Chapter: Basic Radiology : Musculoskeletal Imaging
Exercise: Local Musculoskeletal Disease
EXERCISE 6-2.
LOCAL DISEASE
6-6. Based on the
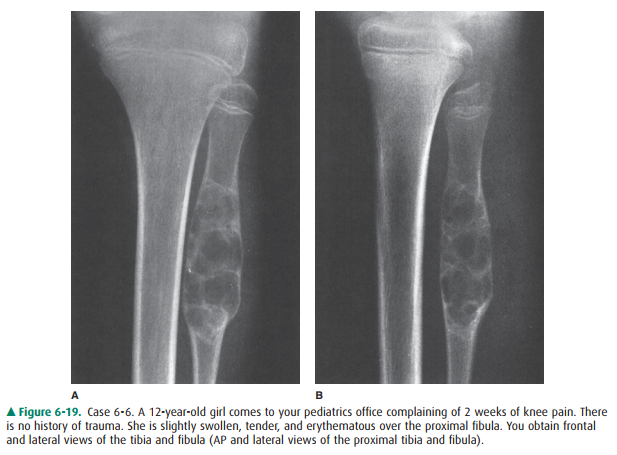
history, physical examination, and radi-ographs for Case 6-6 (Figure 6-19),
which of the fol-lowing choices is the best working diagnosis?
A.
A bone tumor, most likely benign
B.
A bone tumor, most likely malignant
C.
An infection of the bone
D.
A stress fracture of the proximal fibula
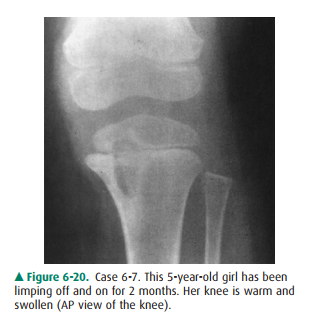
6-7. What is the most
likely diagnosis for Case 6-7 (Figure 6-20)?
A.
Osteomyelitis
B.
A malignant bone tumor
C.
A Salter-Harris IV fracture
D.
Langerhans cell histiocytosis
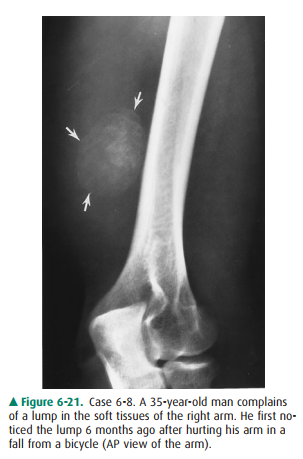
6-8. What should you do
about the calcified lump in the patient’s arm in Case 6-8 (Figure 6-21)?
A.
Needle biopsy
B.
Open excisional biopsy
C.
Reassure the patient
D.
Bone scan
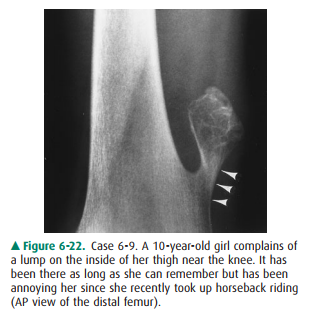
6-9. What is the lump in
Case 6-9 (Figure 6-22)?
A.
An osteosarcoma
B.
An osteochondroma
C.
A normal variant
D.
A soft tissue sarcoma
Radiologic Findings
6-6. Focal lytic lesion
in the proximal fibular metadia-physis with an intact shell of new cortex and a
well-defined, short zone of transition between itself and adjacent normal bone
(A is the correct answer to Question 6-6).
6-7. There is a
well-defined lytic lesion in the proximal tibia. Its edges are slightly
sclerotic. It extends across the physis to involve portions of both the
metaph-ysis and epiphysis (A is the correct answer to Ques-tion 6-7).
6-8. A well-defined
ossified mass projects in the muscula-ture of the posterolateral arm. It has a
thin but dis-tinct cortex (arrows) surrounding trabeculae (Figure 6-21) (C is
the correct answer to Question 6-8).
6-9. Arising from the
medial cortex of the femur is an ossified mass topped by a cauliflower-like
thin shell of cortex (Figure 6-22). The cortex of the remainder of the femur is
continuous with the cortex of the tumor (arrowheads), and the trabecular bone
of the femoral metaphysis blends imperceptibly with that of the mass. The mass
has grown away from its metaphyseal place of origin and points toward the
diaphysis and away from the joint (B is the correct answer to Question 6-9).
Discussion
Case 6-6: The radiographs
demonstrate a focal lytic lesion in the proximal fibular metadiaphysis. The
cortex appears intact around the lesion, and the bone is widened. Cortex is not
pli-able; it will not stretch to accommodate a growing lesion. In-stead it will
slowly remodel by resorption of endosteal bone and deposition of periosteal new
bone. The process takes time, so intact but extended cortex implies a slow
growth rate for this lesion. Another indication of a slow growth rate is the
sharp demarcation or short zone of transition between the lesion and adjacent
normal bone.
In general, osteomyelitis will
not cause apparent expan-sion of bone the way this lesion has. Stress fractures
are usu-ally linear lesions and usually are oriented transversely across the
bone, though there are exceptions. Stress fractures may be lucent, if a gap in
cortical bone is their primary man-ifestation, or sclerotic, either due to
compression of trabecu-lae with resultant overlap or due to healing. The
periosteal reaction that they engender may cause them to be mistaken for bone
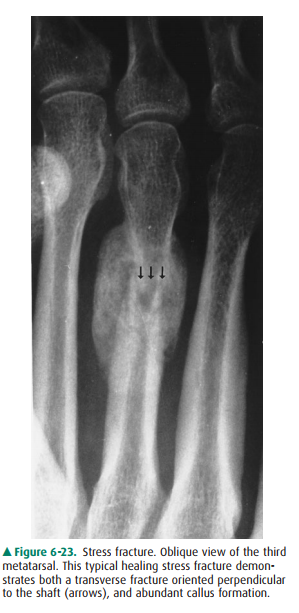
tumors, but they will not look like this particular le-sion (Figure 6-23).
Of the choices given in the
question, the remaining ones are benign and malignant bone tumor. For the most
part, malignant bone tumors in children have a rapid growth rate. This will
cause them to have poorly defined borders. In addi-tion, where they destroy
cortex, the periosteum will be unable to contain them with solidly mineralized
new bone, as has occurred here. There may be gaps in the cortex where tumor has
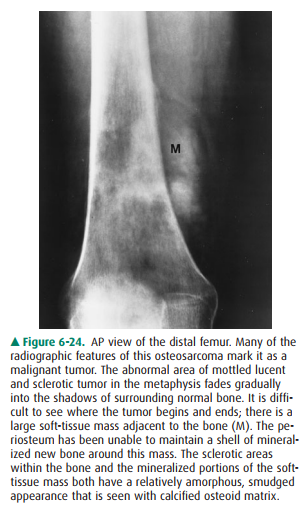
broken through (Figure 6-24). The periosteal new bone may mineralize at 90-degree
angles to the diaphysis or may be lamellated (like onion skin) or incomplete.
The intact shell of periosteal new bone seen in this patient and the short zone
of transition are more typical of a benign than a malignant tumor.
A primary bone tumor, no matter
how benign its appear-ance, is most appropriately handled by an orthopedic
oncologic surgeon experienced with tumors. As you have been stipulated to be a
pediatrician, the patient should be sent to an orthopedic surgeon who
specializes in the treatment of tumors.
Performing a percutaneous needle
biopsy has the poten-tial to cause great harm if a poorly chosen route is
taken. For example, if the needle passed close to the common peroneal nerve and
then the lesion proved unexpectedly to be malig-nant, the nerve might have to
be sacrificed in order to obtain a curative resection.
Obtaining additional imaging
studies to evaluate this lesion further is not a bad idea. It is better,
however, to allow the orthopedic surgeon to whom the patient will be referred
(in consultation with the radiologist) to decide which imaging tests are most
appropriate to evaluate the lesion more thoroughly rather than ordering
additional tests before referral.
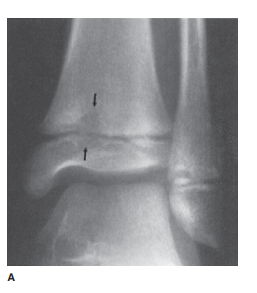
Case 6-7: The edges of malignant
tumors are usually not as well defined as those of this lesion. Malignant
tumors may extent across the growth plate, but it is uncommon for them to do so
while they are still as small as this lesion.
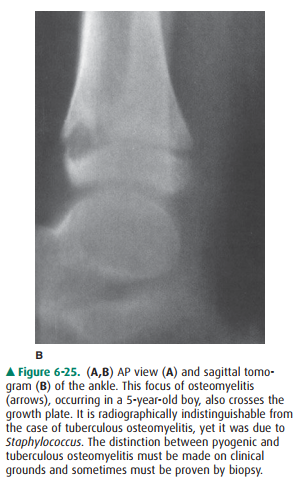
Osteomyelitis, on the other hand,
often breaches the growth plate. The most common organisms to cause
os-teomyelitis are species of Staphylococcus
and Streptococcus (Figure 6-25). The
relatively long history of limping, how-ever, should suggest a more indolent
organism. This case was due to Mycobacterium
tuberculosis. Skeletal tuberculo-sis is uncommon and thus often is
overlooked as a diagnos-tic possibility. Today, it is most commonly seen in the
immunocompromised individual. Because it is curable yet responds to very
different drugs than would be used for pyogenic osteomyelitis, it is important
to keep it in mind. It may occur at any site, but it is most common in the
spine. In the extremities, it most often occurs in or near the hip and knee.
Langerhans cell histiocytosis
(eosinophilic granuloma) is much less common than osteomyelitis and is thus not
as likely a diagnosis. When it does occur, its favorite location is the skull.
Case 6-8: This ossified mass represents
myositis ossificans, also known as heterotopic new bone formation. Though often
associated with trauma, it may also be seen in patients without a distinct
history of trauma. When it resembles ma-ture bone as closely as in this
patient, it is not a diagnostic dilemma, and you can reassure the patient that
there is a be-nign cause for his lump.
Occasionally, myositis ossificans
warrants excision on the basis of mechanical interference with the use of a
mus-cle or joint. Recurrence is less likely if excision is per-formed after the
lesion has matured. A bone scan may help to distinguish between mature and
immature lesions. An immature lesion that is still undergoing ossification will
exhibit marked radionuclide uptake. Once ossification is complete, radionuclide
accumulation will resemble that of other bones.
Myositis ossificans may be
diagnosed more confidently with radiography than with histology. An immature
lesion will be full of immature, rapidly proliferating cells that may be
mistaken for sarcoma by the pathologist. Radiologically, however, there is a
distinct difference between the two. Myositis ossificans ossifies from the
outside in. Sarcomas os-sify from the inside out. See Figure 6-24 and notice
that the central portion of the soft-tissue mass is denser (more ossi-fied)
than the outer portion. If it is not entirely clear from conventional
radiographs where and how the ossification is occurring, a CT scan is the test
of choice because of its ability to demonstrate calcium.
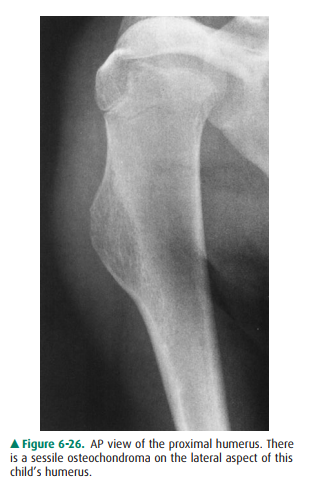
Case 6-9: This mass has the
characteristic appearance of an osteochondroma, the most common of all benign,
carti-laginous neoplasms. Osteochondromas may be very large or very small,
pedunculated or sessile (Figure 6-26). They grow as the child grows and should
cease growth by adulthood. Often asymptomatic, they may be an incidental
finding. They may, however, cause a wide range of symptoms. The most common
complaint is that they interfere with activities or with wearing certain
clothes, such as tight blue jeans. They may be painful as a result of
irritation of an overlying bursa (Figure 6-27). And they are subject to
fracture. An un-common (1% or less) but feared complication is malignant
transformation, usually resulting in chondrosarcoma. Signs of such
transformation include enlargement of the osteo-chondroma in an adult,
thickening of the cartilaginous cap that covers the tumor, development of a
soft-tissue mass, and destruction of bone.
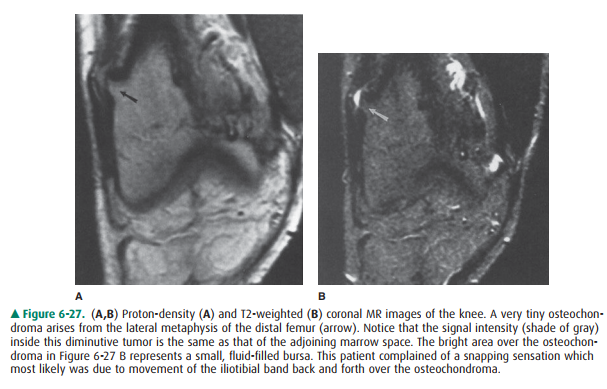
When further radiologic studies
are needed, MR imaging or ultrasound are probably the most useful modalities.
Both can demonstrate the cartilage cap and any associated soft tis-sue mass.
When the diagnosis is not as obvious as in this case by conventional
radiography, MRI can assist in confirming the identity of the tumor by
demonstrating continuity be-tween the cortices and medullary spaces of the
tumor and the host bone.
Related Topics