Chapter: Health Management in Aquaculture: Disease development
Disease Diagnosis - Health Management in Aquaculture
DISEASE DIAGNOSIS
Disease diagnosis involves recognizing the occurrence of an abnormality and identifying its cause. Diagnosis of fish disease is a relatively new service avail-able only fairly recently and the range of laboratory procedures is still limited. The eventual findings are not always very definitive, though the elimination of certain possibilities may still prove useful. A meaningful diagnosis is most likely where the fish are under the closest supervision, and where the fish farmer is able to provide data on environmental parameters and management practices employed.
Signs of Diseases
A sick fish often exhibits some disease signs before it dies. The first indicationmay be reduced feeding. Abnormal changes in fish color and behavior are among the earliest signs seen in affected fish. The fish may stay away from the school, or swim at the surface or along the tank sides. The fish may also exhibit flashing, scraping on the bottom of projecting objects, darting, whirling or twisting, and final loss of equilibrium. In addition to these changes, body sur-face abnormalities and lesions may be observed externally or internally. Spe-cific disease signs associated with various diseases are presented with the dis-eases covered in this book.
Diagnosis
Fish disease diagnosis follows a format similar to that applied to other animal species, with more importance given to water quality parameters because of their direct effect on fish. A good history of the disease should be supported by personal observations before performing postmortem examinations. Following a gross appraisal, the most usual routine procedures are parasitological exami-nations, bacteriology, and histopathology. The last two procedures may require laboratory support. The following lists of on-site and laboratory procedures to investigate disease outbreaks are recommended by Anderson and Barney (1991):
On-site investigation:
· Examine fresh materials from healthy, moribund, and dead fish;
· Collect fish tissue samples;
· Measure environmental conditions (temperature, oxygen, etc.);
· Investigate physical factors and rearing conditions; and
· Gather information on time-course of mortalities.
· Deliver suitable samples with accompanying information to the diagnostic laboratory as soon as possible.
Laboratory procedures:
· Presumptive identification of pathogens (viral, bacterial, fungal, parasitic, etc.);
· Positive identification and confirmation;
· Test for drug sensitivity and effectiveness; and
· Evaluation of recommendations and reports of additional analysis (histopa-thology, toxicology, etc.).
Correct diagnosis is essential in selecting the best management approach to correct the problem and the best possible treatment for the disease. It will be useful as a reference for installing future disease-preventive aquaculture proce-dures and practices. Because diagnostics require suitable laboratory facilities and trained personnel, final diagnosis of fish disease should be performed in accredited Fish Health Laboratories.
Surveillance and Monitoring
Hatchery or farm personnel can initiate disease diagnosis at the farm level.This is done through regular monitoring of cultured populations, feed, and en-vironmental inputs to detect the presence of opportunistic pathogens. One must be familiar with the normal condition of the fish to be able to easily spot any changes or abnormalities. Without an adequate record of the normal health status of the fish, deviations from the normal cannot be assessed. Con-sistent record keeping is vital in monitoring the health status of any cultured population. The keeping of records is also necessary to enable farm personnel to identify environmental problems, learn from past mistakes, and to minimize production costs.
The need for regular monitoring is directly related to the intensification of the production system. For extensive systems without feed inputs, it may be enough to provide minimal monitoring. In hatchery operations, and when en-gaged in the semi-intensive or intensive systems, however, regular monitoring procedures should be applied. Some examples of needed monitoring proce-dures are periodic microscopic evaluation of gill tissues to assess the level of microbial gill fouling, daily microscopic examination of larvae from culture tanks, and evaluation of culture tank water algae and other feed inputs.
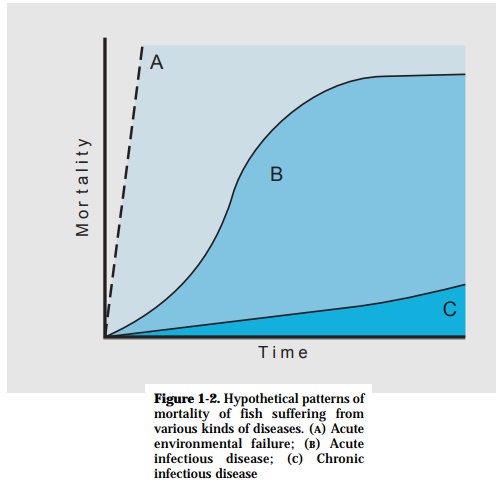
When a disease outbreak is encountered, data gathered on the pattern of losses, the species and sizes of fish affected, and the duration of the epizootic can provide a great deal of useful information. Sudden explosive die-off involv-ing all fish present usually indicates the presence of an acute environmental problem. This may be manifested by lack of oxygen, the presence of a lethal chemical toxicant, or by lethal temperatures. Mortality that starts with the ap-pearance of a few sick fish, unusual behavior, or loss of appetite can signal the onset of infectious disease. These diseases can be more or less acute or pro-longed in duration and the mortality ranging from high percentage of fish dying on a daily basis to low percentage of mortality over several weeks (Fig. 1-2).
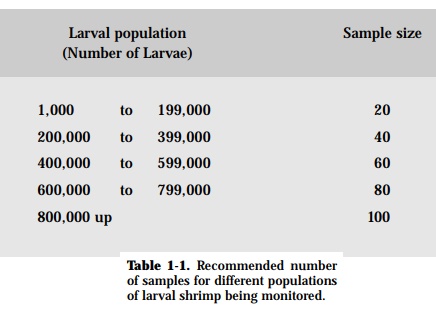
Ease in monitoring and collecting samples depends on the development stage and market value of the fish. The number of samples obtained from specific batches of fish is important. This is because adequate monitoring and diagno-sis are impossible without sacrificing animals. Table 1-1 shows the recom-mended number of samples needed to represent a certain population of hatch-ery-reared shrimp larvae being monitored.
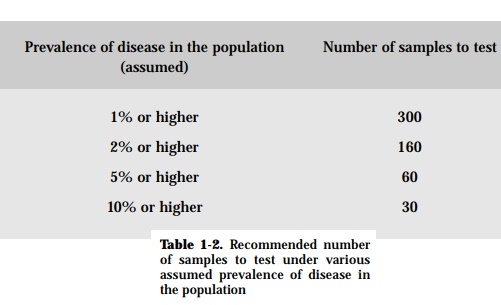
However, if the prevalence of a certain disease without clinical manifestation needs to be assessed, then the number of animals to be examined should be based on assumptions that would yield statistically significant results. Table 1-2 gives the number of samples to be examined given an assumed prevalence of the disease.
Related Topics