Chapter: Pharmaceutical Drug Analysis: Flame Spectroscopy
Applications of Flame Emission Spectroscopy in Pharmaceutical Analysis
APPLICATIONS OF FLAME EMISSION SPECTROSCOPY IN PHARMACEUTICAL ANALYSIS
A few typical example of ‘flame-emission spectroscopy’ are given
below.
1. ASSAY OF SODIUM, POTASSIUM AND CALCIUM IN BLOOD SERUM AND WATER
(i) Standard potassium and sodium solutions,
approximately 500 ppm : Weigh accurately 0.95 g of dried KCl and 1.25 g
of dried NaCl into separate 1-litre volumetric flasks. Dissolve in water and
dilute to the mark.
(ii) Standard calcium solution, approximately
500 ppm : Weigh accurately 1.25 g of CaCO3, which has been dried
at 110°C, into a 500-ml breaker. Add about 200 ml of DW and 10 ml of conc. HCl.
Cover the breaker with a watch-glass during addition of acid to prevent loss of
solution as CO2 is evolved. After the solution is complete, transfer
it quantitatively into a 1-litre volumetric flask and dilute to the mark with
DW.
(iii) Radiation buffer* for sodium determination
: Prepare a saturated solution with reagent-grade CaCl2, KCl,
MgCl2, in that order.
(iv) Radiation buffer for potassium
determination : Prepare a saturated solution with reagent-grade NaCl, CaCl2
and MgCl2, in that order.
(v) Radiation buffer for calcium determination
: Prepare a saturated solution with reagent-grade NaCl, KCl, MgCl2
in that order.
Procedure
(a) Preparation of working curves :
Transfer 5 ml of the appropriate radiation buffer to each series of 100-ml
volumetric flasks. Add a volume of the standard solution which will cover a
concentra-tion ranging between 0 to 100 ppm. Dilute to 100 ml with DW and mix
well. Measure the emis-sion intensity of these samples by taking at least three
readings for each. Between each set of measurements, aspirate DW through the
burner. Correct the average values for background lumi-nosity, and prepare a
working curve from these data.
(b) Analysis of blood serum/water sample :
Prepare aliquot portions of the sample as described in the above paragraph (a). If necessary, use a standard to
calibrate the response of the spectrometer to the working curve. Then measure
the emission intensity for the unknown. After correcting the data for
background, determine the concentration by comparison with the working curve.
2. ASSAY OF BARIUM, POTASSIUM AND SODIUM IN CALCIUM ACETATE
The technique of flame emission spectroscopy is used to
determine the concentration of Ba, K, and Na ions by measuring the intensity of
emission at a specific wavelength by the atomic vapour of the element generated
from calcium acetate i.e., by
introducing its solution into a flame.
2.1. For Emission Measurements
Introduce water into the atomic vapour generator, adjust
the instrument reading to zero, introduce the most concentrated solution into
the generator and adjust the sensitivity to give a suitable reading ; again
introduce water or the prescribed solution into the generator and when the
reading is constant readjust, if necessary, to zero.
2.2. Method of Standard Addition
The various steps are as follows :
Place in each of not fewer than three similar graduated
flasks equal volumes of the solutions of the substance being examined, prepared
as follows :
(a) Prepare a
5.0% w/v solution and use barium solution ASp*, suitably diluted with water to
prepare the standard solution.
(b) Prepare a
1.25% w/v solution and use potassium solution ASp**, suitably diluted with
mater, to prepare the standard solution.
(c) Prepare a
1.0% w/v solution and use sodium solution ASp***, suitably diluted with water,
to prepare the standard solution.
Add to all but one these
flasks a measured quantity of the specified standard solution (marked *; **;
***; above) to produce a series of solution containing increasing amounts of
the element being determined.
Dilute the contents of each
flask to the required volume with water.
After having calibrated the
instrument as directed above, introduce each solution into the genera-tor three
times and record the steady reading. If the generator is a flame, wash the
apparatus thoroughly with water ; if a furnace is used fire it after each
introduction.
Plot the mean of the readings
against concentration on a graph the axes of which intersect at zero added
element and zero reading.
Extrapolate the straight line
joining the points until it meets the extrapolated concentration axis. The
distance between this point and the intersection of the axes represents the
concentration of the element (e.g.,
Mg, K, Na) being determined in the solution of the substance being examined.
2.3. Limits of Elements present in Calcium Acetate Sample
Mg : Not more than 500 ppm of Mg ;
K : Not more than 0.1% of K,
and
Na : Not more than 0.5% of Na.
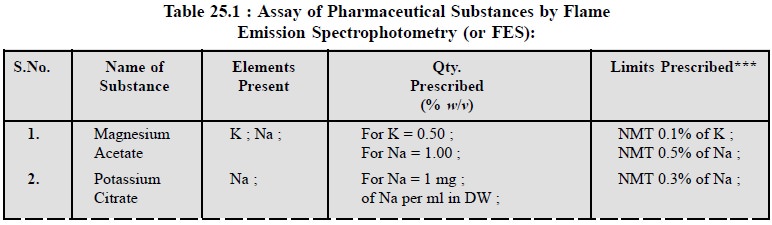
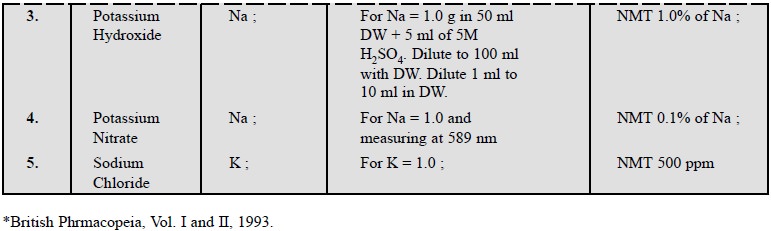
3. COGNATE ASSAYS
The following substance, namely : magnesium acetate ;
potassium citrate ; potassium hydroxide ; potassium nitrate and sodium chloride
can also be assayed for their respective elements as shown in Table 25.1 below.
However, all the respective solutions of the said pharmaceutical substance and
their standard solutions must be prepared as prescribed in BP (1993) strictly
to obtain the best results.
Related Topics