Chapter: Health Management in Aquaculture: Viral diseases
Characteristics of Virus - Health Management in Aquaculture
CHARACTERISTICS OF VIRUS
Viruses are ultramicroscopic organisms with size ranges of 10 to 300 nano-microns (nm). An electron microscope is required to visualize viruses. Because of their size, viruses are able to pass through filters of 0.5 micron pore size.
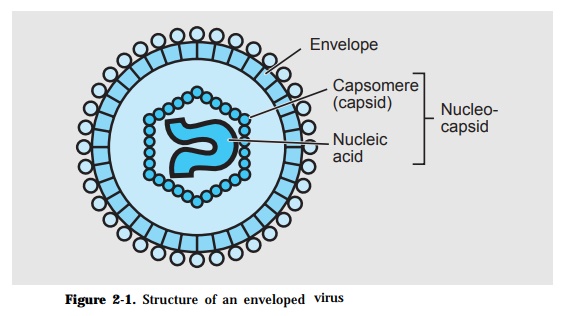
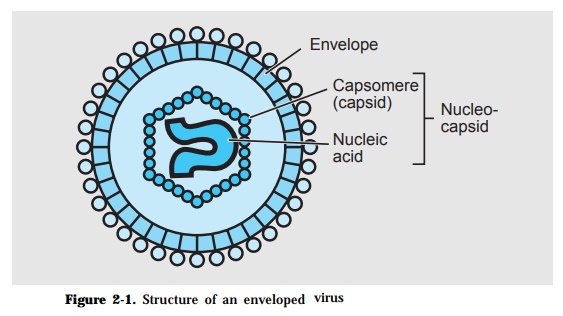
The basic structure of a virion consists of a capsid which encloses a nucleic acid genome (Fig. 2-1). The capsid is made up of identical protein subunits called capsomeres while the genome is either a ribonucleic acid (RNA) or a deoxyribonucleic acid (DNA). The combined viral components is called the nucleocapsid. This may have an envelop which is lipid in nature. Viruses that do not have an envelop are considered naked viruses.
Viruses have cubical/polyhedral, helical or complex morphology/symmetry. These microbes are obligately parasitic, multiplying only in its live host. In animal cells, the virus initially attaches on specific cell surface components called receptors. Subsequently, the whole virion penetrates the cell then makes use of the metabolic machinery and pathways of the living cell to make copies of its nucleic acid and synthesize protein subunits. Thereafter, these basic structural components are assembled to package the new viral particles. The new virions are usually released from the cell by lysis.
Cells infected by some virus develop inclusion/occlusion bodies within the in-fected cell as the replication cycle progresses. These are formed as a result of an accumulation of virions or viral components, although some inclusion bodies may not contain virions. These could be singular, multiple, intracytoplasmic or intranuclear and can be detected by histopathology using a compound micro-scope.
Aquatic viruses are transmitted from fish/shrimp to other fish/shrimp, from water to fish/shrimp or from reservoir to fish/shrimp by horizontal transmis-sion. Disease transmission can also result from brooder to eggs/fry via vertical transmission. Known reservoirs of viral pathogens are farmed fish/crustacean, imported fish/crustacean, wild fish/crustacean, other aquatic animals/plants and survivors of viral epizooties.
Diagnosis of viral infections can be made by a combination of various methods such as signs of disease, detection of inclusion/occlusion bodies, electron mi-croscopy (EM) or infection enhancement bioassay. Tissue culture techniques using established fish cell lines i.e. BB (Brown Bullhead), BF2 (Bluegill Fin), CCO (Channel Catfish Ovary), CFS (Catfish Spleen), CHSE (Chinook Salmon Embryo), EPC (Epithelioma Papulosum Cyprini), FHM (Fathead Minnow), GCK-84 (Grass Carp Kidney), GCG (Grass Carp Gonad) and GCF (Grass Carp Fin), RTG-2 (Raibow Trout Gonad), SBK-2 (Sea Bass Kidney), SHS (Snakehead Spleen), SSN-1 (Striped Snakehead Fry). To date, however, no continuous cell line from shrimp has been established.
Filtrates prepared from homogenized tissues of virus infected fish when inocu-lated onto monolayers of susceptible cells will result in lysis/destruction of the cells known as cytopathic effect (CPE) . Serial dilution of the filtrate will pro-vide information on the estimated titer of the virus expressed as tissue culture infection dose (TCID). Alternatively, the focal site of CPE induced by each virion will cause the development of a clear zone called plaque. In such plaque assay, the number of plaques formed indicates the estimated viral particles in a given sample.
Other techniques based on the principle of serology are also applied in the diagnosis of viral infections. The more commonly used tests include neutraliza-tion index (NI) determination, Western Blot, Enzyme-linked Immunosorbent Assay (ELISA), Fluorescent Antibody Technique (FAT) and Indirect Fluorescent Antibody Technique (IFAT). Recent molecular biology techniques such as Poly-merase Chain Reaction (PCR), Reverse Transciptase-Polymerase chain reaction (RT-PCR), DNA Probe have been developed and is currently widely applied for diagnosis of viral infections in shrimp.
In establishing the pathogenicity of a virus, provisions embodied in the River’s Postulates are followed:
· The virus must be present in the host cells, blood, or body fluids showing specific lesions at the time of the disease.
· Filtrates of infectious material, blood or tissue, shown not to contain bacte-ria or other visible cultivable pathogen in inanimate media, must produce the disease or specific antibody in appropriate animals.
· Similar filtrates from animals or plants must transmit the disease.
It is only when these provisions are complied with that a virus can be consid-ered pathogenic to fish or shrimp.
Related Topics