Chapter: Pharmaceutical Drug Analysis: Estimation of Phenols and Related Compounds
Estimation of Phenols and Related Compounds
ESTIMATION OF PHENOLS AND RELATED COMPOUNDS
INTRODUCTION
In oxidation-reduction methods bromine is employed as an
oxidizing agent in place of iodine, because it is reduced quantitatively be the
readily oxidized pharmaceutical organic substances in a reaction which results
in either water-insoluble bromine substitution products, for instance :
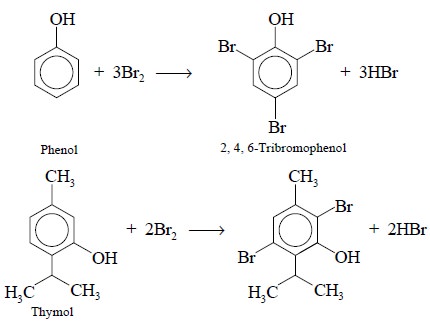
or corresponding water-insoluble bromine-addition
products, such as :
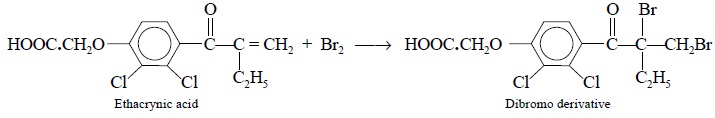
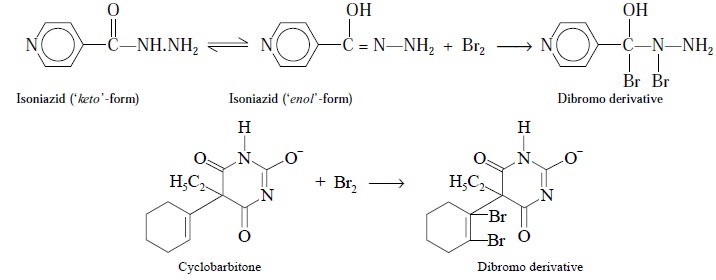
However, the standard solution used does not have bromine
(Br2) as such but it does contain an equivalent amount of potassium
bromate and an excess of potassium bromide and the resulting mixture on
subsequent acidification liberates bromine. The reaction may be expressed as
follows :
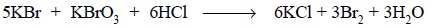
The liberated bromine helps in oxidizing iodide to an
equivalent amount of iodine as shown below :

The free iodine thus produced is titrated with previously
standardized sodium thiosulphate solution as depicted below :
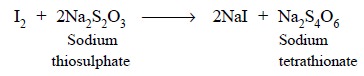
THEORY
In oxidation-reduction assays the use of bromine is
judiciously carried out as an oxidizing agent effectively for such specific
compounds which ultimately results into the formation of both bromine
substitution and bromine additive compounds. These products of reaction are
produced quantitatively and are mostly water-insoluble in characteristics ; and
more interestingly they take place in an acidic medium.
As it has been discussed earlier, iodine cannot be used
directly as an oxidizing agent in such type of assays, whereas the liberated
iodine quantitatively produced by the oxidation of iodide with bromine (excess)
may be assayed by titrating against sodium thiosulphate solution.
Related Topics