Chapter: Medical Microbiology: An Introduction to Infectious Diseases: Chlamydia
Chlamydiatrachomatis : Clinical Aspects
Chlamydiatrachomatis : CLINICAL ASPECTS
MANIFESTATIONS
Eye Infections
Trachoma and inclusion conjunctivitis are distinct diseases of the eye that have some overlap in their clinical manifestations. Trachoma, a chronic conjunctivitis caused byC. trachomatis strains A, B, Ba, and C, is usually seen in less developed countries andoften leads to blindness. Inclusion conjunctivitis, an acute infection commonly caused by strains D to K, is usually not associated with chronicity or permanent eye damage. It occurs in newborns and adults worldwide.
Trachoma
Chronic inflammation of the eyelids and increased vascularization of the corneal conjunc-tiva are followed by severe corneal scarring and conjunctival deformities. Visual loss of-ten occurs 15 to 20 years after the initial infection, due to repeated scarring of the cornea.
Inclusion Conjunctivitis
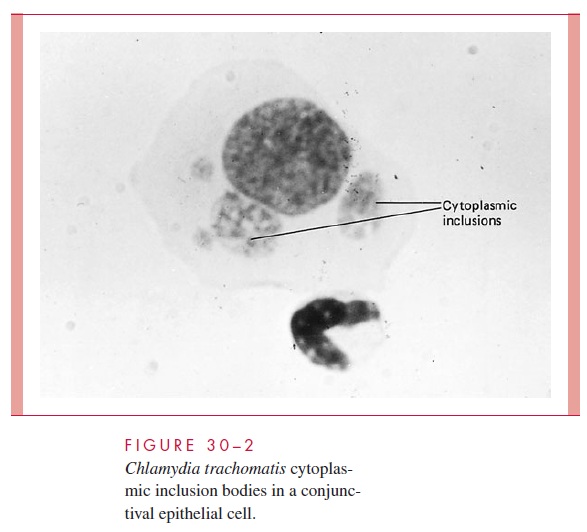
Inclusion conjunctivitis usually presents as an acute, copious, mucopurulent eye dis-charge 5 to 25 days after birth. Infection occurs in roughly two thirds of infants born vaginally to infected mothers, and one third of these become overtly ill. Inclusion con-junctivitis is clinically similar but less common in adults, and is usually associated with concomitant genital tract disease. Diagnosis can be made by demonstrating characteristic cytoplasmic inclusions in smears of conjunctival scrapings (Fig 30 – 2), by demonstration of antigen by direct immunofluorescence, or by culture from conjunctival swabs. Sys-temic therapy is preferred because the nasopharynx, rectum, and vagina may also be colo-nized and other forms of disease may develop, such as an infant pneumonia syndrome.
Genital Infections
The clinical spectrum of sexually transmitted infections with C. trachomatis is similar to that of Neisseria gonorrhoeae. C. trachomatis can cause urethritis and epididymitis in men and cervicitis, salpingitis, and a urethral syndrome in women. In addition, three strains of C. trachomatis cause LGV, another sexually transmitted disease (see Table 30 – 2).

C. trachomatis urethritis is manifested by dysuria and a thin urethral discharge. Infec-tions of the uterine cervix may produce vaginal discharge but are usually asymptomatic. Ascending infection in the form of salpingitis and pelvic inflammatory disease occurs in an estimated 5 to 30% of infected women. The scarring produced by chronic or repeated infection is an important cause of sterility and ectopic pregnancy.
More than 50% of all infants born to mothers excreting C. trachomatis during labor show evidence of infection during the first year of life. Most develop inclusion conjunc-tivitis (see earlier discussion), but 5 to 10% develop an infant pneumonia syndrome. C. trachomatis accounts for about one third to one half of all cases of interstitial pneumo-nia in infants. The illness usually develops between 6 weeks and 6 months of age and has a gradual onset. The child is usually afebrile, but develops difficulty in feeding, a charac-teristic staccato (pertussis-like) cough, and shortness of breath. The disease is rarely fatal but may be associated with decreased pulmonary function later in life.
LGV is a sexually transmitted infection caused by C. trachomatis strains L1, L2, or L3. It occurs principally in South America and Africa, although small outbreaks have recently occurred in North America. The clinical course is characterized by transient genital lesions followed by multilocular suppurative involvement of the inguinal lymph nodes. The primary genital lesion is usually a small painless ulcer or papule, which heals in a few days and may go unnoticed. The most common presenting complaint is inguinal adenopathy. Nodes are initially discrete, but as the disease progresses they become matted and suppurative (bubos). The skin over the node may be thinned, and multiple draining fistulas develop. Systemic symptoms such as fever, chills, headaches, arthralgia, and myalgia are common. Late complications include urethral or rectal strictures and perirec-tal abscesses and fistulas. In homosexual men, LGV strains can cause a hemorrhagic ulcerative proctitis. Lymph nodes may need to be aspirated to prevent rupture.
DIAGNOSIS
All direct C. trachomatis diagnostic tests require the collection of epithelial cells from the site of infection. Inflammatory cells are not useful and should be cleaned away as much as possible. For genital infections, cervical specimens are preferred in females and ure- thral scrapings in males. Eye infections require conjunctival scrapings.
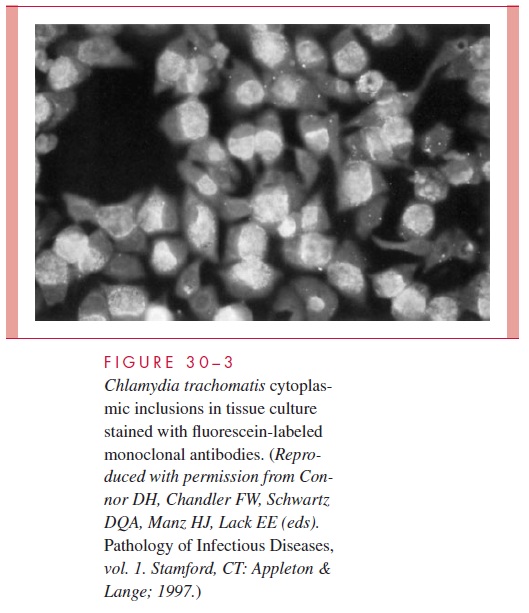
Isolation of C. trachomatis has been the “gold standard” for diagnosis. It is achieved in cell culture using idoxuridine- or cycloheximide-treated McCoy cells. Treatment of the cells with antimetabolites inhibits host cell replication but allows chlamydiae to use available cell nutrients for growth. After inoculation with samples and incubation for 3 to 7 days, the cells are stained with fluorescein-labeled monoclonal antibodies to detect intracytoplasmic chlamydiae (Fig 30 – 3).
C. trachomatis reticulate bodies synthesize large amounts of glyco-gen, and the inclusion bodies in the cell thus stain reddish-brown with iodine. C. psittaci and C. pneumoniae inclusions do not contain glycogen.
A large number of procedures are now available for noncultural direct detection of C. trachomatis in clinical specimens. These include direct fluorescent antibody (DFA)methods using monoclonal antibodies directed against outer membrane proteins of ele-mentary bodies in epithelial cells, enzyme immunoassays that detect chlamydial lipopolysaccharide, and assays for C. trachomatis DNA. These tests are faster, and, in the case of DNA assays, also more sensitive than culture. Ligase chain reaction (LCR) or polymerase chain reaction (PCR) are the most sensitive methods of diagnosis. The latter procedures can be used on urine samples, which are easier to obtain than cervical or urethral samples. This greatly aids the detection of chlamydial genital infections, es-pecially in adolescents, and ultimately facilitates control of the spread of this sexually transmitted disease.
Serodiagnostic methods have little use in diagnosis of chlamydial genital infection be-cause of the difficulty of distinguishing current from previous infection. Detection of IgM antibodies against C. trachomatis is helpful in cases of infant pneumonitis. Chlamydial serology is also useful in the diagnosis of LGV, where a single high complement fixation antibody titer ( >1 32) or a fourfold rise supports a presumptive diagnosis. The most sat-isfactory method for diagnosis of LGV is isolation of an LGV strain of C. trachomatis from aspirated bubos or tissue biopsies. In 80 to 90% of patients, the LGV complement fixation test is positive (titer> 1 64) shortly after the appearance of the bubo.
TREATMENT
Strains of C. trachomatis are sensitive to tetracyclines, macrolides and related com-pounds, and some fluoroquinolones. Azithromycin is given as a single oral dose for non-LGV C. trachomatis infection. Erythromycin is used for pregnant women and infants because of the tooth staining that may result from tetracycline therapy and less experience with the newer agents. Doxycycline is an alternative for C. trachomatis and is the drug of choice for treating LGV.
For trachoma, a single dose of azithromycin is now the treatment of choice, al-though a tetracycline for 14 days is an alternative. Corrective surgery may prevent blindness and is required for severe corneal and conjunctival scarring. Control of tra-choma is directed toward prevention of continued reinfection during early childhood. Improvement in general hygienic practices is the most important factor in decreasing transmission of infection within families and, of course, one of the most difficult to im-plement on a broad scale.
PREVENTION
Prophylaxis for infants using topical erythromycin or silver nitrate on the conjunctiva has limited effectiveness for Chlamydia, because 15 to 25% of exposed infants still develop inclusion conjunctivitis. The primary approach to prevention of all forms of genital and infant C. trachomatis infection comprises detection of this infection in sexually active in-dividuals and appropriate treatment, including infected women late in pregnancy. No vac-cine is available or under development.
Related Topics