacteria (e.g., Yersinia species, N. gonorrhoeae, Chlamydiatrachomatis) invade specific types of the host’s
epithelial cellsand may subsequently enter the tissue. Once inside the host
cell, the bacteria may remain enclosed in a vacuole composed of the host cell
membrane, or the vacuole membrane may dis-solved and bacteria may disperse
within the cell and from one cell to another.
Invasion
of tissues followed by inflammation is enhanced by many factors, which include:
(a) enzymes, (b) antiphagocytic factors, (c)
biofilms, (d) inflammation, and (e) intracellular survival.
1. Enzymes: Invasion of bacteria is enhanced by many enzymes.Many species of
bacteria produce enzymes that are not intrin-sically toxic but do play
important roles in the infectious process. Some of these enzymes are discussed
below:
Hyaluronidases
and collagenase: Hyaluronidases
and col-lagenase are the enzymes that hydrolyze hyaluronic acid and degrade
collagen, respectively; thereby allowing the bacteria to spread through
subcutaneous tissues.
Hyaluronidases
are produced by many bacteria (e.g., staphylococci, streptococci, and
anaerobes) and aid in their spread through tissues. For example, hyaluroni-dase
produced by S. pyogenes degrades
hyaluronic acid in the subcutaneous tissue, thereby facilitating the organ-ism
to spread rapidly.
Clostridium perfringens produces the proteolyticenzyme collagenase,
which degrades collagen (the major protein of fibrous connective tissue), and
promotes the spread of infection in tissue.
Coagulase:
Staphylococcus aureusproduces the enzymecoagulase, which in association with blood
factors coagulates the plasma. Coagulase contributes to the formation of fibrin
walls around staphylococcal lesions, which protects bacteria from phagocytosis
by walling off the infected area. The enzyme also causes deposition of fibrin
on the surfaces of individual staphylococci, which may help protect them from
phagocytosis or from destruction within phagocytic cells.
Streptokinase
( fibrinolysin): Many
hemolytic strepto-cocci produce enzyme streptokinase, which activates a
proteolytic enzyme of plasma. This enzyme is then able to dissolve coagulated
plasma and thereby possibly aids in the rapid spread of streptococci through
tissues. Streptokinase has been used in the treatment of acute myocardial
infarction to dissolve fibrin clots.
IgA1 proteases: Certain pathogenic bacteria produceenzymes IgA1
proteases that split IgA1 at specific pro-line–threonine or proline–serine
bonds in the hinge region and inactivate its antibody activity. IgA1 protease
is an important virulence factor of the pathogens, such as gonorrhoeae, Neisseria
meningitidis, Haemophilus influenzae,and
Streptococcus pneumoniae. Production
of IgA1 protease allows the pathogens to inactivate the primary antibody found
on mucosal surfaces and thereby facilitates the attachment of these bacteria to
the mucous membrane.
2. Antiphagocytic factors: Many bacterial pathogens arerapidly killed once
they are ingested by polymorphonuclear cells or macrophages. Some pathogens evade
phagocyto-sis or leukocyte microbicidal mechanisms by several anti-phagocytic
factors; the most important being (a)
capsule, (b) cell wall proteins, (c) cytotoxins, and (d) surface antigens.
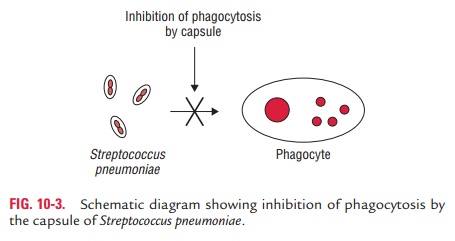
Capsule:
The capsule surrounding
bacteria, such as pneumoniae
(Fig. 10-3) and N. meningitidis, is
the mostimportant antiphagocytic factor. It retards the phago-cytosis of
bacteria by preventing the phagocytes from adhering to the bacteria.
Cell wall proteins: The cell wall
proteins, such as theprotein A and protein M, of S. aureus and S. pyogenes
especially are antiphagocytic. For example, protein A of S. aureus binds to IgG and prevents the activation of complement. M
protein of S. pyogenes is
antiphagocytic.
Cytotoxins: Certain bacteria produce
cytotoxins thatinterfere with chemotaxis or killing of phagocytes. For example,
S. aureus produces hemolysins and
leukocidins that lyse and damage RBCs and WBCs.
Surface antigens: Surface antigens
of bacteria, such as Viantigen of S.
typhi and K antigen of E. coli make
the bacteria resistant to phagocytosis and lytic activity of complement.
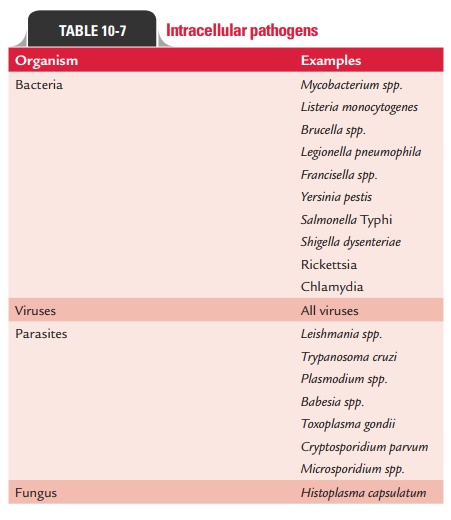
A list
of intracellular pathogens is given in Table 10-7
3. Biofilms: The biofilm is an aggregate of interactive bacteriaattached to a
solid surface or to each other and encased in an exopolysaccharide matrix.
Biofilms consist of single cells and microcolonies of bacteria, all found
together in a highly hydrated, predominantly anionic exopolymer matrix. This is
distinct from planktonic or free-living bacterial growth, in which interactions
of the microorganisms do not occur. Biofilms form a slimy coat on solid
surfaces and occur throughout the nature. A single species of bacteria may be
involved, or more than one species may coaggregate to form a biofilm. Fungi,
including yeasts, are occasionally involved.
Biofilms
are important in human infections that are persis-tent and difficult to treat.
A few such infections include:
a)
S. epidermidis and S.
aureus infections of central venouscatheters;
b) Eye infections that occur with contact lenses
and intraocular lenses;
c)
Infections
in dental plaque; and
d) Pseudomonas
aeruginosa airway
infections in cysticfibrosis patients.
4. Inflammation: Inflammation is an important host
defenseinduced by the presence of bacteria in the body. It is of two types:
pyogenic and granulomatous. Pyogenic inflammation is the host
defense seen primarily against pyogenic or pus-producing bacteria, such as S. pyogenes. It typically consists of
neutrophils and the production of specific antibodies and elevated level of
complement. Granulomatous inflam-mation is the host defense seen primarily
against intracel-lular granuloma-producing bacteria, such as Mycobacteriumtuberculosis, Mycobacterium leprae, etc. The response
consistsof production of macrophages and CD4+ T cells.
5. Intracellular survival: A few mechanisms that aresuggested for
intracellular survival of bacteria include (a)
inhibition of phagolysosome fusion, (b)
resistance to action of lysosomal enzymes, and (c) adaptation to cyto-plasmic replication as follows:
·
Bacteria
(such as Chlamydia, M. tuberculosis) that interfere with the
formation of phagolysosomes in a
hagocyte can survive intracellularly and evade host defense pro-cess.
These bacteria live within cells and are protected from attack by macrophages
and neutrophils. The bac-teria that do not interfere with the formation of
phagoly-sosomes are otherwise killed inside the phagocytes.
·
Presence
of capsular polysaccharide in Mycobacteriumlepraemurium
and mycoside in M. tuberculosis makesthese
bacteria resistant to action of lysosomal enzymes.
·
Certain
bacteria, such as rickettsiae, escape from the phagosome into the cytoplasm of
the host cell before the fusion of phagosome with lysosome takes place and
hence continue to remain intracellular
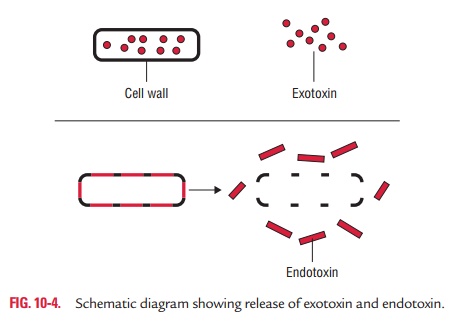
◗ Toxin production
Toxins
produced by bacteria are generally classified into two groups: exotoxins and
endotoxins.
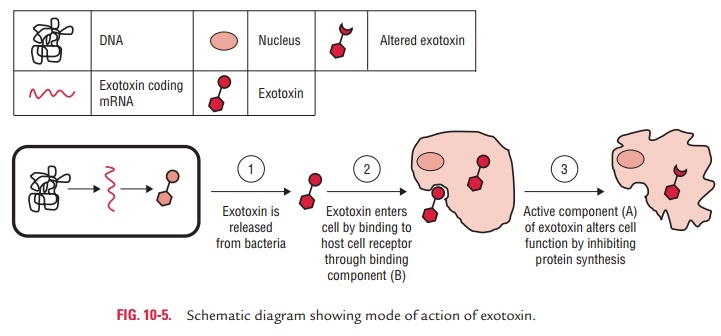
1. Exotoxins: Exotoxins are heat-labile proteins that are pro-duced by several Gram-positive and Gram-negative bacteria. These are bacterial products, which are secreted into tissues and directly harm tissues or trigger destructive biological activities (Fig. 10-4). The genes coding for these proteins are frequently encoded on plasmid or on bacteriophage DNA. Some important toxins encoded by plasmids are tetanus toxin of C. tetani and heat-labile and heat-stable toxins of enterotoxigenic E. coli. Toxins encoded by bacteriophage DNA are cholera toxins, diphtheria toxins, and botulinum toxin.
Superantigens:
Superantigens are special group of toxins.These
molecules activate T-cell nonspecifically by binding simultaneously to a T-cell
receptor and major histocom-patibility complex class II (MHC II) molecules on
another cell, without requiring antigen. Nonspecific activation of T cells
results in a life-threatening autoimmune-like response by producing a large
amount of interleukins, such as IL-1 and IL-2. Furthermore, stimulation of T
cells by superantigen can also lead to the death of activated T cell, resulting
in loss of specific T-cell clones and that of their immune response.
Staphylococcal enterotoxin (toxic shock syndrome toxin) of S. aureus and erythrogenic toxin of type A or C of S. pyogenes are examples of
superantigens.
2. Endotoxins: The term endotoxin was coined in 1893
byPfeiffer to distinguish the class of toxic substances released after lysis of
bacteria from the toxic substances (exotoxins) secreted by bacteria.
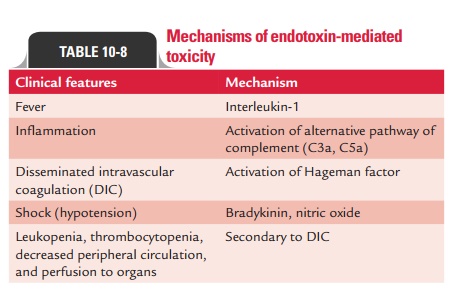
Biological
activity of endotoxin: Gram-negative
bacteriaproduce endotoxin during infection. The toxicity of endo-toxin is low
in comparison with exotoxins. All endotoxins usually produce the same
generalized effect of fever and shock. The lipid A protein of LPS is
responsible for endo-toxin activities (Table 10-8). The endotoxin binds to
specific receptors, such as CD14 and TLR4, present on macro-phages, B cells,
and other cells. Endotoxin exerts profound biological effects on the host and
may be lethal. Biological activities of the endotoxins include the following:
·
Mitogenic
effects on B lymphocytes that increase resis-tance to viral and bacterial
infections.
·
Production
of gamma interferon by T lymphocytes, which may enhance the antiviral state,
promote the rejection of tumor cells, and activates the macrophages and natural
killer cells.
·
Activation
of the complement cascade with the forma-tion of C3a and C5a.
·
Production
and release of acute-phase cytokines, such as IL-1, TNF-a (tumor necrosis factor-alpha), IL-6, and prostaglandins.
Endotoxic
shock: Endotoxins
at low concentration inducea protective response, such as fever, vasodilation,
and activation of immunity and inflammatory response. However, endotoxins at
very high concentration, as seen in blood of patients with Gram-negative
bacterial sepsis, cause a syndrome of endotoxic shock. Endotoxic shock is
characterized by fever, leukopenia, thrombocytope-nia, sudden fall of blood
pressure, circulatory collapse, and sudden death. This is because high
concentration of endotoxin can activate the alternative pathway of complement
and cause vasodilatation and capillary leak-age, resulting in high fever,
hypertension, and shock. It also causes activation of blood coagulation
pathway, leading to disseminated intravascular coagulation. Endotoxins are not
destroyed by autoclaving; hence infu-sion of sterile solution containing
endotoxins can cause serious illness.
Detection
of endotoxins in medical solutions: Endotoxins areomnipresent in the environment. Solutions for human
use (such as intravenous fluids) are prepared under carefully controlled
conditions to ensure sterility and to remove endotoxin. Representative samples
of every manufacturing batch are checked for endotoxins by one of two
procedures: (a) limulus lysate test
or (b) rabbit pyrogenicity test.
·
Limulus lysate test: The test depends on the ability ofendotoxin to
induce gelation of lysates of amoebocyte cells from the horseshoe crab Limulus polyphemus. Test kits are
commercially available. It is simple, fast, and sensitive to detect endotoxin
at a level of 1 ng/mL.
·
Rabbit pyrogenicity test: The test depends on the exquisitesensitivity of
rabbits to the pyrogenic effects of endo-toxin. In this test, a sample of the
solution to be tested is injected intravenously into the ear veins of adult
rab-bits, while the rectal temperature of the animal is moni-tored. Careful
monitoring of the temperature responses provides a sensitive and reliable
indicator of the pres-ence of endotoxin in the solution.
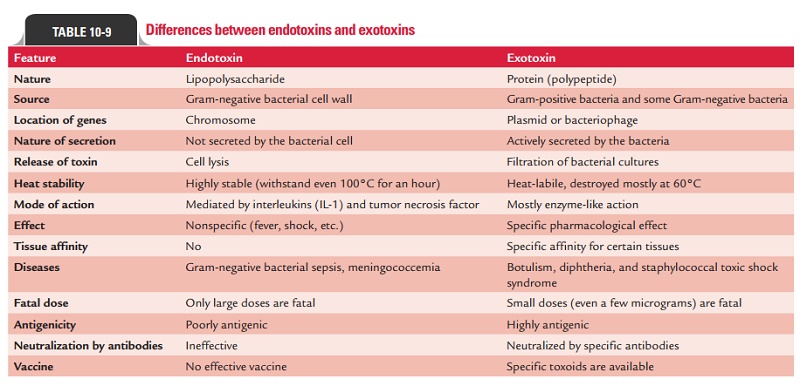
Table
10-9 summarizes differences between exotoxins and endotoxins.
◗ Immunopathogenesis
In certain diseases, the symptoms are caused not by the organism
itself, but due to immune response to the presence of organisms. For example,
immune complexes deposited in the glomerulus of the kidney cause
poststreptococcal glo-merulonephritis. Antibodies that are produced against the
M proteins of S. pyogenes cross-react
with joint, heart, and brain tissues producing disease manifestations of
rheumatic fever.
Similarly, the host immune response is an
important cause of disease symptoms in patients suffering from syphilis caused
by T. pallidum, Lyme disease caused
by Borrelia, and other diseases.
Related Topics