Chapter: Organic Chemistry: Stereochemistry
Conformational isomers
CONFORMATIONAL ISOMERS
Key Notes
Definition
Conformational
isomers are different shapes of the same molecule resulting from rotation round
CŌĆōC single bonds. Conformational isomers are not dif-ferent compounds and are
freely interconvertable.
Alkanes
Alkanes
can take up different shapes or conformations due to rotation around the CŌĆōC
bonds. The most stable conformations are those where the bonds are staggered,
rather than eclipsed. The torsional angle in butane is the angle between the
first and third CŌĆōC bonds when viewed along the middle CŌĆōC bond. The most
stable conformation of butane has a torsional angle of 180 where the carbon
atoms and the CŌĆōC bonds are as far apart from each other as possible. The other
possible staggered conformation has a torsional angle of 60┬░which results in
some steric and electronic strain ŌĆō called a gauche interaction. The most
stable conformation for a straight chain alkane is zigzag shaped where all the
torsional angles are at 180 .
Cycloalkanes
Cycloalkanes
can adopt different conformations or shapes. The most stable conformation for
cyclohexane is the chair. Each carbon in the chair has two CŌĆōH bonds, one of
which is equatorial and one of which is axial. A chair structure can invert
through a high energy boat intermediate such that the equatorial bonds become
axial and the axial bonds become equatorial. If a substituent is present, the
most stable chair conformation is where the sub-stituent is equatorial. In the
axial position, the substituent experiences two gauche interactions with CŌĆōC
bonds in the ring.
Definition
Conformational isomers are essentially
different shapes of the same molecule resulting from rotation round CŌĆōC single
bonds. Since rotation round a single bond normally occurs easily at room
temperature, conformational isomers are not different compounds and are freely
interconvertable. Unlike constitutional and configurational isomers,
conformational isomers cannot be separated.
Alkanes
Conformational isomers arise from the rotation of CŌĆōC single bonds. There are many different shapes which a molecule like ethane could adopt by rotation around the CŌĆōC bond. However, it is useful to concentrate on the most distinctive ones (Fig.1). The two conformations I and II are called ŌĆśstaggeredŌĆÖ and ŌĆśeclipsedŌĆÖ respectively. In conformation I, the CŌĆōH bonds on carbon 1 are staggered with respect to the CŌĆōH bonds on carbon 2. In conformation II, they are eclipsed. Newman projections (Fig 2) represent the view along the C1ŌĆōC2 bond and emphasize the difference. Carbon 1 is represented by the small black circle and carbon 2 is represented by the larger sphere. Viewed in this way, it can be seen that the CŌĆōH bonds on carbon 1 are eclipsed with the CŌĆōH bonds on carbon 2 in conformation II.
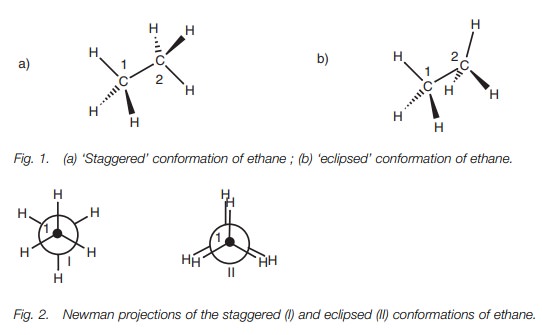
Of these two conformations, the staggered
conformation is the more stable since the CŌĆōH bonds and hydrogen atoms are as
far apart from each other as possible. In the eclipsed conformation, both the
bonds and the atoms are closer together and this can cause strain due to
electron repulsion between the eclipsed bonds and between the eclipsed atoms.
Therefore, the vast majority of ethane molecules are in the staggered conformation
at any one time. However, it is important to realize that the energy difference
between the staggered and eclipsed conformations is still small enough to allow
each ethane molecule to pass through an eclipsed con-formation (Fig. 3) ŌĆō otherwise CŌĆōC bond rotation
would not occur.
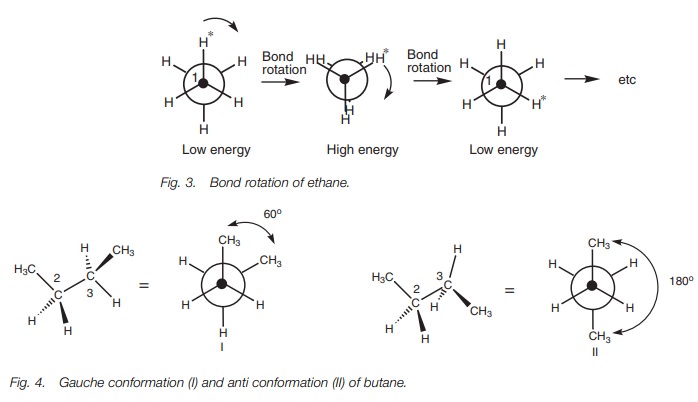
Ethane has only one type of staggered conformation, but different staggered conformations are possible with larger molecules such as butane (Fig. 4).
The first and the third CŌĆōC bonds in isomer I are at an
angle of 60┬░with respect to each other when viewed along the middle CŌĆōC bond.
In isomer II, these bonds are at an angle of 180┬░ . This angle is known as the torsional angle or dihedral angle. Iso-mer II is more stable than isomer I. This is
because the methyl groups and the CŌĆōC bonds in this conformer are as far apart
from each other as possible. The methyl groups are bulky and in conformation I
they are close enough to interact with each other and lead to some strain.
There is also an interaction between the CŌĆōC bonds in isomer I since a
torsional angle of 60┬░ is
small enough for some electronic repul-sion to exist between the CŌĆōC bonds.
When CŌĆōC bonds have a torsional angle of 60┬░ , the steric and electronic repulsions which
arise are referred to as a gaucheinteraction.
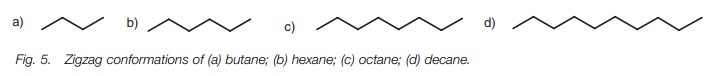
As a result, the most stable conformation for
butane is where the CŌĆōC bonds are at torsional angles of 180┬░which results in a
ŌĆśzigzagŌĆÖ shape. In this conformation, the carbon atoms and CŌĆōC bonds are as far
apart from each other as possible. The most stable conformations for longer
chain hydrocarbons will also be zigzag (Fig.5).
However, since bond rotation is occurring all the time for all the CŌĆōC bonds,
itis unlikely that many molecules will be in a perfect zigzag shape at any one
time.
Cycloalkanes
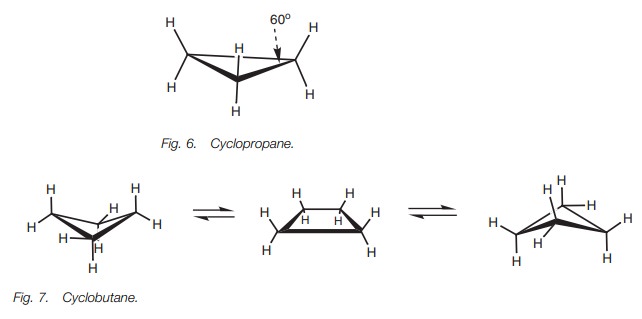
Cyclopropane (Fig. 6) is a flat molecule as far as the carbon atoms are
concerned, with the hydrogen atoms situated above and below the plane of the
ring. There are no conformational isomers. Cyclobutane (Fig. 7) on the other hand can form three distinct shapes ŌĆō a planar
shape and two ŌĆśbutterflyŌĆÖ shapes. Cyclopentane (Fig. 8) can also form a variety of shapes or conformations. The
planar structures for cyclobutane and cyclopentane are too strained to exist in
practice due to eclipsed CŌĆōH bonds.
The two main conformational shapes for cyclohexane are known as the chair and the boat (Fig. 9). The chair is more stable than the boat since the latter has eclipsed CŌĆōC and CŌĆōH bonds. This can be seen better in the Newman projections (Fig. 10) which have been drawn such that we are looking along two bonds at the same time ŌĆō bonds 2ŌĆō3 and 6ŌĆō5. In the chair conformation, there are no eclipsed CŌĆōC bonds.
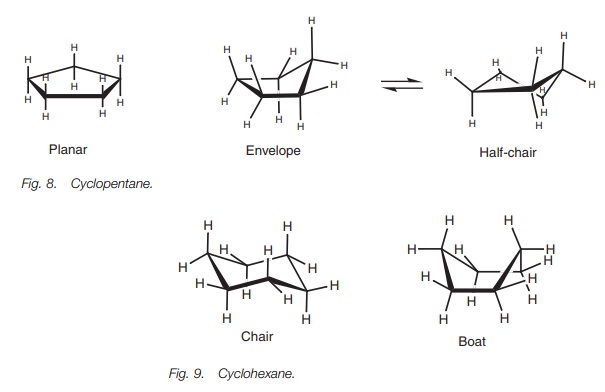
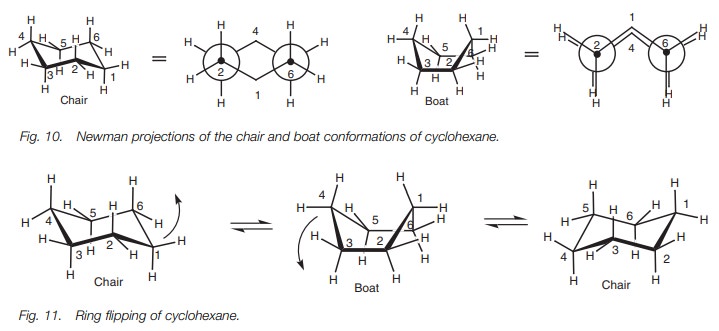
However, in the boat conformation,
bond 1ŌĆō2 is eclipsed with bond 3ŌĆō4, and bond 1ŌĆō6 is eclipsed with bond 5ŌĆō4.
This means that the boat conforma-tion is less stable than the chair
conformation and the vast majority of cyclohexane molecules exist in the chair
conformation. However, the energy barrier is small enough for the cyclohexane
molecules to pass through the boat conformation in a process called ŌĆśring
flippingŌĆÖ (Fig. 11). The ability of a
cyclohexane molecule to ring-flip is important when substituents are present.
Each carbon atom in the chair structure has two CŌĆōH bonds, but these are not
identical (Fig. 12). One of these
bonds is termed equatorial since it
is roughly in the plane of the ring. The other CŌĆōH bond is vertical to the
plane of the ring and is called the axial
bond.
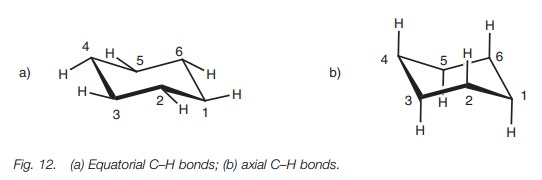
When ring flipping takes place from one chair to another, all the axial bonds become equatorial bonds and all the equatorial bonds become axial bonds. This does not matter for cyclohexane itself, but it becomes important when there is a substituent present in the ring. For example, methylcyclohexane can have two chair structures where the methyl group is either on an equatorial bond or on an axial bond (Fig. 13).
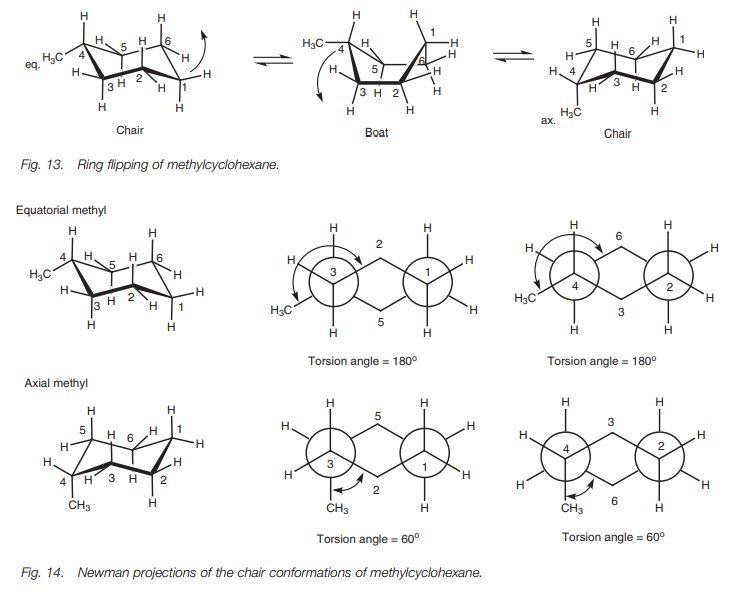
These are different shapes of the same molecule which are interconvertable due to rotation of CŌĆōC single bonds (the ring flipping process). The two chair structures are conformational isomers but they are not of equal stability. The more stable conformation is the one where the methyl group is in the equatorial position. In this position, the CŌĆōC bond connecting the methyl group to the ring has a torsional angle of 180┬░with respect to bonds 5ŌĆō6 and 3ŌĆō2 in the ring. In the axial position, however, the CŌĆōC bond has a torsional angle of 60┬░with respect to these same two bonds. This can be illustrated by comparing Newman diagrams of the two methylcyclohexane conformations (Fig. 14).
A torsion angle of 60┬░between CŌĆōC bonds
represents a gauche interaction and so an axial methyl substituent experiences
two gauche interactions with the cyclo-hexane ring whereas the equatorial
methyl substituent experiences none. As a result, the latter chair conformation
is preferred and about 95% of methylcyclo-hexane molecules are in this
conformation at any one time, compared to 5% in the other conformation.
Related Topics