Thermal Physics | Science - Book Back Questions with Answers | 10th Science : Chapter 3 : Thermal Physics
Chapter: 10th Science : Chapter 3 : Thermal Physics
Book Back Questions with Answers
Thermal Physics - Science
I. Choose the correct answer
1. The value of universal gas constant
a) 3.81 mol–1 K–1
b) 8.03 mol–1 K–1
c) 1.38 mol–1 K–1
d) 8.31 mol–1 K–1
2. If a substance is heated or cooled, the change in mass of that substance is
a) positive
b) negative
c) zero
d) none of the above
3. If a substance is heated or cooled, the linear expansion occurs along the axis of
a) X or –X
b) Y or –Y
c) both (a) and (b)
d) (a) or (b)
4. Temperature is the average ___________ of the molecules of a substance
a) difference in K.E and P.E
b) sum of P.E and K.E
c) difference in T.E and P.E
d) difference in K.E and T.E
5. In the Given diagram, the possible direction of heat energy transformation is
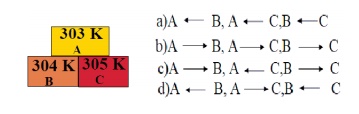
a) A ← B,A ← C,B ← C
b) A → B,A → C,B → C
c) A → B,A ← C,B → C
d) A ← B,A → C,B ← C
II. Fill in the blanks:
1. The value of Avogadro number
2. The temperature and heat are Scalar quantities
3. One calorie is the amount of heat energy required to raise the temperature of 1 gm of water through 1°C.
4. According to Boyle’s law, the shape of the graph between pressure and reciprocal of volume is
III. State whether the following statements are true or false, if false explain why?
1. For a given heat in liquid, the apparent expansion is more than that of real expansion. - False
The real expansion is more (or) less than that of apparent expansion.
2. Thermal energy always flows from a system at higher temperature to a system at lower temperature. - True
3. According to Charles’s law, at constant pressure, the temperature is inversely proportional to volume.
Volume is directly proportional to temperature at constant pressure.
IV. Match the items in column-I to the items in column-II
Column-I Column-II
1. Linear expansion - (a) change in volume
2. Superficial expansion - (b) hot body to cold body
3. Cubical expansion - (c) 1.381 X 10-23 JK-1
4. Heat transformation - (d) change in length
5. Boltzmann constant - (e) change in area
Answer:
1. Linear expansion - Change in length
2. Superficial expansion - change in area
3. Cubical expansion - change in volume
4. Heat transformation - hot body to cold body
5. Boltzmann constant - 1.381 x 10-23 JK-1
V. Assertion and reason type questions
a. Both the assertion and the reason are true and the reason is the correct explanation of the assertion.
b. Both the assertion and the reason are true but the reason is not the correct explanation of the assertion.
c. Assertion is true but the reason is false.
d. Assertion is false but the reason is true.
1. Assertion: There is no effects on other end when one end of the rod is only heated.
Reason: Heat always flows from a region of lower temperature to higher temperature of the rod.
Answer: a. Both the assertion and the reason are true and the reason is the correct explanation of the assertion.
2. Assertion: Gas is highly compressible than solid and liquid
Reason: Interatomic or intermolecular distance in the gas is comparably high.
Answer: c. Assertion is true but the reason is false.
VI. Answer in briefly
1. Define one calorie.
One calorie is the amount of heat required to rise the temperature of 1 gram of water through 1°C.
2. Distinguish between linear, arial and superficial expansion.
Liner Expansion
1. Also called longitudinal expansion
2. αL = ΔL/L0ΔT
arial Expansion and superficial expansion
1. Also called as superficial expansion
2. αA = ΔA/A0ΔT
3. What is co-efficient of cubical expansion?
The ratio of increase in volume of the body per degree rise in temperature to its unit volume is called as co-efficient of cubical expansion. It's unit is K-1.
4. State Boyle’s law
When the temperature is kept constant, the volume of a fixed mass of gas is inversely proportional to its pressure. P ∝ 1/V
PV = constant
5. State-the law of volume
According to this law, when the pressure of gas is kept constant, the volume of a gas is directly proportional to the temperature of the gas.
V ∝ T or V/T = constant
6. Distinguish between ideal gas and real gas.
Ideal gas
1. If the atoms or molecules of a gas do not interact with each other, then the gas is said to be an ideal gas or a perfect gas.
2. Ideal gas does not have volume.
Real gas
1. If the molecules or atoms of a . gas interact with each other with a definite amount of intermolecular or inter atomic force of attraction, then the gas is said to be a real gas.
2. Real gas has volume.
7. What is co-efficient of real expansion?
The ratio of true rise in the volume of the liquid to unit original volume when the temperature rises by one kelvin is called as “Coefficient of real expansion”. Its SI unit is per K or K-1.
8. What is co-efficient of apparant expansion?
Co-efficient of apparent expansion is defined as the ratio of the apparent rise in the volume of the liquid per degree rise in temperature to its unit volume. The SI unit of coefficient of apparent expansion is K-1.
VII. Numerical problems
1. Find the final temperature of a copper rod. Whose area of cross section changes from 10 m2 to 11 m2 due to heating. The copper rod is initially kept at 90 K. (Coefficient of superficial expansion is 0.0021 /K)
Given
Area of copper rod, A = 10 m2
After expansion, A2 = 11 m2
Initial temperature T1 = 90 K
Coefficient of superficial expansion = 0.0021 / K
To find: Final temperature T2 = ?
Solution
ΔA/A = α ΔT
(A2-A1)/A = α Δ(T2-T1)
11-10 / 10 = 0.0021(T2 - 90)
1 / [ 10 x 0.0021 ] = (T2 - 90)
T2 = (1/0.021) + 90 = 47.6 + 90
Final Temperature, T2 = 137.6 K
2. Calculate the coefficient of cubical expansion of a zinc bar. Whose volume is increased 0.25 m3 from 0.3 m3 due to the change in its temperature of 50 K.
Given
Volume of the zinc bar V = 0.3 m3
Change in volume ΔV = 0.25 m3
Change in temperature ΔT = 50 K
To find : Coefficient of cubic expansion = ?
Solution
ΔV/V = αVΔT
αV = ΔV / V. ΔT
αV = 0.25 / (0.3 x 50) = 0.25 / 15 = 0.0166 K-1
Coefficient of cubical expansion,
αV = 0.0166 K-1.
VIII. Answer in detail
1. Derive the ideal gas equation.
Answer:
(i) The ideal gas equation is an equation which
relates all the properties of an ideal gas such as pressure (P), volume (V),
temperature (T) and the amount of the gas.
(ii) An ideal gas obeys Boyle’s law and Charles’s law
and Avogadro’s law.
(iii) According to Boyle’s law,
PV
= constant ... (1)
(iv) According to Charles’s law,
V/T
= constant ... (2)
(v) According to Avogadro’s law,
V/n
= constant ... (3)
(vi) After combining equations (1), (2) and (3), we
can write the following equation.
PV/nT
= constant ... (4)
The
above relation is called the combined gas law.
(vii) If we consider a gas which contains μ moles of
the gas, the number of atoms contained will be equal to μ times the Avogadro
number, NA. i.e., n = μNA.
(viii) Using this value, equation (4) can be written as
PV/ μNAT = constant
(ix) The value of constant in the above equation is
taken to be kB, which is called as Boltzmann’s constant.
Its
value is 1.381 × 10-23 JK-1.
(x) Hence, we have the following equation:
PV/
μNAT = kB
PV
= μNA kB T
(xi) Here, μNA kB = R, which is
termed as universal gas constant whose value is 8.31
J
mol-1K-1.
PV
= RT ...
(5)
This
is called ideal gas equation.
2. Explain the experiment of measuring the real and apparent expansion of a liquid with a neat diagram.
Answer:
(i) The liquid whose real and apparent expansion is
to be determined is poured in a container up to a level. Mark this level as L1.
(ii) Now, heat the container and the liquid using a
burner. Initially, the container receives the thermal energy and it expands.
(iii) As a result, the volume of the liquid appears to
have reduced. Mark this reduced level of liquid as L2.
(iv) On further heating, the thermal energy is
supplied to the liquid resulting in the expansion of the liquid. Hence, the
level of liquid rises to L3.
(v) Now, the difference between the levels L1
and L3 is called as apparent
expansion, and the difference between the levels L2 and L3
is called real expansion.
(vi) The real expansion is always more than the
apparent expansion.
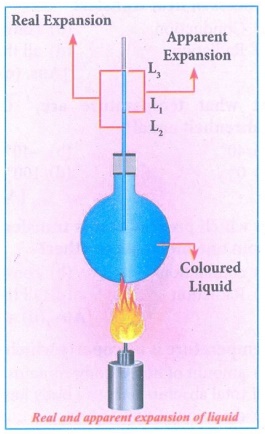
(vii) Real expansion = L3 - L2
Apparent
expansion = L3 - L1
IX. HOT question
If you keep ice at 0° C and water at 0°C in either of your hands, in which hand you will feel more chillness? Why?
We will feel more chillness in the hand ice is placed at 0° C latent heat absorbed by the ice is greater than that absorbed by water.
Reason : The latent heat of vaporization absorbed by the ice is greater than water, at the same temperature.
Related Topics