Chapter: Medical Physiology: Transport of Oxygen and Carbon Dioxide in Blood and Tissue Fluids
Effect of Hemoglobin to ÔÇťBufferÔÇŁ the Tissue PO2
Effect of Hemoglobin to ÔÇťBufferÔÇŁ the Tissue PO2
Although hemoglobin is necessary for the transport of oxygen to the tissues, it performs another function essential to life. This is its function as a ÔÇťtissue oxygen bufferÔÇŁ system. That is, the hemoglobin in the blood is mainly responsible for stabilizing the oxygen pressure in the tissues. This can be explained as follows.
Role of Hemoglobin in Maintaining Nearly Constant PO2 in the Tissues. Under basal conditions, the tissues requireabout 5 milliliters of oxygen from each 100 milliliters of blood passing through the tissue capillaries. Refer-ring back to the oxygen-hemoglobin dissociation curve in Figure 40ÔÇô9, one can see that for the normal 5 mil-liliters of oxygen to be released per 100 milliliters of blood flow, the PO2 must fall to about 40 mm Hg. Therefore, the tissue PO2 normally cannot rise above this 40 mm Hg level, because if it did, the amount of oxygen needed by the tissues would not be released from the hemoglobin. In this way, the hemoglobin nor-mally sets an upper limit on the oxygen pressure in the tissues at about 40 mm Hg.
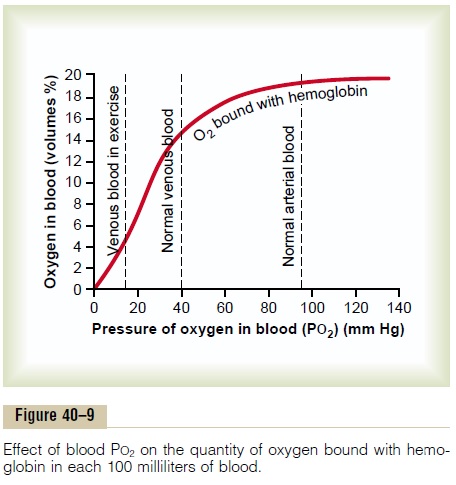
Conversely, during heavy exercise, extra amounts of oxygen (as much as 20 times normal) must be deliv-ered from the hemoglobin to the tissues. But this can be achieved with little further decrease in tissue PO2 because of (1) the steep slope of the dissociation curve and (2) the increase in tissue blood flow caused by the decreased PO2; that is, a very small fall in PO2 causes large amounts of extra oxygen to be released from the hemoglobin. It can be seen, then, that the hemoglobin in the blood automatically delivers oxygen to the tissues at a pressure that is held rather tightly between about 15 and 40 mm Hg.
When Atmospheric Oxygen Concentration Changes Markedly, the Buffer Effect of Hemoglobin Still Maintains Almost Constant Tissue PO2. The normal PO2in the alveoli is about104 mm Hg, but as one ascends a mountain or ascends in an airplane, the PO2 can easily fall to less than half this amount. Alternatively, when one enters areas of compressed air, such as deep in the sea or in pressur-ized chambers, the PO2 may rise to 10 times this level. Even so, the tissue PO2 changes little.
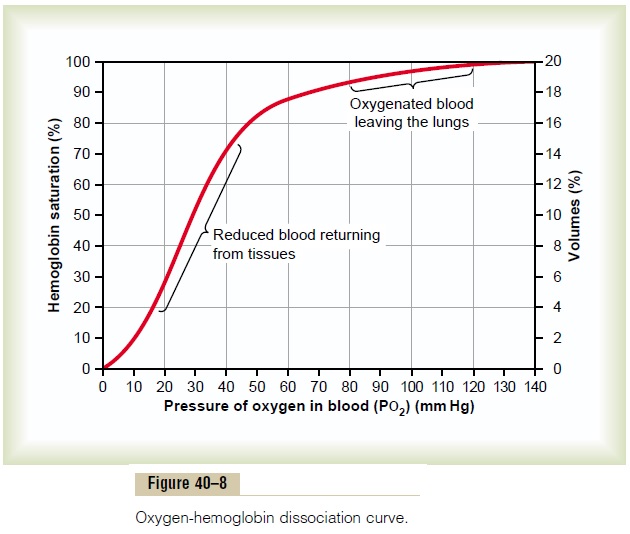
It can be seen from the oxygen-hemoglobin dissoci-ation curve in Figure 40ÔÇô8 that when the alveolar PO2 is decreased to as low as 60 mm Hg, the arterial hemo-globin is still 89 per cent saturated with oxygenÔÇöonly 8 per cent below the normal saturation of 97 per cent. Further, the tissues still remove about 5 milliliters of oxygen from each 100 milliliters of blood passing through the tissues; to remove this oxygen, the PO2 of the venous blood falls to 35 mm HgÔÇöonly 5 mm Hg below the normal value of 40 mm Hg. Thus, the tissue PO2 hardly changes, despite the marked fall in alveolar PO2 from 104 to 60 mm Hg.
Conversely, when the alveolar PO2 rises as high as 500 mm Hg, the maximum oxygen saturation of hemoglobin can never rise above 100 per cent, which is only 3 per cent above the normal level of 97 per cent. Only a small amount of additional oxygen dissolves in the fluid of the blood, as will be discussed subse-quently. Then, when the blood passes through the tissue capillaries and loses several milliliters of oxygen to the tissues, this reduces the PO2 of the capillary blood to a value only a few millimeters greater than the normal 40 mm Hg. Consequently, the level of alve-olar oxygen may vary greatlyÔÇöfrom 60 to more than 500 mm Hg PO2ÔÇöand still the PO2 in the peripheral tissues does not vary more than a few millimeters from normal, demonstrating beautifully the tissue ÔÇťoxygenbufferÔÇŁ function of the blood hemoglobin system.
Related Topics