Chapter: Introduction to Human Nutrition: Food Safety: A Public Health Issue of Growing Importance
Chemicals affecting food safety
Chemicals affecting food safety
Chemicals may be present in food owing to their natural occurrence in soil (e.g., cadmium, lead) or from fungal contamination (e.g., aflatoxins, ochra-toxin), from algal contamination [e.g., amnesic shell-fish poisoning (ASP), diarrhetic shellfish poisoning (DSP), azaspiracid shellfish poisoning (AZP), para-lytic shellfish poisoning (PSP)], from industrial or other pollution [e.g., lead, mercury, polychlorinated biphenyls (PCBs), dioxins], from agricultural and vet-erinary practices (e.g., pesticides, fertilizers, veteri-nary drugs) or from food processing and packaging techniques [e.g. acrylamide, polycyclic aromatic hydrocarbons (PAHs), 3-monochloropropane-1,2-diol (3-MCPD), bisphenol A diglycidyl ether (BADGE)].
Toxicological assessment of these substances is largely carried out on an international basis by expert groups such as the Joint Expert Committee on Food Additives and Contaminants (JECFA) or Joint Meeting on Pesticide Residues (JMPR), both jointly organized by the WHO and FAO. These expert groups advise on acceptable or tolerable levels of intake of these substances.
Acceptable and tolerable levels of intake
The acceptable daily intake (ADI) level of a chemical is the daily intake that, during a lifetime, would pose no appreciable risk to the consumer, on the basis of all facts known at the time. It is expressed in mg/kg of body weight.
The tolerable weekly intake (TWI) represents per-missible human weekly exposure to those contami-nants unavoidably associated with the consumption of otherwise wholesome and nutritious foods. The term tolerable signifies permissibility rather than acceptability for the intake of contaminants that have no necessary function in food, in contrast to those of permitted pesticides or food additives. For cumulative toxicants, such as lead, cadmium, and mercury, the tolerable intakes are expressed on a weekly basis to allow for daily variations in intake levels, the real concern being long-term exposure to the contaminant.
The maximum tolerable daily intake (MTDI) has been established for food contaminants that are not known to accumulate in the body, such as tin, arsenic, and styrene. The value assigned to the provisiona MTDI represents permissible human exposure as a result of the natural occurrence of the substance in food and drinking water.
One of the most difficult issues in food safety is to advise on the potential risks to human health for substances found in food which are both genotoxic (damaging DNA, the genetic material of cells) and carcinogenic (leading to cancer). For these substances, it is generally assumed that even a small dose can have an effect. JECFA addressed this issue in 1978 and introduced the concept of an “irreducible level,” which it defined as “that concentration of a substance which cannot be eliminated from a food without involving the discarding of that food altogether, severely com-promising the ultimate availability of major food sup-plies” (FAO/WHO, 1978).
Until now the risk assessors have advised to keep the exposure to such substances at the lowest possible level. This approach is known as the ALARA principle (“as low as reasonably achievable”). A disadvantage of this approach is that it cannot be used to compare risks posed by different substances. Furthermore, the application of the ALARA principle does not take into account the effectiveness of a substance and the actual (sometimes extremely low) level of occurrence in food.
A different approach, “the margin of exposure” (MoE) approach, which can be used to assess the risks to human health of exposure to a substance in the absence of a tolerable daily intake or similar guidance value, has recently been endorsed by the EFSA Scien-tific Committee (EFSA, 2005) and the WHO/FAO
Setting the acceptable daily intake
JECFA generally sets the ADI of a substance on the basis of the highest no-observed-effect level in animal studies. In calculating the ADI, a “safety factor” is applied to the no-observed-effect level to provide a conservative margin of safety on account of the inher- ent uncertainties in extrapolating animal toxicity data to potential effects in humans and for variation within the human species. JECFA traditionally uses a safety factor of 100 (10 × 10) in setting ADI values based on long-term animal studies. It is intended to provide an adequate margin of safety for the consumer by assum-ing that the human being is 10 times more sensitive than the test animal and that the difference in sensi-tivity within the human population is in a 10-fold range. However, different safety factors apply depend-ing on the substance and test species in question.
Maximum levels for food commodities
These levels are calculated taking the above-mentioned levels into consideration. Depending on the substance, different principles apply. Residues such as pesticides and residues of veterinary drugs in foodstuffs are limited by setting a maximum residue limit (MRL). Additives are regulated by setting maximum limits or by applying the “quantum satis” principle (the least amount required to exert the desired technological function).
For contaminants, maximum levels/limits are established for those foods that provide a significant contribution to the total dietary exposure. However, as a general principle the levels in all foods should always be kept as low as reasonably achievable (the ALARA principle).
In Europe, additives, pesticides, veterinary residues, and a wide range of contaminants are regulated by EU legislation in the form of directives or regulations that are transposed into national legislation by each member state.
Pesticide residues
Pesticides are chemicals or biological products used to control harmful or undesired organisms and plants, or to regulate the growth of plants as crop protection agents.
Pesticides can also be toxic to humans since certain biochemical pathways are relatively conserved across species, as are some enzymes and hormones. In the context of food safety, exposure to pesticides is clas-sified as acute or chronic. An acute intoxication usually has an immediate effect on the body, whereas a chronic effect may reveal itself over the lifespan. The severity depends on the dose and the toxicity of the pesticide compound or breakdown product. Toxic effects that have been identified include enzyme inhi-bition, endocrine disruption, and carcinogenic action, depending on the compound in question.
In Europe the control of pesticides is based on Council Directive 91/414/EEC. Under this legislation, pesticides must be evaluated for safety based on dos-siers prepared by their manufacturers. If a pesticide is accepted it is placed on a positive list with an MRL assigned to it.
This is the maximum intake level, which is judged to result in no adverse toxicological effect following such exposure. The ARfD value includes a safety factor to ensure that older people, infants and children, and those under stress due to illness are protected.
Veterinary drug residues
Veterinary drugs include antibacterial compounds, hormones, and nonsteroidal anti-inflammatory prep-arations. As animal husbandry practices have intensi-fied over the past few decades, antibacterial substances have been increasingly used as growth promoters to increase feed conversion efficiency, and for pro-phylaxis and therapy to prevent outbreaks and treat disease. Similarly, hormones are administered to increase growth rate and meat yield. Table 14.6 shows the main types of antibacterial and hormonal compounds.
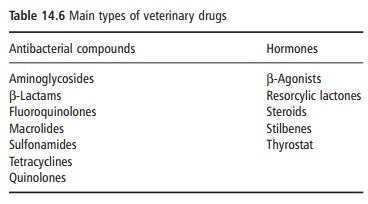
Veterinary drugs are metabolized in the animal and are excreted in the urine and feces over time as the detoxification process continues. Hence, residue traces of drugs or their metabolites can be found in major organs, muscles, and body fluids. In general, antibacterial drugs are found in greatest concentration in the kidney, lesser concentrations in the liver and lowest concentrations in the muscle tissue, whereas hormones tend to concentrate in the liver.
The excessive use of antibacterial compounds in animal husbandry has raised concerns about the development of resistant bacteria and the effect that this may have on the usefulness of antibiotics in human medicine. There have also been concerns about the risk of allergic reactions in humans to anti-bacterial residues in food of animal origin. The use of hormones has raised issues surrounding the effects of hormone residues in foods of animal origin on human metabolism.
Environmental and industrial contaminants
These contaminants are of environmental origin or are by-products of industrial processes.
Polyhalogenated hydrocarbons (PHHs) are a cate-gory of environmental contaminants that includes toxaphene, dioxins, and polychlorinated biphenyls (PCBs). Certain polyhalogenated hydrocarbons are manufactured for use in plastics, paints, transformers, and herbicides; although their use is now either banned or severely restricted. In most industrialized nations the compounds have become ubiquitous in the environment. Hence, contamination of the food chain is inevitable and it has been estimated that in Western industrialized countries 90% of human exposure is through ingestion of contaminated foods such as fish and milk.
Foods that are rich sources of fats and oils tend to accumulate PHHs because the compounds are lipo-philic and bioaccumulate in lipid-rich tissues and fluids. Oily fish from areas such as the Baltic Sea, where levels of PHHs in the water are high, may contain elevated levels of these contaminants. Simi-larly, cows that graze on polluted pasture can accu-mulate unacceptable concentrations of PHHs in their milk. A recent incident in Belgium introduced PCBs and dioxins into the food chain via contaminated animal feed resulting from the accidental incorpora-tion of industrial oil into the feed ration. The biologi-cal half-life of PHHs can range from a matter of months to 20 years in human adipose tissue. Hence, they are persistent and accumulate in the body. Expo-sure to PHHs can result in a variety of toxic effects that can be carcinogenic, including dermal toxicity, immunotoxicity, reproductive effects, and endocrine disruption.
Metals, metalloids, and their compounds have long been associated with food poisoning, with lead and mercury probably the best documented hazards. Metals are released into the environment as a result of natural geological action and also as a result of man-made pollution from industrial processes.
Metals have an affinity for biological tissue and organic compounds, and hence they are often easily absorbed into the body and can often accumulate in organs and fat deposits. Table 14.7 shows some of the main metals linked with food-borne toxicity.
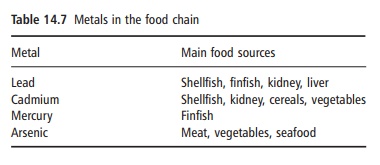
Lead toxicity has many symptoms, but the main issue relates to its effects on the nervous system of children. Here, lead interferes with the transmission of nervous signals around the body. This can manifest itself in a reduced intelligence quotient (IQ) and coordination problems. In adults, exposure to lead can result in hypertension and other blood effects such as anemia. Cadmium is most often accumulated from occupational exposure or smoking and is known to affect the respiratory system. However, food expo-sure tends to be at a low level over longer periods. In this regard, cadmium bioaccumulates in the kidney and can cause renal damage. Mercury and its com-pounds also bioaccumulate in the body, where they are most frequently associated with neural effects and renal damage. In particular, methylmercury is highly toxic particularly to the nervous system, and the developing brain is thought to be the most sensitive target organ for methylmercury toxicity.
Arsenic is most often an occupational hazard, but it can also be ingested with food and is responsible for acute and chronic poisoning. The toxicity of arsenic depends on its oxidation state and the type of complex that it forms with organic molecules in the body. Chronic effects include gastroenteritis, nephri-tis, and liver damage. Arsenic is also considered to be a carcinogen. Other metals are also known contami-nants and their toxic effects are diverse. Although this is not an exhaustive list, these metals include alumi-num, copper, tin, zinc, and chromium.
Process contaminants
These types of contaminant occur during the process-ing and production of foods, and include acrylamide, PAHs, chloropropanols, and nitrosamines.
Acrylamide is a reactive unsaturated amide that has found several industrial uses. In 2002, it was discov-ered to occur in a variety of fried and baked foods, in particular carbohydrate-rich foods that had been subjected to high-temperature cooking/processing.
Acrylamide has been shown to be neurotoxic in humans. It has been shown to induce tumors in labo-ratory rats and has been classified as a probable human carcinogen, and as such several international bodies have concluded that dietary exposure should be as low as reasonably achievable. The most signifi-cant pathway of formation of acrylamide in foods has been shown to arise from the reaction of reducing sugars with asparagines via the Maillard reaction at temperatures above c. 120°C. Acrylamide has been found in a wide range of heat-treated foods; it is found in both foods processed by manufacturers and foods that are cooked in the home. Acrylamide has been found to be most prevalent in fried potato prod-ucts (such as French fries and potato chips), cereals, bakery wares, and coffee.
PAHs are a group of over 100 different chemicals that are formed during certain technological pro-cesses and are common environmental contaminants. They are formed during incomplete combustion of coal and oil. They are also formed during barbecuing or grilling meat. Human exposure usually results from air pollution and from cigarette smoke. Foods most likely to be contaminated by PAHs are grilled or charred meats. PAHs are toxins that have been docu-mented by the WHO as genotoxic, immunotoxic, and carcinogenic. Long-term exposure to foods contain-ing PAHs can lead to serious health risks. In a recent Europe-wide incident, PAHs were found in pomace olive oil, which resulted in a major product recall.
3-Monochloro-propane-1,2-diol (3-MCPD) is a member of a group of contaminants known as chloropropanols, which includes known genotoxic animal carcinogens such as 1,3-dichloropropan-2-ol. 3-MCPD is a by-product in soy sauce and in hydro-lyzed vegetable protein produced through acid hydro-lysis. It can also be present as a contaminant in some food additives, and in epichlorhydrin/amine copoly-mers, used as flocculants or coagulant aids in water treatment, and may be present in drinking water (opinion of the European Scientific Committee on Food, adopted on 30 May 2001).
Microbial toxins
Food poisoning can occur as a result of the ingestion of food containing preformed toxins that originate from bacterial growth, fungal growth, or algal growth.
In the case of bacteria the toxin is absorbed into the bloodstream via the intestine and therefore illness results from intoxication rather than infection. In the case of fungi, several species are involved in the pro-duction of toxic substances during growth on food-stuffs. These toxins are known as mycotoxins. Algal toxins are usually associated with seafood, most notably molluscan shellfish.
Bacterial toxins
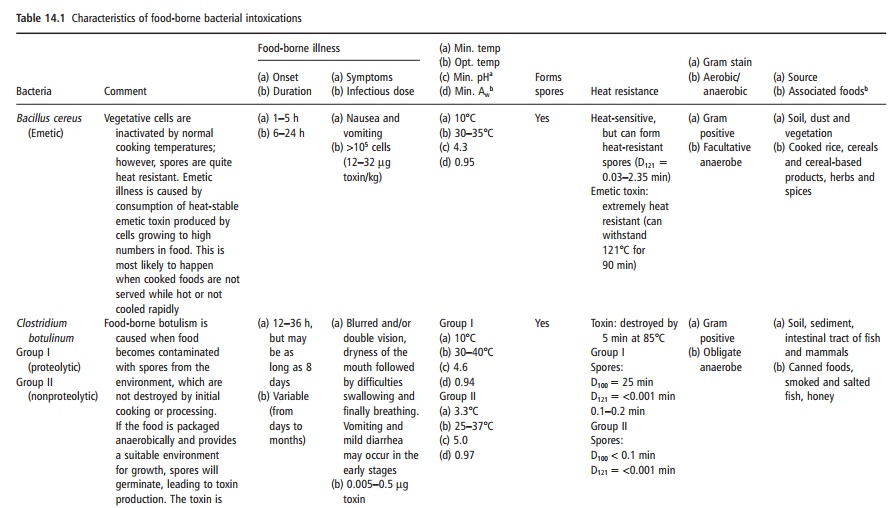
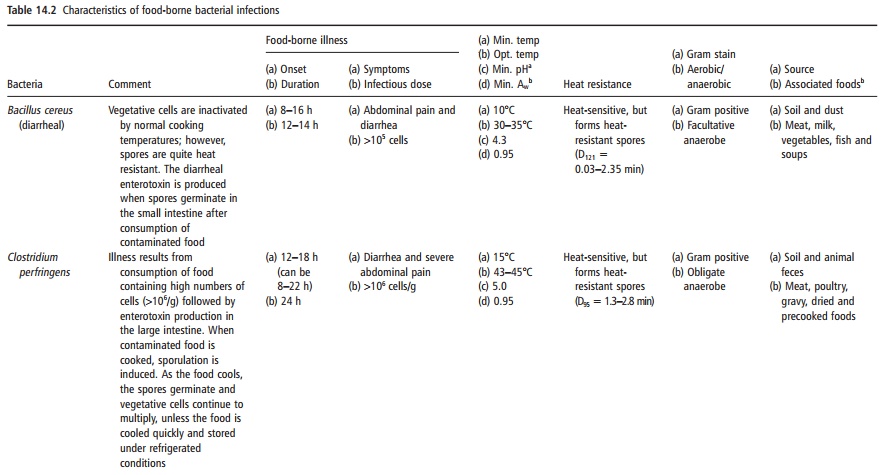
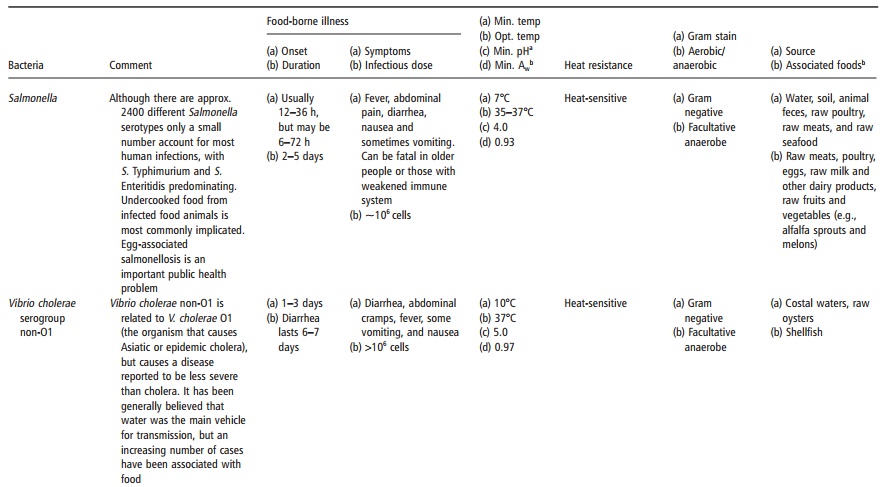
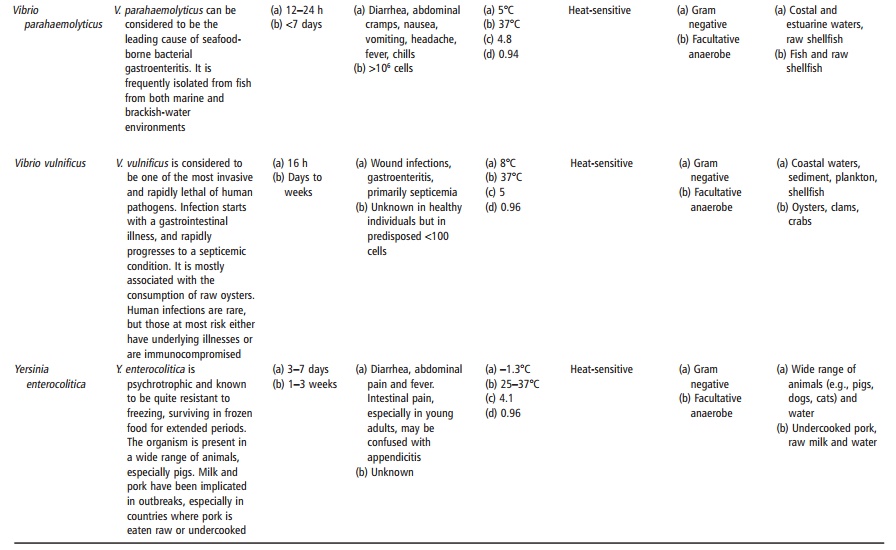
Three bacteria are most commonly associated with preformed toxin production: Clostridium botu-linum, Staphylococcus aureus and Bacillus cereus (see Table 14.1).



Fungal toxins (mycotoxins)
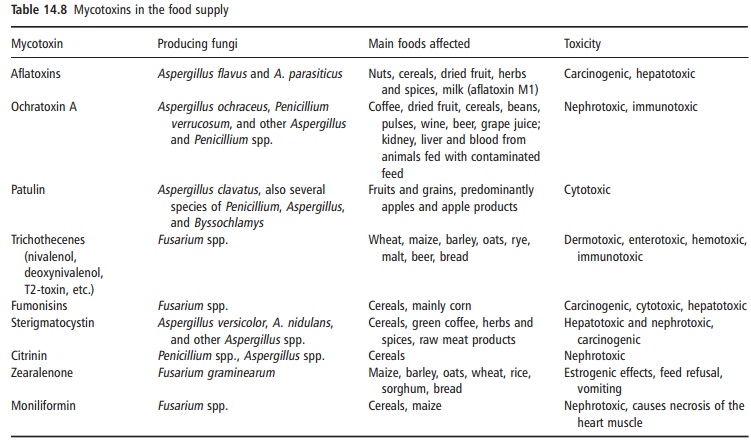
Mycotoxins are secondary metabolites of molds that can induce acute and chronic symptoms, such as carcinogenic, mutagenic, and estrogenic effects in humans and animals. Acute toxicity due to mycotox-ins is associated with liver and kidney damage. Chronic toxicity resulting from the exposure of low levels of mycotoxins in the human diet is a major food safety concern. In nonindustrialized countries myco-toxins have been reported to be responsible for increased morbidity and mortality in children owing to suppression of their immune systems and greater susceptibility to disease.
The principal fungi that are associated with myco-toxin production are the genera Aspergillus, Penicil-lium, and Fusarium. Aspergillus and Penicillium are sometimes referred to as storage fungi as they can grow at low water activity levels and are associated with the post-harvest spoilage of stored food commodities such as cereals, nuts, and spices. Fusarium species are plant pathogens and can infect plants in the field and produce mycotoxins preharvest. Table 14.8 provides an over-view of the most important mycotoxins.
Seafood toxins
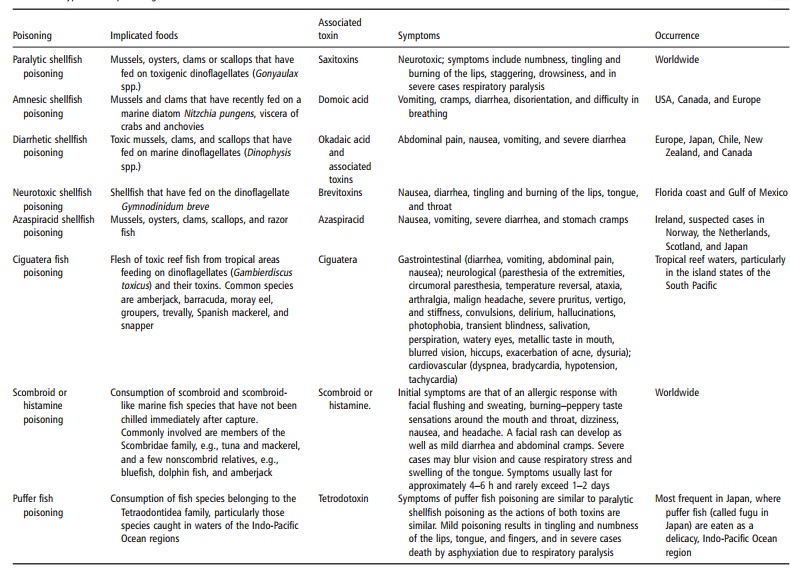
Fish and fishery products are nutritious foods and are desirable components of a healthy diet. Food-borne illnesses resulting from the consumption of seafood are associated with both finfish and molluscan shell-fish. The major risk of acute illness is associated with the consumption of raw shellfish, particularly bivalve molluscs. The consumption of these toxic shellfish by humans can cause illness, with symptoms ranging from mild diarrhea and vomiting to memory loss, paralysis, and death. Toxins associated with phyto
Various seafood poisoning syndromes are associ-ated with toxic marine algae and these include para-lytic shellfish poisoning (PSP), amnesic shellfish poisoning (ASP), diarrhetic shellfish poisoning (DSP), neurotoxic shellfish poisoning, and azaspiracid shell-fish poisoning (AZP). There are also different types of food poisoning associated with finfish and these include ciguatera poisoning, scombroid or histamine poisoning, and puffer fish poisoning. Consumption of raw molluscan shellfish poses well-known risks of food poisoning, but intoxication from finfish is not so well known. Most of the algal toxins associated with seafood poisoning are heat stable and are not inactivated by cooking. It is also not possible visually to distinguish toxic from nontoxic fish. Many coun-tries rely on biotoxin monitoring programs to protect public health and close harvesting areas when toxin algal blooms or toxic shellfish are detected. In nonin-dustrialized countries, particularly in rural areas, monitoring for harmful algal blooms does not rou-tinely occur and deaths due to red-tide toxins com-monly occur. Table 14.9 provides an overview of the most important types of fish poisoning.
Naturally occurring plant toxins
Certain plants contain naturally occurring com-pounds that are toxic to humans or that reduce the bioavailability of nutrients in foods. Examples of naturally occurring toxins are listed in Table 14.10. Some species of mushroom also contain toxic com-pounds, for instance agaritine. Some cereal-based diets have restricted bioavailability of nutrients as a result of the presence of antinutritional factors such as phytate and tannins or polyphenols.
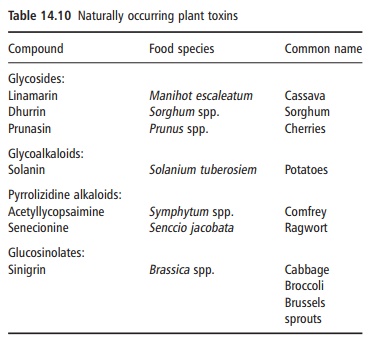
Food processing methods have evolved that reduce human exposure to both natural toxins and antinu-tritional compounds. For instance, cassava is a staple food of over 500 million people worldwide. Certain bitter varieties of cassava contain high levels of lin-amarin, a cyanogenic glycoside. The consumption of these varieties has been associated with health defects such as goiter and paralysis of the legs. Traditional processing of cassava in Africa that involves grating, soaking roots in water, and lactic acid fermentation completely removes the cyanide.
Adequate cooking of legume seeds such as kidney beans and disposal of the cooking water will remove the natural toxins present in these food products.
Antinutritional factors are those components of plants that interfere with metabolic processes and can lead to deficiencies of key nutrients in the diet. These are generally classified as enzyme inhibitors and mineral binding agents. Enzyme inhibitors are poly-peptides and proteins that inhibit the activities of digestive enzymes, and most are thermolabile and are reduced by cooking. For example, trypsin inhibitors may cause poor protein digestion and a shortage of sulfur-containing amino acids in the diet. Lectins are proteins that occur in beans that alter the absorption of nutrients in the intestinal wall. Cooking beans will inactivate lectins. Tannins (polyphenols) occur in cereals, specifically in the seed coat. These form complexes with proteins and inhibition of digestive enzymes. Phytate is a natural component in plants and on digestion forms insoluble complexes with metal ions in the body. The result is reduced bioavail-ability of essential minerals such as iron. In addition, a range of natural plant toxins can cause allergic reac-tions in humans, but there is a general lack of knowl-edge about their properties and modes of action.
Food additives
Food additives are added to foods for a specific tech-nological purpose during manufacture or storage and become an integral part of that foodstuff. Additives can be natural or synthetic and are usually catego-rized by their function (Table 14.11). For example, preservatives prevent the growth of bacteria, gelling agents maintain the structure of some foods during storage, and emulsifiers maintain the stability of fat structures. Without additives it would not be possible to manufacture many of the foods available today, especially convenience foods and low-fat foods.
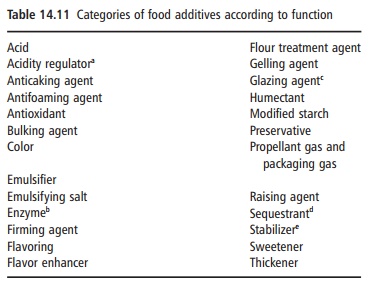
At the international level, additives are controlled by means of safety evaluation and the development of a positive list. To ensure transparency and choice, all additives that are used in prepackaged food should be labeled with their function and their name or
Related Topics