Chapter: Physics : Conducting Materials
Conducting Materials
CONDUCTING MATERIALS
1
Introduction
1.1.Basic
Terminologies
2
Conducting Materials
3
Electron Theory of solids
4
Classical Free Electron Theory
4.1.
Drude – Lorentz Theory
4.2.
Postulates of Classical Free Electron Theory
4.3.
Derivation of Electrical conductivity
4.4.
Thermal Conductivity
5
Wiedemann - Franz Law
6
Lorentz Number
7
Merits of classical free electron theory
8
Drawbacks of classical free electron theory
9
Quantum Free Electron Theory
10
Fermi Dirac Distribution Function
10.1.
Effect of Temperature
11
Density of Energy States
11.1.
Definition
11.2.
Derivation
11.3.
Calculation of Carrier Concentration at 0 K
11.4.
Calculation of Fermi Energy
11.5.
Average Energy of an Electron at 0 K
1 INTRODUCTION
Conducting materials
are low resistivity materials, which conduct heat as well as electricity.
Electrical conduction is due to free electrons, whereas normal conduction is
due to free electrons as well as phonons.
1.1. BASIC
TERMINOLOGIES
Conductors
Experimental measurements showed that the metals and
their alloys exhibit large electrical conductivity in the order of 10 Ω m .
Hence they are known as conductors Conducting materials are the materials
having high electrical and thermal conductivities. Low resistive materials are
also generally known as conducting materials.
Bound Electrons
All the valence
electrons in an isolated atom are bound to their parent nuclei which are called
as ‘ bound electrons’
Free Electrons
In a solid, due to the
boundaries of neighbouring atoms overlap each other, the valence electrons find
continuity from atom to atom. Therefore, they can move easily throughout the
solid. All such valence electrons of its constituent atoms in a solid are
called free electrons.
Difference Between Ordinary Gas and Free
Electron Gas
The molecules
of ordinary gas are neutral. But, the free electron gas is charged. The density
of molecules is smaller than the density of free electrons.
Electric Field (E)
The
electric field (E) of a conductor having uniform cross section is defined as the potential
drop (V) per unit length (l).
E = V/ l
Unit –Vm-1
Current Density (J)
Current density (J) is
defined as the current per unit area of cross section of an imaginary plane
hold normal to the direction of flow of current in a current carrying
conductor.
If ‘I’ is
the current, and
‘A’ is the
area of
J = I / A Unit –A m-2
2 CONDUCTING MATERIALS
Conducting
materials are classified in to three major categories based on the
conductivity.
(i). Zero resistive
materials (ii). Low resistive materials (iii). High resistive materials
(i)Zero resistive materials
The super conductors like alloys of
aluminium, zinc, gallium, niobium, etc., are a special class
of materials. These
materials conduct electricity almost with zero resistance blow transition
temperature. Thus, they are called zero resistive materials.
These materials are
used for saving energy in the power systems, super conducting magnets, memory
storage elements etc.,
(ii)Low resistive materials
The metals like silver,
aluminium and alloys have high electrical conductivity. These materials are
called low resistive materials.
They are used as conductors, electrical co
nduct etc., in
electrical devices and electrical power transmission and distribution, winding
wires in motors and transformers.
(iii)High resistive materials
The materials like
tungsten, platinum, nichrome etc., have high resistive and low temperature
co-efficient of resistance. These materials are called high resistive
materials.
Such a metals and
alloys are used in the manufacturing of resistors, heating elements, resistance
thermometers.
The conducting
properties of solid do not depend on the total number of the electrons
available because only the valance electrons of the atoms take part in the
conduction. When these valance electrons detached from the orbit they are
called free electrons or conduction electrons.
In a metal, the number
of free electrons available is proportional to its electrical conductivity.
Hence, electronic structure of a metal determines its electrical conductivity.
3 ELECTRON THEORY OF SOLIDS
We know that the
electrons in the outermost orbit of the atom determine the electrical
properties in the solid. The free electron theory of solids explains the
structure and properties of solids through their electronic structure.
This theory is applicable to all solids, both metals
and non metals. It explains
(i). The behavior of
conductors, semiconductors, and insulators. (ii). The electrical, thermal and
magnetic properties of solids.
So far three electron theories have been proposed.
(i). Classical free electron theory
It is a macroscopic
theory, proposed by Drude and Lorentz in1900. According to this theory, the
free electrons are mainly responsible for electrical conduction in metals. This
theory obeys laws of classical mechanics.
(ii). Quantum free electron theory
It is a microscopic
theory, proposed by sommerfeld in 1928. According to this theory, the electrons
in a metals move in a constant potential. This theory obeys laws of quantum
mechanics.
(iii). Zone theory or band theory of solids
Bloch proposed this theory in the year
1928. According to this theory, the free electrons move in a periodic
potential. This theory explains electrical conductivity based on the energy
bands.
4 CLASSICAL FREE ELECTRON MODEL
Free electron theory of
metals was proposed by P. Drude in the year 1900 to explain electrical
conduction in metal. This theory was further extended by H.A. Lorentz in the
year 1909.
4.1. DRUDE LORENTZ THEORY
Principle
According to this
theory, a metal consists of a very large number of free electrons. These free
electrons can move freely throughout the volume of the metal. They are fully
responsible for the electrical conduction in the metal.
Explanation
We know that an atom consists of a
central nucleus with positive charge surrounded by the electrons of negative
charge. The electros in the inner shells are called core electros and those in
the outermost shell are called valence electrons.
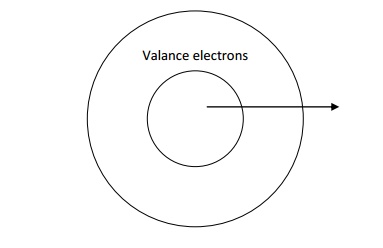
4.2.POSTULATES OF FREE ELECTRON THEORY
Drude assumed that the
free electrons in a metal form an electron gas. These free electrons move
randomly in all possible directions just like the gas molecules in a container.
In the absence of electrical field
When an electrical field is not applied,
the free electrons move everywhere in a random manner. They collide with other
free electrons and positive ion core. This collision is known as elastic
collision.
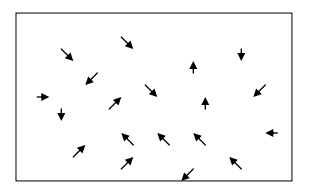
In the presence of electric field
When the electrical field is applied,
the electrons get some amount of energy from the applied electric field and
they begin to move towards the positive potential. (In the opposite direction
to the applied electric field).
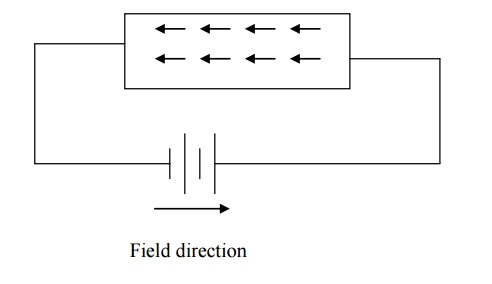
Since electrons are assumed to be a
perfect gas, they obey the laws of kinetic theory of gases.
Drift velocity (v d)
It is defined as the average velocity
acquired by the free electrons in a metal in a particular direction by the
application of an electrical field.
Mean free path
(λ)
The average distance travelled by a free
electron between any two successive collisions in the presence of an applied
electric field is known as mean free path. It is the product of drift velocity
of free electron and collision time ( ι c).
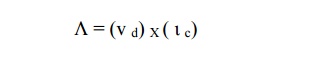
Collision time ( ι c)
The average time taken by a free
electron between any two successive collisions is known as collision time. It
is expressed mathematically as
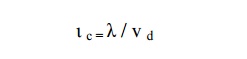
Relaxation time (ι)
The average time taken by a free
electron to reach its equilibrium position from its disturbed position due to
the application of an external electrical field is called relaxation time. It
is approximately equal to 10-14 second.
4.3 DERIVATION OF ELECTRICAL
CONDUCTIVITY
Definition
The amount of electrical charge conducted (Q) per
unit time across unit area (A) of a solid given by
σ = Q
/ tAE
If
t = 1second E = 1 volt, A = 1 metre 2
σ = Q
σ = Q / tAE = J / E
Where J is the current density and it is
given by Q / tA. Also J = σ E ( according to ohm’s law )
Expression for electrical conductivity
We know in the absence
of external electric field, the motion of electrons in a metal moves randomly
in all directions. When electric fi rod, the electron moves in opposite
direction to the applied field with velocity v d. this velocity is
known as drift velocity.
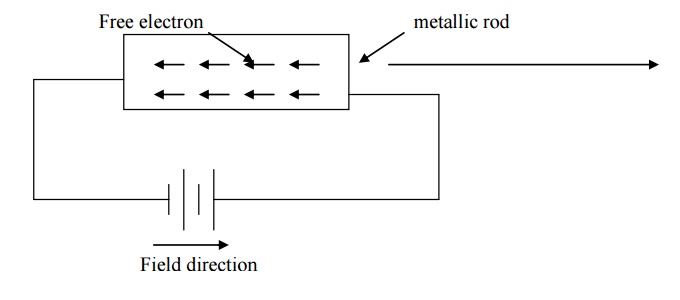
Lorentz force acting on
the electron F = eE --- (1)
This force is known as
driving of the electron. Due to this force, the electron gains acceleration
‘a’. From motion, the Newton’s force second law of
F= ma --- (2)
From equations 1 &
2
ma =
eE(or)
a = eE / m -- (3)
The acceleration of
electron is given by
Acceleration (a) = Drift velocity (v d) /
Relaxation time (τ)
a = v d / τ
v d =
a τ
Substituting equation (3) in( 4)
v d
=[ e τ
/ m ]
E ---(5)
Where ‘σ’ is the electrical conductivity of velocity
is given as
J = ne v d --(6)
Substituting equation (5)in (7), we have
J = ne [e τ / m] E ---(7)
J = [ne2 τ / m] E , in terms of effective
mass m* of an electron,
J = [ne2 τ / m* ] E ---(8)
From microscopic form of Ohm’s law, the current
density ‘J’ is expressed as,
J = σ E
---(9)
On comparing equations (8) & (9) , we have
Electrical conductivity σ = ne2 τ / m*
From equation (10), we know that with increase of
electron concentration ‘n’ , the conductivity ‘σ’ increases. As m* increases,
the motion of electron becomes slow and the electrical conductivity.
4.4.THERMAL CONDUCTIVITY
Definition
It is defined as the
amount of heat flowing per unit time through the material having unit area of
cross section per unit temperature gradient.
ie).,
Q = K dT/ dx
Thermal conductivity of
the material
K = Q / dT/dx
Q
–Amount of heat flowing per unit time through unit cross sectional area
dT/dx
–Temperature gradient.
Expression for thermal conductivity
Let us consider a
uniform rod AB with temperatures T1 (Hot) at end A and T2
(cold) at end B. Heat flows from hot end A to the hold end B. Let us consider
cross sectional area C which is at a distance equal to the mean free path (λ)
of t in fig.
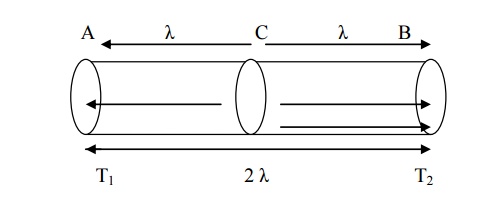
The conduction electron
per unit value is n and average velocity of these electrons is v.
During the movement of
electrons in the rod, the collision takes place. Hence, the electrons near A
lose their kinetic energy while electrons near B gain kinetic energy.
At A, average kinetic
energy of en electron = 3/2 kT ------(1)
Where k-
Boltzmann’s constant and
T is temperature
At B average kinetic
energy of the electron = 3/2 k (T –dT) ------(2)
The excess of kinetic energy
carried by the electron from A to B
= 3/2 kT - 3/2 k (T
–dT)
= 3/2 kT - 3/2 kT + 3/2
k dT ------(3)
Number of electrons
crossing per unit area per unit time from A to B
= 1/6 nv.
The excess of energy
carried from A to B per unit area in unit time
= 1/6 nv X 3/2 kdT
= 1/4 n v k dT
------(4)
Similarly, the
deficient of energy carried from B to A per unit area per unit time
= -1/4 n v kdT
------(5)
Hence, the net amount
of energy transferred from A to B per unit area per unit time
Q =1/4 nvkdT –(-1/4
nvkdT )
Q = 1/4 nvkdT + 1/4
nvkdT
Q = 1/2 nvkdT
------(6)
But from the basic
definition of thermal conductivity, the amount of heat conducted per unit area
per unit time
Q = K
dT/ λ
[ Q
= K dT/
dx ; λ = dx]
½ n u
k dT = K
dT/ λ
K = ½ nv k λ (7)
We
know that for the metals
Relaxation time = collision time
τ = τ c = λ
/ v
τ v = λ ---(8)
Substituting equation 8
in the equation 7, we have
K = ½ n v k τ v
K = ½ n v2 k
τ ---(9)
5
WIDEMANN –FRANZ LAW
Statement
It states that for the metals, the ratio
of thermal conductivity to electrical conductivity is directly proportional to
the absolute temperature. This ratio is a constant for all metals at given
temperature.
K
/ σ ∞
T
Or
Where
L is a proportionality constant. It is known as Lorentz number. Its value is
2.44 X 10-8
-2
WΩK
at T = 293 K.
Derivation
Widemann –Frantz law is derived from the
expressions of thermal conductivity and electrical conductivity of metal.
We know that,
Electrical conductivity
of a metal σ = ne2τ / m
----(1)
Thermal conductivity of
a metal K = ½ n v2k τ
----(2)
Thermal
conductivity = ½ n v2 k τ
Electrical conductivity = ne2 τ / m
K / σ = ½ mv2
k / e2 ---(3)
We know that the
kinetic energy of an electron
Or ½
mv2 = 3/2 kT
---(4)
Substituting equation 4
in equation 3, we have
K / σ = 3/2 kT X k /
e2
= 3/2 [k/e] 2
T
K / σ = LT ---(5)
where L = 3/2 [k/e] 2
is a constant and it is known as Lorentz number
K / σ = αT ----(6)
Thus it is proved that, the ratio of
thermal conductivity to electrical conductivity of a metal is directly
proportional to the absolute temperature of the metal.
Conclusion
Wiedamann –Franz law clearly shows that
if a metal has high thermal conductivity, it will also have high electrical
conductivity.
6
LORENTZ NUMBER
The ratio of thermal conductivity (K) of
of the metal and absolute temperature (T) of the metal is a constant. It is
known as Lorentz number
and
it is given by
L =
K / σ
7MERITS
OF CLASSICAL FREE ELECTRTON THEORY
1. It is
used to verify
Ohm’s Law.
2. The
electrical and Thermal conductivities of metals can be explained by this
theory.
3. It
is used to derive Wiedemann- Franz law.
4. It
is used to explain the optical properties of metals.
8 DRAWBACKS OF
CLASSICAL FREE ELECTRON THEORY
It is a macroscopic theory
Classical theory states that all free electrons will absorb energy, but
quantum theory states that only few electrons will absorb energy.
This theory cannot explain the Compton effect, photoelectric effect,
paramagnetism, ferromagnetism etc.,
The theoretical and experimental values of specific heat and electronic
specific heat are not matched.
By classical theory Kratures,/σT= butconstantbyquanfo constant for all
temperatures.
The Lorentz number by classical theory does not have good agreement with
experimental value and it is rectified by quantum theory.
9 QUANTUM THEORY
The drawbacks of
classical theory can be rectified using quantum theory. In classical theory the
properties of metals such as electrical and thermal conductivities are well
explained on the assumption that the electrons in the metal freely moves like
the particles of gas and hence called free electron gas.
According to classical
theory, the particles of gas (electrons) at zero Kelvin will have zero kinetic
energy, and hence all the particles are at rest. But according to quantum
theory when all particles at rest, all of them should be filled only in the
ground state energy level, which is impossible and is controversial to the
pauli’s exclusion principle.
Thus in order to fill
the electrons in a given energy level, we should know the following. Energy
distribution of electrons
Number of available
energy states Number of filled energy states
Probability of filling
an electron in a given energy state, etc.,
10 FERMI DIRAC DISTRIBUTION FUNCTION
The classical and
quantum free electron theories failed to explain many electrical and thermal
properties of solids. However, these properties can be easily understood using
Fermi –Dirac statistics.
Fermi –Dirac statistics
deals with the particles having half integral spin. The particles like
electrons are the examples of half integral spin and hence they are known as
Fermi particles or Fermions.
Definition
The expression which
gives the distributions of electrons among the various energy levels as a
function of temperature is known as Fermi distribution function. It is the
probability function F(E) of an electron occupying given energy level at
absolute temperature. It is given by
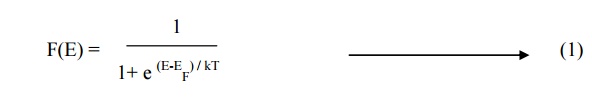
Where
E
–Energy of the level whose occupancy is being considered
EF
–Energy of the Fermi level
K
–Boltzmann’s constant
T
–Absolute temperature
The probability value
of F(E) lies between 0 and 1. If F(E) = 1, the energy level is occupied by an
electron. If F(E) = 0, the energy level is vacant. If F(E) = ½ or 0 .5 then
there is a 50% chance for the electron occupying in that energy level.
Effect of temperature on Fermi function
The
effect of temperature on Fermi function F(E) can be discussed with respect to equation
1.
1. At 0 kelvin
At 0 kelvin, the
electron can be filled only upto a maximum energy level called Fermi energy
level (EF0), above EF0 all the energy levels will be empty. It can be proved
from the following conditions.
When E<EF,
equation 1 becomes
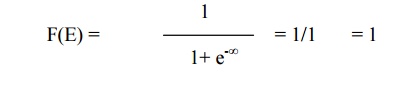
(i.e 100% chance for
the electron to be filled within the Fermi energy level)
When E>EF,
equation 1 becomes
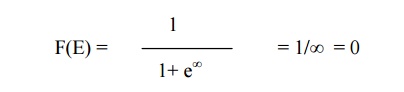
(i.e zero% chance for
the electron not to be filled within the Fermi energy level)
When E = EF,
equation 1 becomes
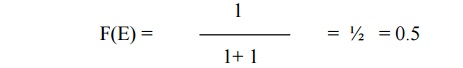
(i.e
50% chance for the electron to be filled within the Fermi energy level)
This clearly shows that
at 0 kelvin all the energy states below EF0 are filled and all those
above it are empty.
The Fermi function at 0
kelvin can also be represented graphically as shown in fig .
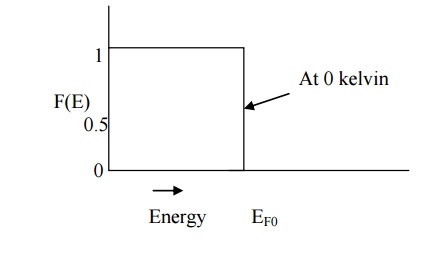
Fermi energy and its
importance Fermi energy level
Fermi energy level is
the maximum energy level upto which the electron can be filled at ok. Importance:
Thus it act as a
reference level which separates the vacant and filled energy states at 0k. It
gives the information about the filled electron states and the empty states.
At 0k, blow Fermi
energy level electrons are filled and above Fermi energy level it will be empty.
When the temperature is
increased, few electrons gains thermal energy and it goes to higher energy
levels.
Conclusions:
In the quantum free
electron theory, though the energy levels are descrete, the spacing between
consecutive energy level is very less and thus the distribution of energy
levels seems to be continuous.
The number of energy
levels N(E) that are filled with electrons per unit energy increases parabolic
ally with increase of energy E as shown in fig.
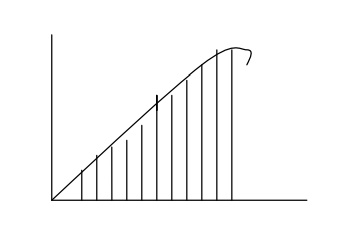
Each energy level can
provide only two states, namely, one for spin up and other for spin down and
hence only two electrons can be exclusion principle.
At T = 0, if there are
N number of atoms, then we have N/2 number of filled energy levels and other
higher energy levels will be completely empty.
This (N/2) th
energy level is the highest filled energy level is known as Fermi energy level
(EFO).
The electrons are
filled in a given energ no two electrons can have the same set of four quantum
numbers.
At room temperature,
the electrons within the range of KBT below the Fermi energy level
will absorb thermal energy = KBT and goes to higher energy states
with energy EF0 + KBT.
11 DENSITY OF STATES
The Fermi function F(E)
gives only the probability of filling up of electrons on a given energy state,
it does not give the information about the number of electrons that can be
filled in a given energy state. To know that we should know the number of
available energy states so called density of states.
11.1.Definition
Density of states
Z(E)dE is defined as the number of available electron states per unit volume in
an energy interval (dE).
Explanation
In order to fill the
electrons in an energy state we have to first find the number of available
energy states within a given energy interval.
We know that a number
of available energy levels can be obtained for various combinations of quantum
numbers nx, ny, and nz . (i.e) n2 =
nx2 + ny2 + nz2
Therefore, let us
construct three dimensional space of points which represents the quantum
numbers nx, ny, and nz as shown in fig. in
this space each point represents an energy level.
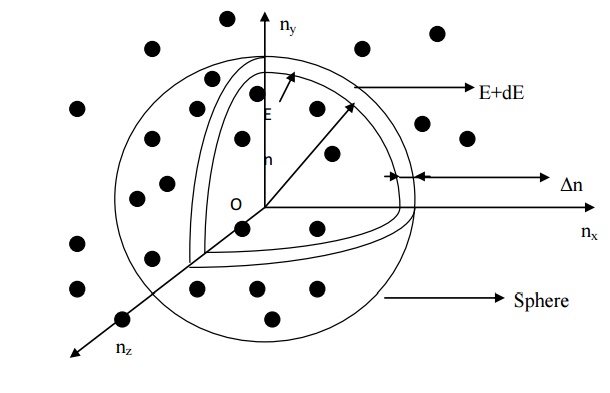
11.2.DERIVATION OF DENSITY OF ENERGY
STATES
To find the number of
energy levels in a cubical metal piece and to find number of electrons that can
be filled in a given energy level, l
The sphere is further
divided in to many shells and each of this shell represents a particular
combination of quantum numbers (nx, n y, and nz)
and therefore represents a particular energy value.
Let us consider two
energy values E and E + dE. The number of energy states between E and E + dE
can be found by finding the number of energy states between the shells of the
radius n and n + Δn, from the origin.
The number of energy states within the sphere of
radius n = 4/3 πn3
Since nx, ny, and nz
will have only positive values, we have to take only one octant of the sphere
(i.e) 1/8 th of the sphere volume.
The number of available
energy states within the sphere of radius n = 1/8 [4/3 π n3]
Similarly the number of
available energy states within the sphere of radius n + dn
n + dn = 1/8 [4/3 π
(n+dn)3 ]
The number of available
energy states between the shells of radius n and n + dn (or) between the energy
levels

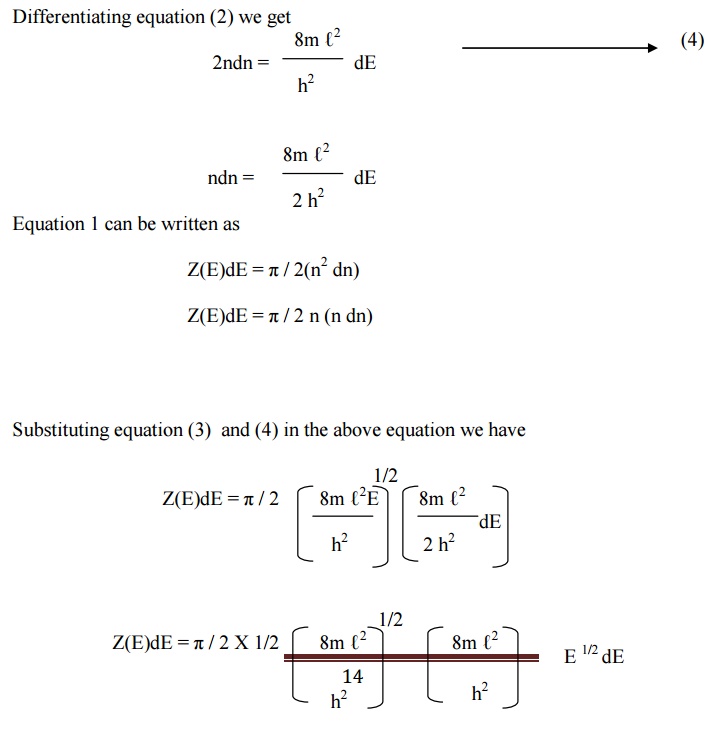
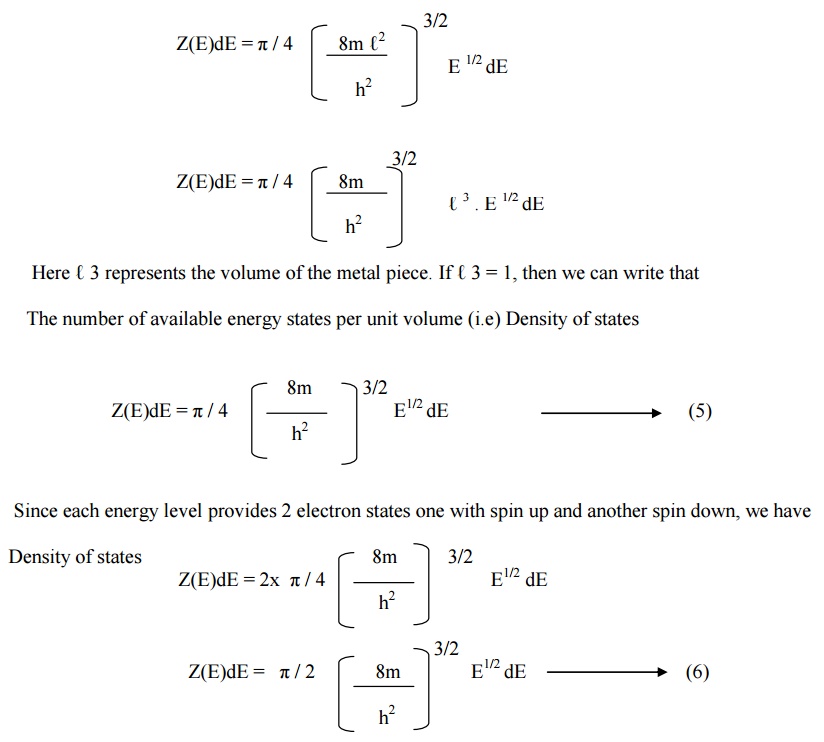
11.3.
CALCULATION OF CARRIER CONCENTRATION
Let N(E) dE represents
the number of filled energy states between the interval of energy dE . Normally
all the energy states will not be filled. The probability of filling of
electrons in a given energy state is given by Fermi function F(E).
N(E) = Z(E)dE .
F(E) ----(7)
Substituting equation
(6) in equation (7), we get
Number of filled energy
states per unit volume
N(E) = π / 2 [8m / h2 ] E ½ dE.
F(E) ----(8)
N(E) is known as
carrier distribution function (or) carrier concentration in metals.
11.4.CALCULATION OF FERMI ENERGY
Case (i) At T= 0K
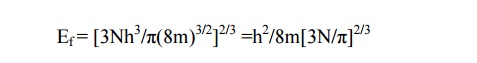
Case(ii) At T> 0K
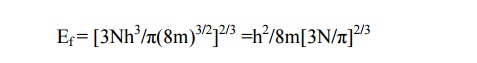
11.5.AVERAGE ENERGY OF
AN ELECTRON AT OK
Average
energy = Total energy /
Carrier Concentration
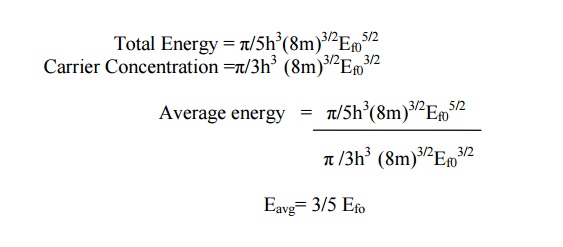
Eavg=
3/5 Efo
Related Topics