Chapter: Aquaculture Engineering : Disinfection
Basis of disinfection - Aquaculture Engineering
Basis of disinfection
Degree of removal
The term percentage removal of actual micro-organisms is used in environmental engineering. In microbiological terms log10 removal or inactivation (decimal removal) is used to define the disinfection yield; normally a reduction of between 99 and 99.99% of the total number of bacteria is wanted, which corresponds to a log disinfection of 2ŌĆō4. However, these terms do not give exact values of the number of micro-organisms left; they only indicate by how much numbers are reduced from the starting concentration.
Example
The normal concentration of bacteria is 107/ml and a reduction of 99.9% is required. Find the concen-tration of bacteria present after disinfection.
Solution
concentration of bacteria after infection = 107(1 ŌłÆ 0.999)

Example
The starting concentration of bacteria is 107/ml. A log disinfection of 3 is wanted. Calculate the new concentration of bacteria.
Solution
Let the starting concentration be N1 and the end con-centration N2; log(disinfection) = 3.
log(disinfection) = log(N1/N2)
= logN1 ŌłÆ logN2log N2= log N1ŌłÆlog(disinfection)
=7 ŌĆō 3
=4
N2=104/ml
ChickŌĆÖs law
Inactivation of micro-organisms in a disinfection plant depends on the time that the micro-organism are exposed to the disinfectant. This is described by ChickŌĆÖs law:
dn / dt = kN
where:
dn/dt= necessary time to inactivate n micro-organisms
k =time constant depending on disinfectant,type of micro-organism and water quality
N =number of live micro-organisms t =time.
This differential equation can be integrated within limits to give the following equation:
N1= N0eŌłÆkt
where:
N0=number of micro-organisms at the start
N1=number of micro-organisms after time t.
WatsonŌĆÖs law
Based on the results of ChickŌĆÖs law, WatsonŌĆÖs law can be developed:
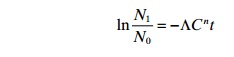
where:
╬ø = coefficient of specific toxicity
C =concentration of disinfectant
n =exponent (normally around 1)
t =time after start-up.
This means that the relation between the number of active and inactive micro-organisms is a product of the concentration of the disinfectant and the exposure time.
Dose-response curve
Based on WatsonsŌĆÖs law a dose-response relation may be established for specific types of micro-organism. This gives the proportion of micro-organisms inactivated by fixed doses of disinfectant over various time periods. Exact dose-response relationships are difficult to determine in practice for several reasons. It is often difficult to isolate new pathogens and the response to a certain dose depends, amongst other factors, on the immune status of the organism, environmental conditions and population density.
Related Topics