Chapter: Biotechnology: Plant Cell Culture and Applications
Gene transfer methods in plants
Gene transfer methods in plants
For achieving genetic transformation in plants, the basic pre-requisite is the construction of a vector (genetic vehicle) which carrys the genes of interest flanked by the necessary controlling sequences, i.e. promoter and terminator, and deliver the genes into the host plant. There are two kinds of gene transfer methods in plants as discussed below.
Vector-mediated or indirect gene transfer
Among the various vectors used in plant transformation, the Ti-plasmid of Agrobacteriumtumefacienshas been used extensively. This bacterium contains large size plasmid, known as Ti-plasmid (tumour-inducing plasmid) and portion of this plasmid referred as T-DNA(transferred DNA) is transferred to plant genome in the infected cells and cause plant tumours (crown galls -Fig. 7A). This means thatA. tumefacienshas natural ability to transfer T-DNA of its plasmid intoplant genome. (plant chromosomes) upon infection of cells at the wound site, and therefore this bacterium is known as 'natural genetic engineer of plants'. Because of this unique property, Ti-plasmid can be used as gene vectors for delivering useful foreign genes into plant cells and tissues. The foreign genes (transgenes), i.e., the gene of interest (e.g. Bt gene for insect resistance) and plant selection marker gene, usually an antibiotic gene like nptII which confer resistance to kanamycin are cloned in the T-DNA region of Ti-plasmid in place of unwanted DNA sequences(Fig. 7B).
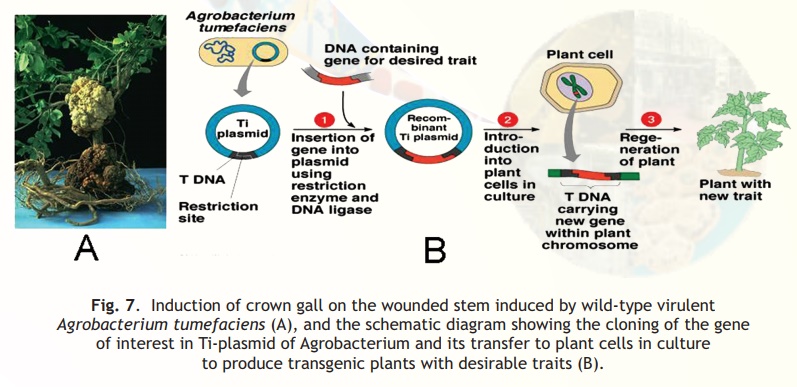
The general strategy for transforming plants is to collect leaf discs (in case of dicots) or embryogenic callus (in case of monocots like cereals) and then infect the tissue with Agrobacterium carrying recombinant disarmed Ti-plasmid vector (Fig. 7B). The infected tissue can then be cultured (co-cultivation) on shoot regeneration medium for 2-3 days during which time the transfer of T-DNA along with foreign genes takes place. Later, the transformed tissues (leaf discs/calli) are transferred onto selection cum plant regeneration medium supplemented with usually lethal concentration of an antibiotic (e.g. kanamycin) to selectively eliminate non-transformed tissues. This selection medium also contain a bacteriostatic agent, i.e. an antibiotic like cefotaxime which suppresses or kills the Agrobacterium present with the transformed tissues and is no longer needed as the transfer of foreign genes has already taken place during co-cultivation. After 3-5 weeks, the regenerated shoots (from leaf discs) are transferred to root-inducing medium, and after another 3-4 weeks, complete plants can be transferred to soil following the hardening (acclimatisation) of regenerated plants. The presence of foreign genes in the putative transgenic plants can be confirmed by molecular techniques like PCR and Southern blot hybridisation.
Vectorless or direct gene transfer
In the direct gene transfer methods, the foreign gene of interest is delivered into the host plant cell without the help of a vector. The following are some of the common methods of direct gene transfer in plants.
Chemical mediated gene transfer: Certain chemicals like polyethylene glycol (PEG) anddextran sulphate induce DNA uptake into plant protoplasts.
Microinjection: Here, the DNA is directly injected into plant protoplasts (specifically into thenucleus) using fine tipped (0.5-10 micrometer diameter) glass needle or micropipette to transform plant cells. The protoplasts are immobilized on a solid support (e.g. agarose on a microscopic slide) or held with a holding pipette under suction, and the DNA is injected into the protoplasts with the help of a fine microscope.
Electroporation: In this case, a pulse of high voltage is applied to protoplasts/cells/tissues,which makes transient (temporary) pores in the plasma-membrane which facilitates the uptake of foreign DNA. The DNA is mixed with the plant protoplasts in a specially made cuvettes before the application of the electric field.
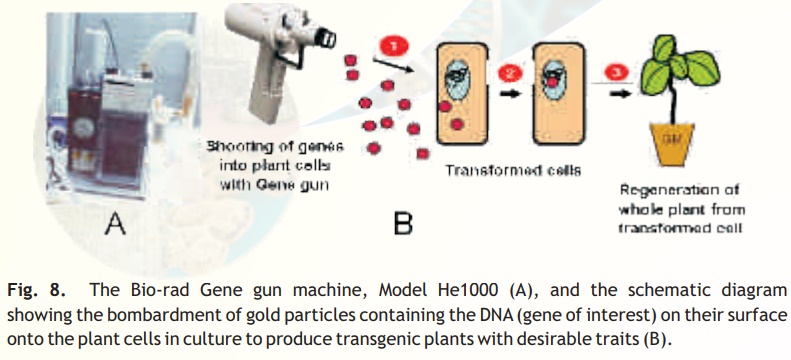
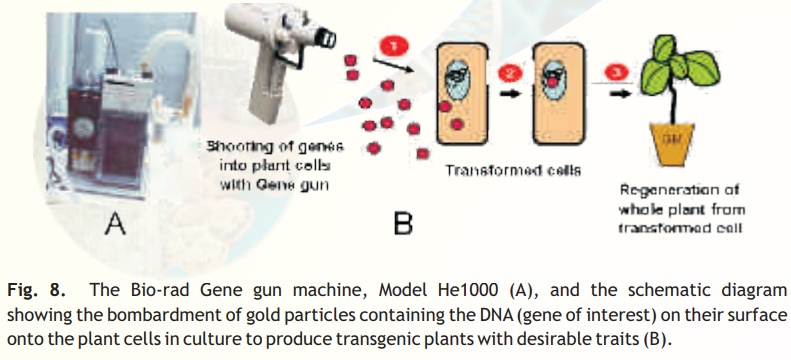
Particle gun: A popular and widely used direct gene transfer method for delivering foreign genesinto virtually any tissues and cells or even intact seedlings. In this method, the foreign DNA (containing the genes of interest) is coated (precipitated) onto the surface of minute gold or tungsten particles (1-3 micrometers) and bombarded (shot) onto the target tissue or cells using a particle gun (also called as gene gun/shotgun/microprojectile gun). Then, the bombarded cells or tissues are cultured on selection medium to regenerate plants from the transformed cells(Fig. 8).
Transgene analysis
The selection of transformed plant cells from untransformed cells is an important step in the plant genetic engineering. For selection, the gene of interest is transferred along with a genetic marker gene (usually antibiotic resistant genes such as nptII that confer resistance to kanamycin), so that only the transformed cells can be selected and regenerated on the selection medium containing the selection agent (e.g. kanamycin). The untransformed cells are eliminated on the selection medium as they are susceptible to the antibiotic (due to the absence of antibiotic resistance gene). The segregation and stability of the transgene integration and expression in the subsequent generations can be studied by genetic and molecular analyses (PCR, Southern, Northern and Western blot analysis).
Related Topics