Chapter: Clinical Anesthesiology: Perioperative & Critical Care Medicine: Nutrition in Perioperative & Critical Care
Basic Nutritional Needs
BASIC NUTRITIONAL NEEDS
Maintenance of normal body mass, composition, structure, and function
requires the periodic intake of water, energy substrates, and specific
nutrients. Nutri-ents that cannot be synthesized from other nutrients are
characterized as “essential.” Remarkably, relatively few essential nutrients
are required to form the thou-sands of compounds that make up the body. Known
essential nutrients include 8–10 amino acids, 2 fatty acids, 13 vitamins, and
approximately 16 minerals.
Energy is normally derived from dietary or
endogenous carbohydrates, fats, and protein. Meta-bolic breakdown of these
substrates yields the ade-nosine triphosphate required for normal cellular
function. Dietary fats and carbohydrates normally supply most of the body’s
energy requirements. Dietary proteins provide amino acids for protein
synthesis; however, when their supply exceeds requirements, amino acids also
function as energy substrates. The metabolic pathways of carbohydrate, fat, and
amino acid substrates overlap, such that some interconversions can occur
through metabolic intermediates (see Figure 32–4). Excess amino acids can
therefore be converted to carbohydrate or fatty acid precursors. Excess
carbohydrates are stored as glycogen in the liver and skeletal muscle. When
glycogen stores are saturated (200–400 g in adults), excess carbohydrate is
converted to fatty acids and stored as triglycerides, primarily in fat cells.
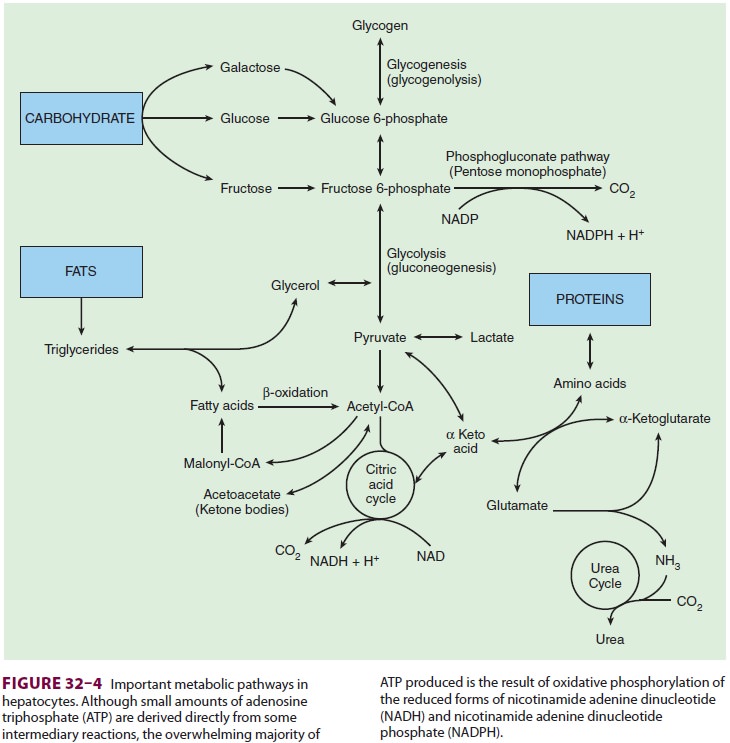
During starvation, the protein content of
essen-tial tissues is spared. As blood glucose concentration begins to fall
during fasting, insulin secretion decreases, and counterregulatory hormones,
such as glucagon, increase. Hepatic and, to a lesser extent, renal
glycogenolysis and gluconeogenesis are enhanced. As glycogen supplies are
depleted (within 24 h), gluconeogenesis (from amino acids) becomes increasingly
important. Only neural tissue, renal medullary cells, and erythrocytes continue
to utilize glucose—in effect, sparing tissue proteins. Lipolysis is enhanced,
and fats become the principal energy source. Glycerol from the triglycerides
enters the gly-colytic pathway, and fatty acids are broken down to
acetylcoenzyme A (acetyl-CoA). Excess acetyl-CoA results in the formation of
ketone bodies (ketosis). Some fatty acids can contribute to gluconeogenesis. If
starvation is prolonged, the brain, kidneys, and muscle also begin to utilize
ketone bodies efficiently.The previously well-nourished
patient under-going elective surgery could be fasted for upto a week
postoperatively without apparent adverse effect on outcomes, provided fluid and
electro-lyte needs are met. The usefulness of nutritional repletion in the
immediate postoperative period is not well defined, but likely relates to the
degree of malnutrition, number of nutrient deficiencies, and severity of the
illness/injury. Moreover, the optimal timing and amount of nutrition support
following acute illness remain unknown. On the other hand, malnourished
patients may benefit from nutritional repletion prior to surgery.
Modern surgical practice has evolved to an expec-tation of an
accelerated recovery. Accelerated recov-ery programs generally include early
enteral feeding, even in patients undergoing surgery on the gastroin-testinal
tract, so prolonged periods of postoperative starvation are no longer common
practice. All well-nourished patients should receive nutritional support after
5 days of postsurgical starvation, and those with ongoing critical illness or
severe malnutrition should be given nutritional support immediately. The
mal-nourished patient presents a different set of issues, and such patients may
benefit from both preoperative and early postoperative feeding. Clearly, the
healing of wounds requires energy, protein, lipids, electro-lytes, trace
elements, and vitamins. Depletion of any of these substrates may delay wound
healing and pre-dispose to complications, such as infection. Nutrient depletion
may also delay optimal muscle functioning, which is important for supporting
increased respira-tory demands and early mobilization of the patient.
The resting metabolic rate can be measured
(but often inaccurately) using indirect calorimetry (known as a metabolic cart)
or by estimating energy expenditure using standard nomograms (such as the
Harris–Benedict equation), yielding an approxima-tion of daily energy
requirements. Alternatively, a simple and practical approach assumes that
patients require 25–30 kcal/kg daily. The weight is usually taken as the ideal
body weight or adjusted body weight. Even though nutritional requirements can
increase greatly above basal levels with certain condi-tions (eg, burns), the
more often relevant reason fordetermining the daily requirements is to ensure
that patients are not unnecessarily overfed. In this regard, obese patients
require adjusting the body weight based on the degree of obesity to prevent
overfeeding.
Related Topics