Chapter: Basic & Clinical Pharmacology : Tetracyclines, Macrolides,Clindamycin,Chloramphenicol,Streptogramins,& Oxazolidinones
Clindamycin
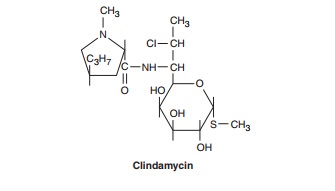
CLINDAMYCIN
Clindamycin
is a chlorine-substituted derivative of lincomycin,
an antibiotic that is elaborated by Streptomyces
lincolnensis.
Mechanism of Action &
Antibacterial Activity
Clindamycin, like
erythromycin, inhibits protein synthesis by inter-fering with the formation of
initiation complexes and with amino-acyl translocation reactions. The binding
site for clindamycin on the 50S subunit of the bacterial ribosome is identical
with that for erythromycin. Streptococci, staphylococci, and pneumococci are
inhibited by clindamycin, 0.5–5 mcg/mL. Enterococci and gram-negative aerobic organisms
are resistant. Bacteroides sp and
other anaerobes, both gram-positive and gram-negative, are usually
sus-ceptible. Resistance to clindamycin, which generally confers
cross-resistance to macrolides, is due to (1) mutation of the ribosomal
receptor site; (2) modification of the receptor by a constitutively expressed
methylase (see section on erythromycin resistance, above); and (3) enzymatic
inactivation of clindamycin. Gram-negative aero-bic species are intrinsically
resistant because of poor permeability of the outer membrane.
Pharmacokinetics
Oral
dosages of clindamycin, 0.15–0.3 g every 8 hours (10–20 mg/ kg/d for children),
yield serum levels of 2–3 mcg/mL. When administered intravenously, 600 mg of
clindamycin every 8 hours gives levels of 5–15 mcg/mL. The drug is about 90%
protein-bound. Clindamycin penetrates well into most tissues, with brain and
cerebrospinal fluid being important exceptions. It penetrates well into
abscesses and is actively taken up and concentrated by phagocytic cells. Clindamycin
is metabolized by the liver, and both active drug and active metabolites are
excreted in bile and urine. The half-life is about 2.5 hours in normal
individuals, increasing to 6 hours in patients with anuria. No dosage
adjustment is required for renal failure.
Clinical Uses
Clindamycin is
indicated for the treatment of skin and soft-tissue infections caused by
streptococci and staphylococci. It is often active against community-acquired
strains of methicillin-resistant S aureus,
an increasingly common cause of skin and soft tissueinfections. Clindamycin is
also indicated for treatment of anaerobic infections caused by Bacteroides sp and other anaerobes that
often participate in mixed infections. Clindamycin, sometimes in com-bination
with an aminoglycoside or cephalosporin, is used to treat penetrating wounds of
the abdomen and the gut; infections origi-nating in the female genital tract,
eg, septic abortion, pelvic abscesses, or pelvic inflammatory disease; and lung
abscesses. Clindamycin is now recommended rather than erythromycin for
prophylaxis of endocarditis in patients with valvular heart disease who
are undergoing certain dental procedures and have significant penicillin
allergies. Clindamycin plus primaquine is an effective alternative to trimethoprim-sulfamethoxazole
for moderate to moderately severe Pneumocystis
jiroveci pneumonia in AIDS patients. It is also used in combination with
pyrimethamine for AIDS-related toxoplasmosis of the brain.
Adverse Effects
Common
adverse effects are diarrhea, nausea, and skin rashes. Impaired liver function
(with or without jaundice) and neutrope-nia sometimes occur. Administration of
clindamycin is a risk fac-tor for diarrhea and colitis due to C difficile.
Related Topics