Chapter: Basic & Clinical Pharmacology : Miscellaneous Antimicrobial Agents; Disinfectants,Antiseptics, & Sterilants
Disinfectants, Antiseptics, & Sterilants
DISINFECTANTS, ANTISEPTICS, &
STERILANTS
Disinfectants
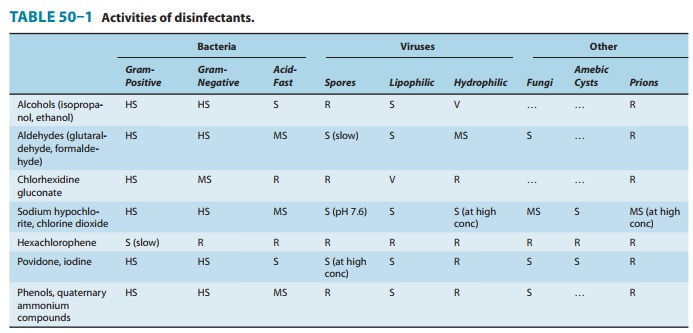
are strong chemical agents that inhibit or kill micro-organisms (Table 50–1).
Antiseptics are disinfecting agents with sufficiently low toxicity for host
cells that they can be used directly on skin, mucous membranes, or wounds.
Sterilants kill both veg-etative cells and spores when applied to materials for
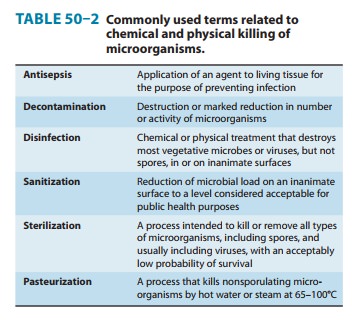
appropriate times and temperatures. Some of the terms used in this context are
defined in Table 50–2.
Disinfection prevents infection by reducing the number of potentially infective organisms by killing, removing, or diluting them. Disinfection can be accomplished by application of chemi-cal agents or use of physical agents such as ionizing radiation, dry or moist heat, or superheated steam (autoclave, 120°C) to kill microorganisms. Often a combination of agents is used, eg, water and moderate heat over time (pasteurization); ethylene oxide and moist heat (a sterilant); or addition of disinfectant to a detergent. Prevention of infection also can be achieved by washing, which dilutes the potentially infectious organism, or by establishing a barrier, eg, gloves, condom, or respirator, which prevents the pathogen from gaining entry to the host.
Hand hygiene is
the most important means of preventingtransmission of infectious agents from
person to person or from regions of high microbial load, eg, mouth, nose, or
gut, to poten-tial sites of infection. Soap and warm water efficiently and
effec-tively remove bacteria. Skin disinfectants along with detergent and water
are usually used preoperatively as a surgical scrub for sur-geons’ hands and
the patient’s surgical incision.
Evaluation of
effectiveness of antiseptics, disinfectants, and sterilants, although seemingly
simple in principle, is very complex. Factors in any evaluation include the
intrinsic resistance of the microorganism, the number of microorganisms
present, mixed populations of organisms, amount of organic material present
(eg, blood, feces, tissue), concentration and stability of disinfectant or
sterilant, time and temperature of exposure, pH, and hydration and binding of
the agent to surfaces. Specific, standardized assays of activity are defined
for each use. Toxicity for humans also must be evaluated. In the United States,
the Environmental Protection Agency (EPA) regulates disinfectants and
sterilants and the FDA regulates antiseptics.
Users of antiseptics,
disinfectants, and sterilants need to con-sider their short-term and long-term
toxicity because they may have general biocidal activity and may accumulate in
the environ-ment or in the body of the patient or caregiver using the agent.
Disinfectants and antiseptics may also become contaminated by resistant
microorganisms—eg, spores, P aeruginosa,
or Serratiamarcescens—and actually
transmit infection. Most topical antisep-tics interfere with wound healing to
some degree. Simple cleansing of wounds with soap and water is less damaging
than the applica-tion of antiseptics. Topical antibiotics with a narrow
spectrum of action and low toxicity (eg, bacitracin and mupirocin) can be used
for temporary control of bacterial growth and are generally pre-ferred to
antiseptics. Methenamine-containing agents releaseformaldehyde in a low
antibacterial concentration at acid pH and can be an effective urinary
antiseptic for long-term control of urinary tract infections.
Some
of the chemical classes of antiseptics, disinfectants, and sterilants are
described briefly in the text that follows. The reader is referred to the
general references for descriptions of physical disinfection and sterilization
methods.
Related Topics