Chapter: Essential Anesthesia From Science to Practice : Applied physiology and pharmacology : Anesthesia and the cardiovascular system
Cardiovascular problems during anesthesia
Cardiovascular problems during
anesthesia
Hypotension
Picture the acutely hypotensive, tachycardic patient (BP 80/50 HR 120 bpm), a fairly common observation. How should you go about treating this patient? After the ABCs,5 we recommend a physiologic approach, rather than a mnemonic laun-dry list of possible causes. First, there are three main ways a patient can become hypotensive: low preload (not enough blood to push forward through the system), low contractility (inadequate force pushing the blood), and low resistance (dilated vascular bed). Other categories are less common and include severe bradycardia, lack of atrial kick, and valvular anomalies, to name a few. To distinguish between these, we start with situational awareness. Did the cross-clamp just come off the aorta? Did we just induce a sympathectomy with a high spinal anesthetic?
Add to that a
quick physical examination to rule out abnormal rhythm or valvular or cardiac
dysfunction and review of the patient’s medical history (chronic CHF or recent
myocardial infarction (MI)?). If these do not lead to a high-probability
diagnosis, invasive monitoring may be indicated.
The
invasive monitors we have available, in addition to the arterial catheter for
blood pressure monitoring, include:
·
filling pressure as an inference of ventricular volume/preload:
central venous pressure (CVP), pulmonary capillary wedge pressure (PCWP);
·
cardiac output: thermodilution via a pulmonary artery catheter
(PAC).
For
example, consider the hypotensive, tachycardic patient above. Assume a CVP of 1
mmHg (normal 5–12 mmHg) and cardiac output of 6.5 L/min. A low filling pressure
(low preload) translates into low ventricular volume – but contractility
appears to be good (a cardiac output of 6.5 L/min is not consistent with a
poorly contracting heart). With a look at the systemic vascular resistance
(SVR) equa-tion above, we see that a low MAP (small numerator) and high cardiac
output (denominator) implies a very low SVR. The baroreflex, though, should be
railing against the low BP and raising
the SVR – we cannot measure the baroreflex activ-ity but assume that it is
straining to raise resistance, without success. Thus, our attention is drawn to
vasodilation (via endotoxin as in septic shock, or blockade of sympathetic
outflow as in spinal shock or neuraxial anesthesia).
Such a
physiologic approach allows tailoring of intervention to the specific problem.
While intravenous fluid administration is routinely our first choice in a
hypotensive patient – particularly in the post-operative setting – and proves
the correct choice 99 times out of 100, it does no favor for the patient
hypotensive from CHF. Thus, with an unclear etiology or a troubling response to
initial treatment, invasive monitoring may be helpful (Table 9.2).
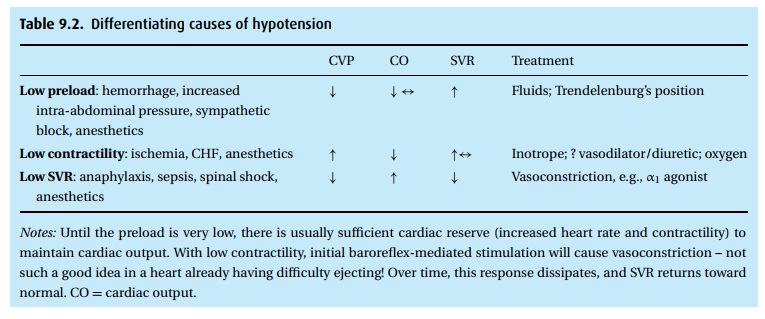
We see
from the table that the typical general inhalation anesthetic can affect blood
pressure from top to bottom, decreasing preload by venous pooling, decreasing
contractility by a direct negative inotropic effect on the heart, decreas-ing
SVR by depressing sympathetic outflow and, with some agents, even decreas-ing
the baroreceptor reflex. The treatment of hypotension under anesthesia – when
not attributed to primary heart disease, hypovolemia from hemorrhage or sepsis
– still consists of “filling up the tank” by giving fluids, lightening
anes-thesia to improve cardiac function. and giving sympthomimetic drugs, such
as ephedrine, to raise SVR and contractility.
Arrhythmias
Rhythm
disturbances occur in up to 70% of patients subjected to general anesthe-sia.
Fortunately, the majority of these, in the otherwise healthy patient, are
benign and transient. A number of factors can be blamed: the effects of
anesthetic agents on the SA and AV nodes, peri-operative ischemia, and
increased sympathetic activity during light anesthesia, e.g., laryngoscopy,
hypoxemia, and hypercarbia (not uncommon during induction of general
anesthesia). In addition to adher-ing to ACLS protocols, potential triggers
must be sought and eliminated: correct ventilation, alter anesthetic agent
selection (no halothane), increase oxygenation, deepen anesthetic, etc.
Hypertension
The
differential diagnosis of intraoperative hypertension is lengthy, but should be
approached by considering the patient and procedure first (Table 9.3).
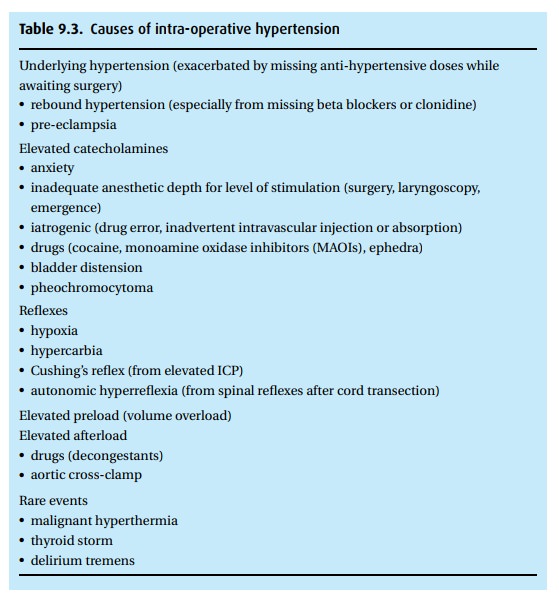
Management
of intra-operative hypertension should focus on three things:
(i) Fix the underlying problem: correct anesthetic
depth, treat hypercarbia, drain the bladder, etc.
(ii) Where correction is not possible: treat
according to the physiologic derange-ment. For example, volume overload should
not be treated with beta-blockade nor anxiety with diuretics.
(iii) Consider the time course of the treatment: if a
patient’s hypertension results from a transient surgical stimulus, a
long-acting anti-hypertensive may cause refractory hypotension when the
stimulus ends.
See the
pharmacology section to review a selection of the myriad anti-hypertensive
agents at our disposal.
Related Topics