Chapter: Medical Microbiology: An Introduction to Infectious Diseases: Host-Parasite Relationships
Avoiding the Host Immune System
AVOIDING THE HOST IMMUNE SYSTEM
The host immune system evolved in large part because of the selective pressure of microbial attack. To be successful, microbial pathogens must escape this system at least long enough to be transmitted to a new susceptible host or to take up residence within the host in a way that is compatible with mutual coexistence.
Serum Resistance
Many bacteria that come into contact with human complement can be destroyed by op-sonization or by direct lysis of the bacterial membrane by complement complexes. Some can avoid this fate by a process called serum resistance. Pathogenic Salmonella possess a lipopolysaccharide inhibiting the C5b – 9 complement complex from attacking the hy-drophobic domains of the bacterial outer membrane. Other bacteria employ different mechanisms, but the end result is the same. These organisms can persist in an environ-ment that is rapidly lethal for nonpathogens.
Antiphagocytic Activity
A fundamental requirement for many pathogenic bacteria is escape from phagocytosis by macrophages and polymorphonuclear leukocytes. It seems likely that the ability to avoid phagocytosis was an early necessity for microorganisms following the evolution of preda-tory protozoans. Some bacteria such as the causative agent of Legionnaires’ disease,Legionella pneumophila, learned how to replicate in free-living amoebae following phagocytosis and used them as part of their life cycle. Legionella uses similar mecha- nisms to outwit human macrophages. In this one example, it can be seen that pathogenic-ity in some microorganisms evolved from a very early time in their development.
The most common bacterial means to avoid phagocytosis is an antiphagocytic capsule. The significance of the bacterial capsule can hardly be overemphasized. Almost all princi-pal pathogens that cause pneumonia and meningitis have antiphagocytic polysaccharide capsules. Nonencapsulated variants of these organisms are usually avirulent. In many cases, it has been found that the capsule of pathogens prevents complement deposition on the bacterial cell surface. Thus, the capsule prevents nonimmune opsonization and con-fers resistance to phagocytosis. As noted earlier, along with encapsulation, a common fac-tor of many organisms that cause pneumonia and meningitis is the elaboration of an enzyme that specifically cleaves human IgA1 molecules. IgA proteases are found in the pathogenic Neisseria, Haemophilus influenzae type b (Hib), and Streptococcus pneumo-niae. The combination of a capsule to avoid opsonization and/or an enzyme that cleavesan important class of secretory antibody is a potent stratagem to avoid phagocytosis.
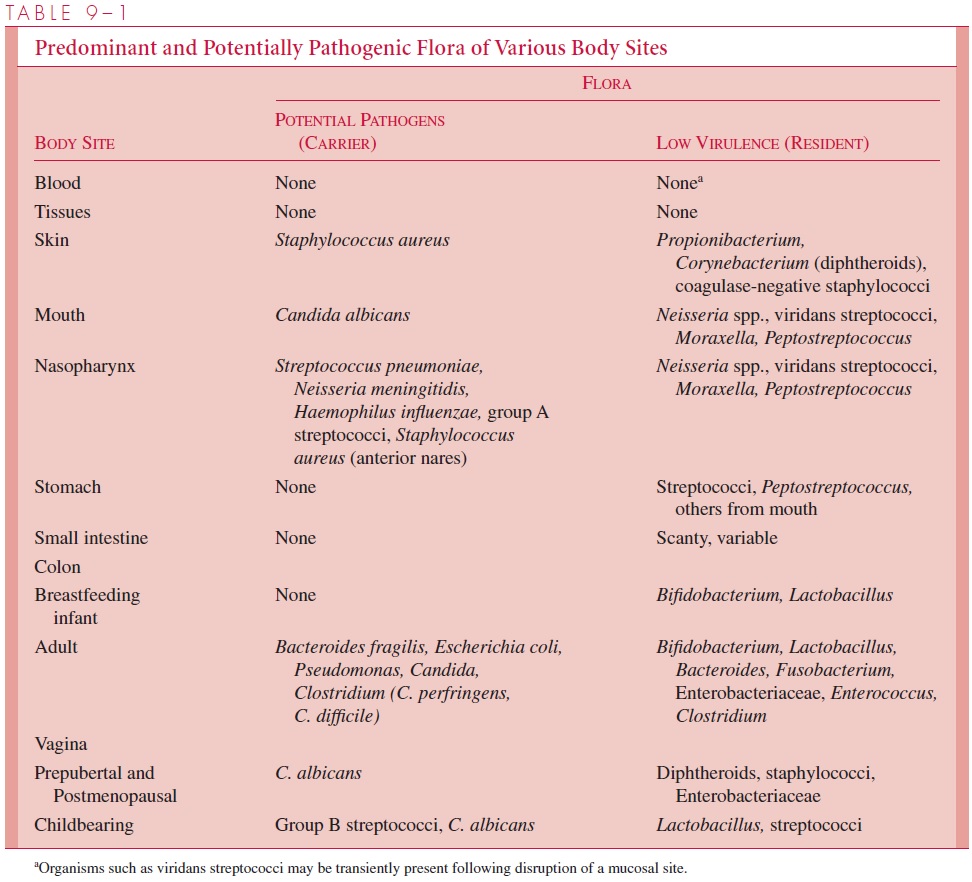
The group A streptococcal M protein is another example of a bacterial surface product employed by the organism to escape opsonization and phagocytosis. In part, this is a re-flection of the ability of M protein to bind fibrinogen and its breakdown product fibrin to the bacterial surface. This sterically hinders complement access and prevents opsoniza-tion. There are many other examples. The principle is clear. If microorganisms can inhibit phagocytosis, they can often gain the upper hand long enough to replicate sufficiently to establish themselves in the host or become transmitted to a new host. It is important to understand that these encapsulated pathogens are often carried asymptomatically in the normal flora (see Table 9 – 1). The capsule is important for the organism to establish itself in the nasopharynx.
The host responds to its initial encounter with the encapsulated organism by elaborat-ing anticapsular antibodies that opsonize and permit efficient phagocytosis and destruc-tion of the microorganism in subsequent encounters. Thus, the initial interaction between an encapsulated microbe and its host usually has two outcomes. First, the host becomes asymptomatically colonized, and, second, the colonization is an immunizing event for the host. The host is protected against serious systemic infection by the organism, but this im-munity may not affect the capacity of the organism to live happily on a mucosal surface. Epidemiologic investigations show that serious disease caused by encapsulated pathogens when it occurs does so shortly after a susceptible individual encounters the microorgan-ism for the first time. This scenario contrasts with the idea that carriers of microorganisms come down with the disease at some time in the future. If a microorganism meets a host with a compromised immune system or some short-term deficit in its defense systems, then the organism’s capacity for replication can overwhelm the host defense mechanisms and cause serious disease. Once colonization and immunity have been established, the steady state is a satisfactory host – parasite relationship. For example, the outcome of en-counters with Neisseria meningitidis in military recruits followed for colonization and an-ticapsular antibody throughout training camp has been demonstrated. Disease developed only in those entering the camp lacking both specific antibody and nasopharyngeal colo-nization with the N. meningitidis serogroup responsible for a subsequent meningitis out-break. Unaffected recruits either had a “successful” encounter followed by development of antibody or already had protective antibody, presumably from a similar experience ear-lier in life.
Cutting Lines of Communication
Pathogens such as Yersinia and Salmonella have evolved means to neutralize phagocytes directly by using the equivalent of eukaryotic signal transduction molecules. PathogenicYersinia synthesize tyrosine phosphatase molecules and serine kinase molecules and in-troduce them into the cytoplasm of macrophages, which leads to a complete loss in the capacity of these cells either to phagocytose microorganisms or to signal other compo- nents of the host immune system by cytokine release. Likewise, bothSalmonellaandYersinia inject bacterial proteins into the cytoplasm of host cells that directly induceapoptosis or disrupt cellular function. As noted earlier, microbial mimicry can have the same effect by concealing the microorganism under a shroud of host proteins;however, the strategy of directly interfering with host cell function by use of an alternative enzyme or modifying and activating existing host cell effectors has been discovered to be a more common pathogenic strategy for the invading microbe than previously realized. This helps resolve the mystery that bacteria not known to produce toxins nonetheless cause cellular toxicity.
Antigenic Variation
Another method by which microorganisms avoid host immune responses is by varying sur-face antigens. N. gonorrhoeae displays an endless array of pili and outer membrane proteins to the host immune system. The organism has learned to preserve its binding specificity but to vary endlessly the molecular scaffolding on which the functional units are placed. The host “sees” a bewildering array of new epitopes, whereas the critical regions of the mole- cule remain hidden from immune surveillance. Of course, among the viruses, antigenic variation is also a common theme; the best known example is the influenza virus. It is instructive that in both the bacterial example and the viral example, recombination mecha-nisms act to bring together novel sequences of genetic material. A number of microorgan-isms known for their antigenic diversity such as those of the genus Borrelia, which causes relapsing fever, and the group A streptococci also use homologous recombination of DNA from repeated sequences to generate the diversity in size and sequence observed in their principal immunodominant antigens.
Related Topics