Chapter: Essential Clinical Immunology: Chronic Lymphocytic Leukemia
Correlations Between the Cellular and Molecular Features of the Disease With the Clinical Course of B-CLL
CORRELATIONS BETWEEN THE CELLULAR AND MOLECULAR FEATURES OF THE
DISEASE WITH THE CLINICAL COURSE OF B-CLL
How might features of the repertoire
of IgVH genes used by B
cells in B-CLL, the muta-tion status of these genes, and expression of
molecules related to cellular activation and BCR signal transduction (CD38 and
ZAP-70) be relevant to the clinical course of B-CLL? The disease manifests
differently in different patients, depending on the utilization of mutated or
unmutated IgVH genes and
the expression of ZAP-70 and CD38 by the leukemic cells (Figure 7.5). One
explanation is that activation via the BCR following recognition of
(self-)antigens activates the cells in vivo, accompanied by expression of CD38
and ZAP-70. Because the majority of U-CLL clones contain a self-reacting BCR,
while most M-CLL clones do not, it is not unexpected that more activa-tion
markers are found on U-CLL cells. In addition, B-CLL clones from patients in
different prognostic subsets differ in sig-naling capacity, with an intact BCR
signal transduction pathway found most frequently among patients exhibiting
unfa-vorable prognostic markers. Thus, con-tinuous (auto)antigenic stimulation
would likely represent a major factor for U-CLL cases and much less likely for
M-CLL.
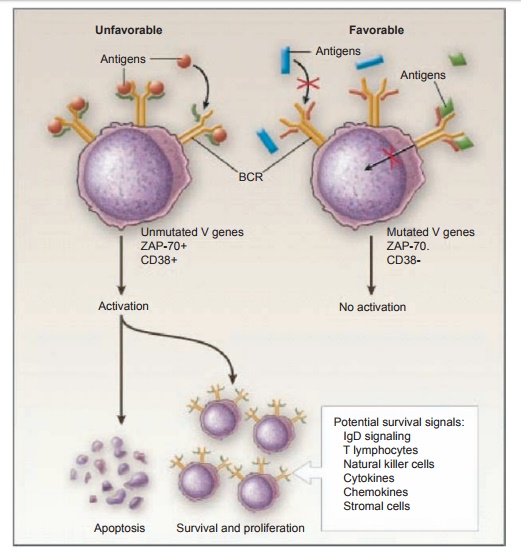
Figure 7.5 Differences in B cell’s receptor for antigen (BCR) signaling in the two subgroups of B-cell–type chronic lymphocytic leukemia (B-CLL). B-CLL clones from patients with unmutated IgVH genes (U-CLL) frequently differ from those from mutated IgVH (M-CLL) patients in the ability to transduce an activation signal through the BCR. In the case of U-CLL, patients that are more likely to have an unfavorable clinical course, BCR-mediated signals deliver an apoptotic signal unless influenced by other survival signals. In the case of M-CLL patients who are more likely to have a more favorable clinical course, BCR-mediated signals are ineffective either because the BCR has been altered and no longer binds available (auto)antigens or because after antigen binding the BCR is incapable of signaling due to anergy. Reprinted with permission from Chiorazzi N, Rai KR, Ferrarini M. Chronic lymphocytic leukemia. N Engl J Med. 2005;352:804–815.
Similarly, there is a rough correspon-dence between
the clinical course of patients and the development of chromosomal
abnormalities in their clones. Recurrent chromosomal lesions typically found in
B-CLL patients include deletions at 13q14.3, 11q22–23, 17p13, and 6q21, and
amplifica-tions of all or portions of chromosome 12. Deletion at 13q14.3 is
found in greater than half of B-CLL cases over time and is linked to loss of
two micro-RNAs that can regulate Bcl-2 expression.
However, this particular chromosomal
abnormality is not especially dangerous because patients exhibiting this
deletion on one allele and no other DNA lesions in their clones have a clinical
course that is benign and comparable to normal age-stratified individuals. In
contrast, deletions at 11q22–23, 17p13, and 6q21 are gen-erally associated
with more aggressive dis-ease, perhaps because these deletions may affect
important genes such as p53 (17p13
deletion), and ataxia telangiectasia
mutated (ATM; 11q22–23 deletion).
Longitudinal studies, albeit on a relatively limited num-ber of patients,
demonstrated that these ominous cytogenetic abnormalities accu-mulate
progressively in the course of the disease and more frequently in patients with
U-CLL.
These considerations are in line with the results of in vivo labeling experiments that involve incorporation of nonradioactive deuterium into newly synthesized DNA of dividing cells. These studies have shown B-CLL clones to be dynamic, hav-ing measurable birth rates from about 0.1 to >1.0 percent of the clone/day. Although only a minority of cells in a B-CLL clone can be shown to divide using this approach, estimates of the leukemic cell burden of a typical B-CLL patient are of the order of about 1012 cells, and therefore about 109 to 1010 new leukemic cells would be generated daily. Such rates of cell division are sufficient to permit more dangerous clonal variants to emerge and to influence clinical course and outcome over time.
Related Topics