Chapter: Medical Microbiology: An Introduction to Infectious Diseases: Viral Structure
Capsid Structure - Viral Structure
CAPSID STRUCTURE
Subunit Structure of Capsids
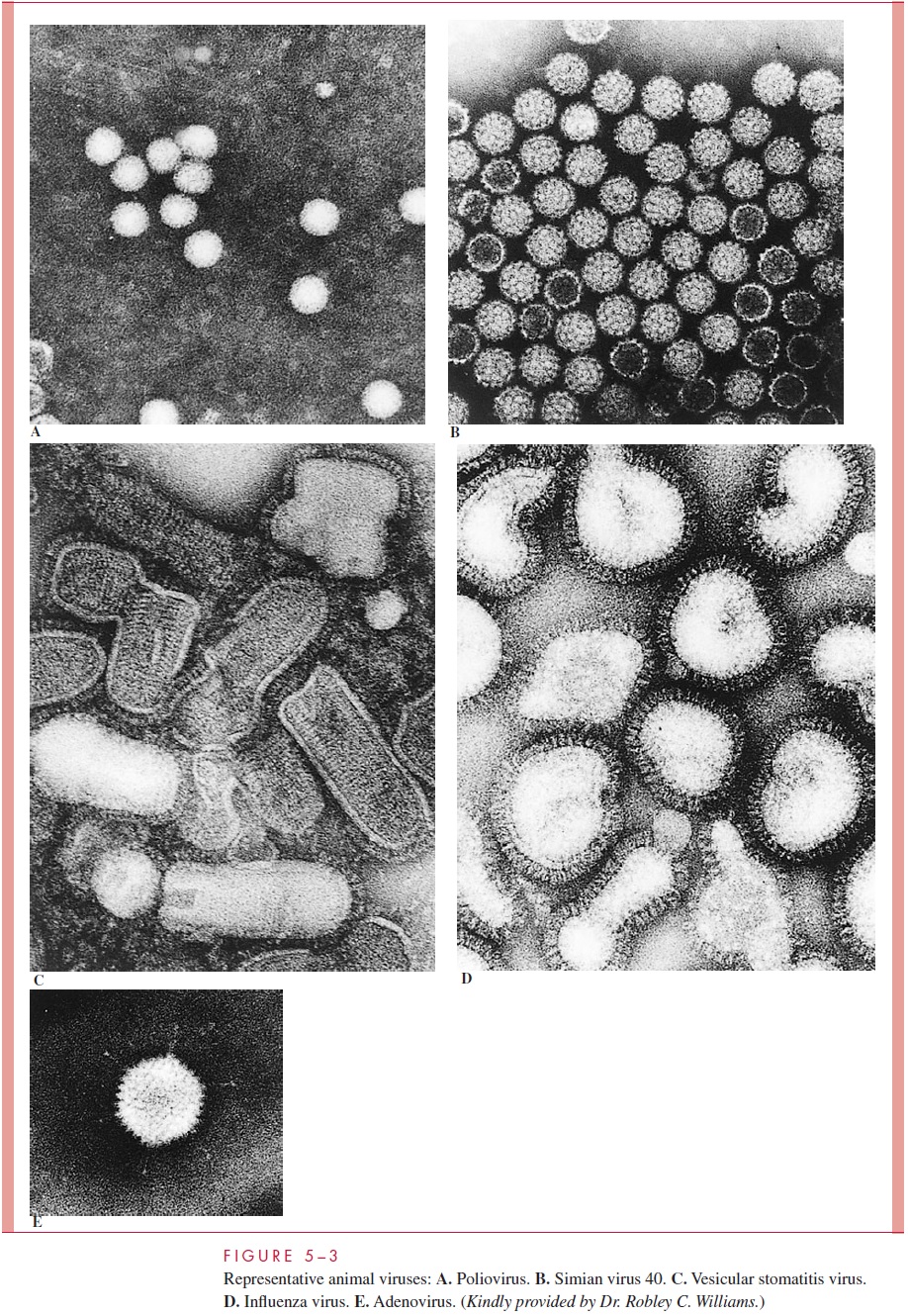
The capsids or nucleocapsids of all viruses are composed of many copies of one or at most several different kinds of protein subunits. This fact follows from two fundamental considerations. First, all viruses code for their own capsid proteins, and even if the entire coding capacity of the genome were to be used to specify a single giant capsid protein, the protein would not be large enough to enclose the nucleic acid genome. Thus, multiple protein copies are needed, and, in fact, the simplest spherical virus contains 60 identical protein subunits. Second, viruses are such highly symmetric structures that it is not un-common to visualize naked capsid viruses in the electron microscope as a crystalline array (eg, simian virus 40 in Fig 5 – 3B). The simplest way to construct a regular symmet-rical structure out of irregular protein subunits is to follow the rules of crystallography and form an aggregate involving many identical copies of the subunits, where each sub-unit bears the same relationship to its neighbors as every other subunit.
The presence of many identical protein subunits in viral capsids or the existence of many identical spikes in the membrane of enveloped viruses has important implications for adsorp-tion, hemagglutination, and recognition of viruses by neutralizing antibodies .
Cylindrical Architecture
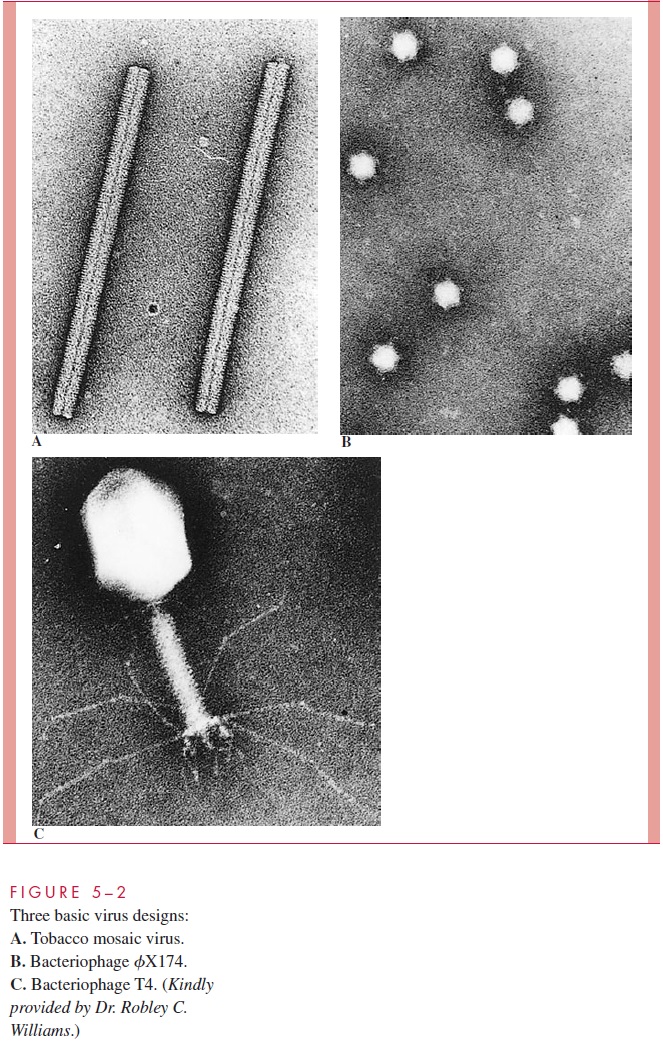
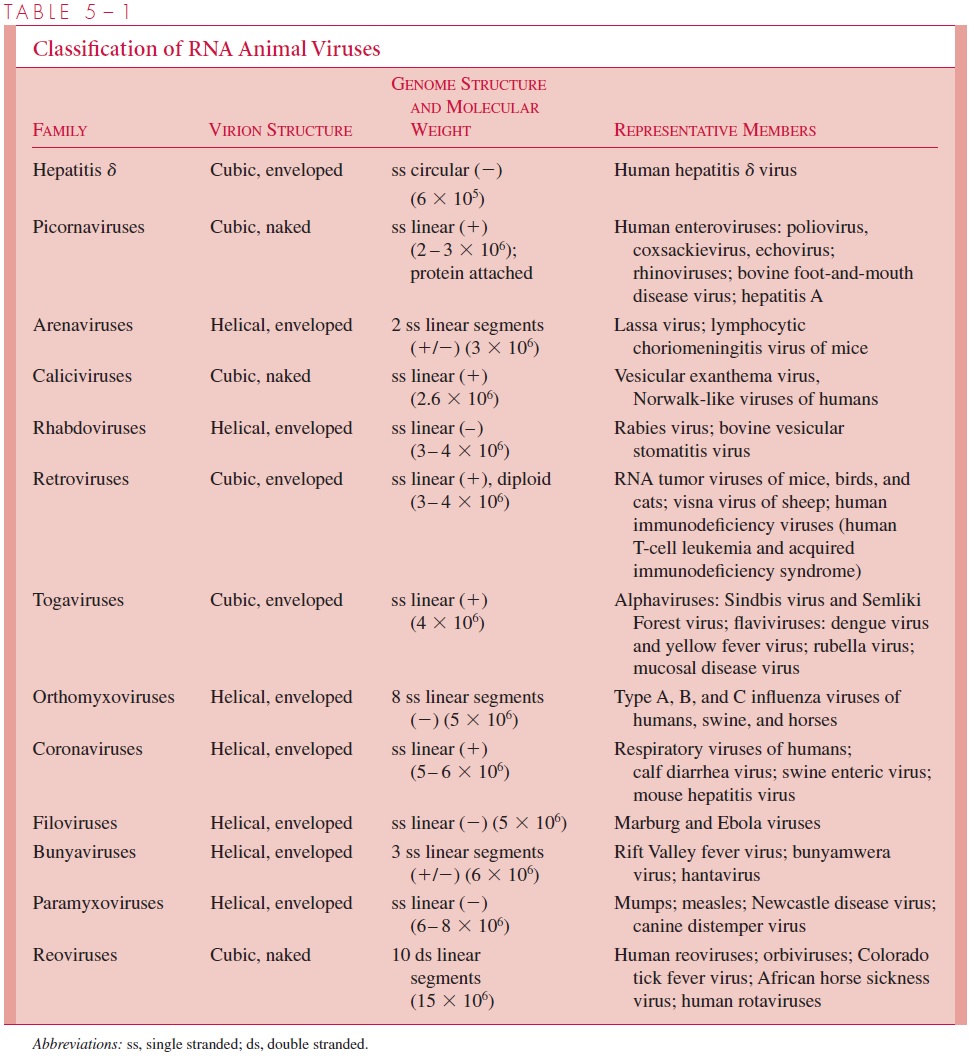
A cylindrical shape is the simplest structure for a capsid or a nucleocapsid. The first virus to be crystallized and studied in structural detail was a plant pathogen, tobacco mosaic virus (TMV) (see Fig 5 – 2A). The capsid of TMV is shaped like a rod or a cylinder, with the RNA genome wound in a helix inside it. The capsid is composed of multiple copies of a single kind of protein subunit arranged in a close-packed helix, which places every sub-unit in the same microenvironment. Because of the helical arrangement of the subunits, viruses that have this type of design are often said to have helical symmetry. Although less is known about the architecture of animal viruses with helical symmetry, it is likely their structures follow the same general pattern as TMV. Thus, the nucleocapsids of influenza, measles, mumps, rabies, and poxviruses (Table 5 – 1) are probably constructed with a heli-cal arrangement of protein subunits in close association with the nucleic acid genome.
Spherical Architecture
The construction of a spherically shaped virus similarly involves the packing together of many identical subunits, but in this case the subunits are placed on the surface of a geo-metric solid called an icosahedron. An icosahedron has 12 vertices, 30 sides, and 20 tri-angular faces (Fig 5 – 4). Because the icosahedron belongs to the symmetry group that crystallographers refer to as cubic, spherically shaped viruses are said to have cubic sym-metry. (Note that the term cubic, as used in this context, has nothing to do with the more familiar shape called the cube.)
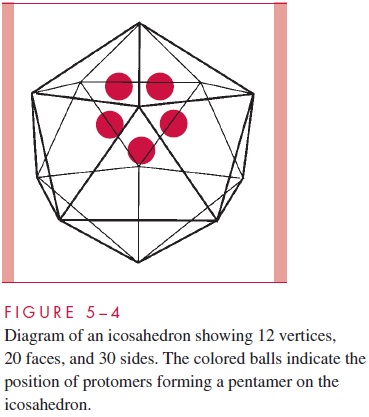
When viewed in the electron microscope, many naked capsid viruses and some nucle-ocapsids appear as spherical particles with a surface topology that makes it appear that they are constructed of identical ball-shaped subunits (see Fig 5 – 3B and E). These visi-ble structures are referred to asmorphological subunits, or capsomeres. A capsomere is generally composed of either five or six individual protein molecules, each one referred to as a structural subunit, or protomer. In the simplest virus with cubic symmetry, five protomers are placed at each one of the 12 vertices of the icosahedron as shown in Figure 5 – 4 to form a capsomere called a pentamer. In this case, the capsid is composed of 12 pentamers, or a total of 60 protomers. It should be noted that as in the case of helical symmetry, this arrangement places every protomer in the same microenvironment as every other protomer.
To accommodate the larger cavity required by viruses with large genomes, the capsids contain many more protomers. These viruses are based on a variation of the basic icosa-hedron in which the construction involves a mixture of pentamers and hexamers instead of only pentamers. A detailed description of this higher level of virus structure is beyond the scope of this text.
Special Surface Structures
Many viruses have structures that protrude from the surface of the virion. In virtually every case these structures are important for the two earliest steps of infection, adsorption and penetration. The most dramatic example of such a structure is the tail of some bacteriophages (see Fig 5 – 2C), which, acts as a channel for the transfer of the genome into the cell. Other examples of surface structures include the spikes of adenovirus (see Fig 5 – 3E) and the glycoprotein spikes found in the membrane of enveloped viruses (see influenza virus in Fig 5 – 3D). Even viruses without obvious surface extensions probably contain short projections, which, like the more obvious spikes, are involved in the specific binding of the virus to the cell surface .
Classification of Viruses
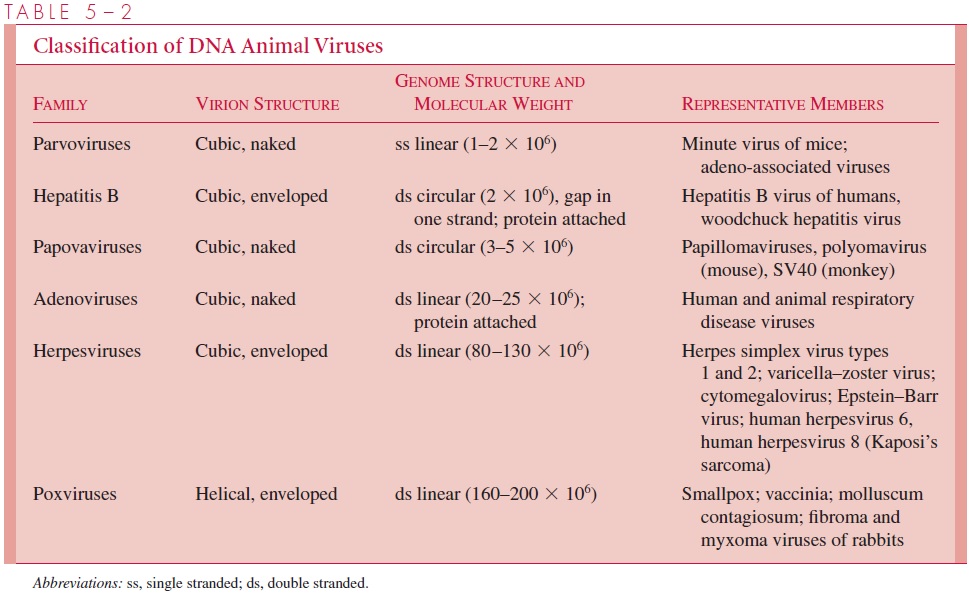
Tables 5 – 1 and 5 – 2 present a classification scheme for animal viruses that is based solely on their structure. The viruses are arranged in order of increasing genome size. It is important to bear in mind that phylogenetic relationships cannot be inferred from this tax-onomic scheme. The tables should not be memorized, but instead used as a reference guide to virus structure.
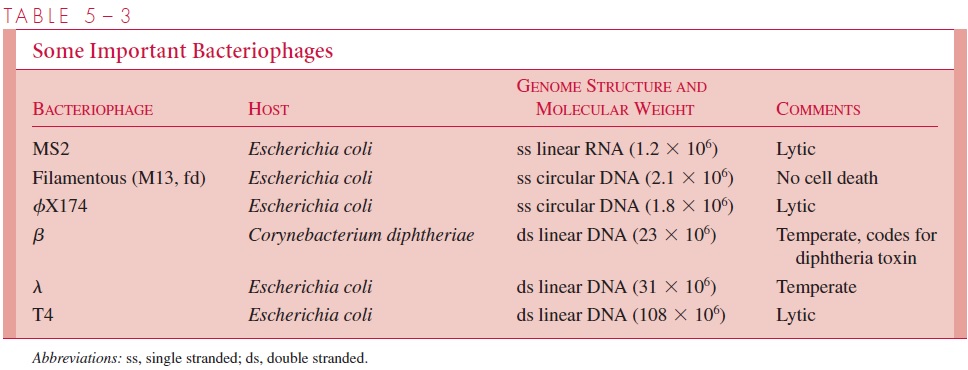
Representative and important bacteriophages are listed along with their properties in Table 5 – 3.
Related Topics