Chapter: Clinical Anesthesiology: Anesthetic Management: Anesthesia for Patients with Neurologic & Psychiatric Diseases
Anesthesia for Cerebrovascular Disease
Cerebrovascular Disease
Preoperative Considerations
Patients
with diagnosed cerebrovascular disease typically have a history of transient
ischemic attacks (TIAs) or stroke. Patients with TIAs undergoing surgery for
other indications have an increased risk of perioperative stroke. Asymptomatic
carotid bruits occur in up to 4% of patients older than age 40 years, but do
not necessarily indicate signifi-cant carotid artery obstruction. Fewer than
10% of patients with completely asymptomatic bruits have hemodynamically
significant carotid artery lesions. An asymptomatic carotid bruit may not
increase the risk of stroke following surgery, but increases the likelihood of
coexisting coronary artery disease. Moreover, the absence of a bruit does not
exclude significant carotid obstruction.
The
risk of perioperative stroke increases with patient age and varies with the
type of surgery. The overall risk of stroke associated with surgery is low, but
is greater in patients undergoing cardiovascu-lar surgery. Rates of stroke
after general anesthe-sia and surgery range from 0.08% to 0.4%. Even in
patients with known cerebrovascular disease, the risk is only 0.4% to 3.3%.
Patients at greatest risk of postoperative stroke are those undergoing open
heart procedures for valvular disease, coro-nary artery disease with ascending
aortic athero-sclerosis, and diseases of the thoracic aorta. Stroke following
open heart surgery is usually due to embolism of air, clots, or atheromatous
debris. Inone study, 6.1% of patients experienced an adverse neurological
outcome following cardiac surgery. Stroke following thoracic aortic surgery may
be due to emboli or ischemia secondary to prolonged circulatory arrest or a
clamp placed close to the ori-gin of the carotid artery.
The pathophysiology of
postoperative strokes following noncardiovascular surgery is less clear, but
may involve severe sustained hypotension or hypertension. Hypotension with
severe hypo-perfusion can result in so-called “watershed” zone infarctions or
thrombosis of cerebral arter-ies, whereas hypertension can result in
intracere-bral hemorrhage (hemorrhagic stroke). Sustained hypertension can
disrupt the blood–brain barrier and promote cerebral edema. Widened pulse
pres-sure (>80 mm Hg) can produce
endothelial vessel injury, potentially resulting in cerebral hypoperfu-sion or
embolism. Perioperative atrial fibrillation can likewise lead to atrial clot
formation and cere-bral embolism. The period of time during which anesthesia
and surgery should best be avoided following a stroke has not been determined.
Abnormalities in regional blood flow and metabolic rate usually resolve after 2
weeks, whereas altera-tions in CO 2 responsiveness and the
blood–brain barrier may require more than 4 weeks. However, urgent surgery is
performed for acute intracranial hemorrhage, symptomatic carotid disease, and
car-diac sources of emboli.
Patients with TIAs
have a history of tran-sient (<24 h) impairment, and,
by definition, no residual neurologic impairment. These attacks are thought to
result from emboli of fibrin-platelet aggregates or atheromatous debris from
plaques in extracranial vessels. Unilateral visual impairment, numbness or
weakness of an extremity, or aphasia is suggestive of carotid disease, whereas
bilateral visual impairment, dizziness, ataxia, dysarthria, bilateral weakness,
or amnesia is suggestive of vertebral–basilar disease. Patients with TIAs have
a 30% to 40% chance of developing a frank stroke within 5 years; 50% of these
strokes occur within the first year. Patients with TIAs should not undergo any
elective surgical procedure with-out an adequate medical evaluation that
generally includes at least noninvasive (Doppler) flow and imaging studies. The
presence of an ulcerative plaque of greater than 60% occlusion is generally an
indication for carotid endarterectomy or endo-vascular intervention.
PREOPERATIVE MANAGEMENT
Preoperative assessment requires
neurologic and cardiovascular evaluations. The type of stroke, the presence of
neurologic deficits, and the extent of residual impairment should be
determined. Thromboembolic strokes usually occur in patients with generalized
atherosclerosis. Most patients are elderly and have comorbid conditions, such
as hyper-tension, hyperlipidemia, and diabetes. Coexisting coronary artery disease
and renal impairment are common. Following nonhemorrhagic strokes or TIAs, many
patients are placed on long-term war-farin and/or antiplatelet therapy.
Management of antiplatelet therapy and antithrombotic therapy should be
reviewed by the anesthesia, primary care, and surgical teams to determine the
risk/benefit of the discontinuation or maintenance of such ther-apy
perioperatively. Other systemic diseases, such as diabetes, hypertension,
coronary artery disease, heart failure, and chronic obstructive lung disease
frequently manifest in the patient with cerebrovas-cular disease.
INTRAOPERATIVE MANAGEMENT
Patients may present for surgery following embolic, thrombotic,
and hemorrhagic strokes.
Management of the patient following
acute embolic stroke is directed toward the embolic source. Cardiac surgery is
performed to remove atrial myxomas. Systemic emboli can also be pro-duced from
endocarditic vegetations, as well as from degenerated heart valves and
intracardiac thrombus.
Patients with acute strokes secondary to carotid occlusive
disease present for carotid end-arterectomy and endovascular procedures. When
an awake carotid endarterectomy is undertaken, the patient serves as a monitor
of the adequacy of cerebral blood flow during application of vessel clamps to
facilitate the surgical repair. When gen-eral anesthesia is used
electroencephalography, evoked potentials, carotid stump pressure, cerebral
infrared spectroscopy, transcranial Doppler, and surgeon subjective sense of
collateral back flow are all used to estimate the adequacy of cerebral oxy-gen
delivery during cross clamp. When monitors or lack of appropriate patient
response indicate hypo-perfusion, the surgeon places a shunt to deliver blood
to the brain around the cross-clamped ves-sel. Even with adequate cerebral
blood flow, peri-operative stroke can occur during carotid surgery secondary to
emboli.
Management of patients following thrombotic or hemorrhagic
stroke for nonneurological surgery must be individualized. Cerebral
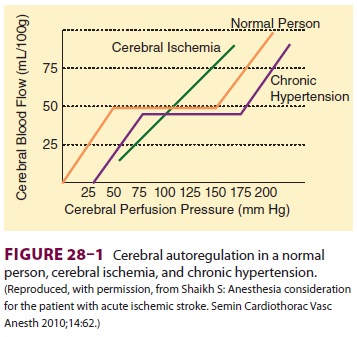
autoregulation of blood flow may fail, leaving flow directly dependent upon
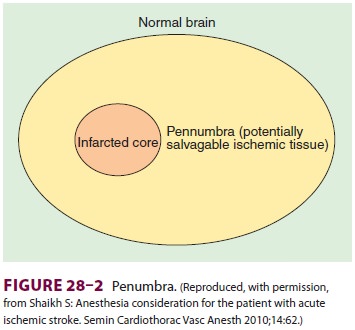
cerebral perfusion pressure (Figure28–1). The penumbra of potentially salvageable neurologic tis-sue
may therefore be very sensitive to injury from the effects of both hypotension
and hypertension (Figure28–2).
Patients taken to surgery following administra-tion of
thrombolytic therapy are at risk of cerebral hemorrhage, and tighter blood
pressure control may be indicated to mitigate the possibility of cerebral
bleeding.Patients with intracerebral hemorrhage or traumatic brain injury
undergo evacuation of
hematoma and decompressive craniectomy. These patients usually
require invasive arterial pressure monitoring to facilitate blood pressure
manage-ment in settings where cerebral autoregulation is likely deranged
(Figure 28–1). Hypertension is fre-quently treated with intravenous
vasodilators and β-blockers.
INTRACRANIAL MASS LESIONS
Patients with intracranial mass lesions
present to surgery with both malignant and nonmalignant lesions. Such patients
frequently present to their pri-mary care physicians with complaints of
headache, vision disturbance, or seizures. Radiologic studies confirm the
presence of a lesion, and initial treat-ment is aimed at decreasing cerebral
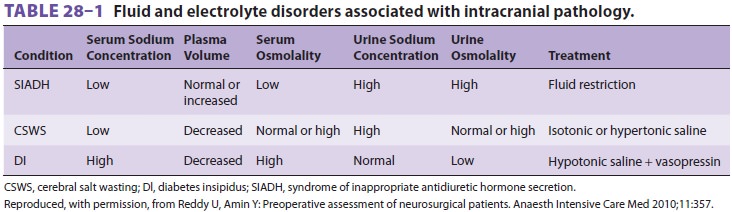
edema with dexamethasone. Electrolytes should be reviewed perioperatively in
all patients undergoing cranial surgery, as both hyponatremia and
hypernatre-mia can develop secondary to cerebral salt wast-ing, inappropriate antidiuretic
hormone secretion, or central diabetes insipidus ( Table28–1). Patients with altered mentation preoperatively
may likewise be dehydrated. Hyperglycemia secondary to steroid use is
frequently seen.
Related Topics