Chapter: Obstetrics and Gynecology: Amenorrhea and Abnormal Uterine Bleeding
Amenorrhea
AMENORRHEA
Amenorrhea
(the
absence of menstruation)
andA abnormal uterine bleeding are the most commongynecologic disorders
of reproductive-age women. Amenorrhea and abnormal uterine bleeding are
discussed as separate topics. However, the pathophysiol-ogy underlying
amenorrhea and abnormal uterine bleeding is often
the same.
If a young woman has never
menstruated by age 13 with-out secondary sexual development or by age 15 with
sec-ondary sexual development, she is classified as having primary amenorrhea. If a menstruating woman has notmenstruated for
3 to 6 months or for the duration of three typical menstrual cycles for the
patient with oligomenor-rhea, she is classified as having secondary amenorrhea. The designation of primary or secondary
amenorrhea has no bearing on the severity of the underlying disorder or on the
prognosis for restoring cyclic ovulation. Terms often confused with these
include oligomenorrhea, defined as a
reduction of the frequency of menses, with cycle lengths of greater than 35
days but less than 6 months, and hypomenorrhea,
defined as a reduction in the number of days or the amount of menstrual flow.
Amenorrhea not caused by
pregnancy occurs in 5% or less of all women during their menstrual lives.
Causes of Amenorrhea
When endocrine function along the
hypothalamic– pituitary–ovarian axis is disrupted or an abnormality devel-ops
in the genital outflow tract (obstruction of the uterus, cervix, or vagina or
scarring of the endometrium), menstru-ation ceases. Causes of amenorrhea are
divided into those arising from (1) pregnancy, (2) hypothalamic–pituitary
dys-function, (3) ovarian dysfunction, and (4) alteration of the genital
outflow tract.
PREGNANCY
Because
pregnancy is the most common cause of amenorrhea, it is essential to exclude
pregnancy in the evaluation of amenorrhea.
A history of breast fullness,
weight gain, and nausea suggest the diagnosis of pregnancy, which is confirmed
by a positive human chorionic gonadotropin (hCG) assay. It is important to rule
out pregnancy to allay the patient’s anxiety and to avoid unnecessary testing.
Also, some treatments for other causes of amenorrhea can be harmful to an
ongoing pregnancy. Lastly, the diag-nosis of ectopic pregnancy should be
entertained in the presence of abnormal menses and a positive preg-nancy test,
as this would necessitate medical or surgical intervention.
Box 35.1
Types of Abnormal Uterine Bleeding
Polymenorrhea—frequent
menstrual bleeding
(frequency,
21 days or less) Menorrhagia—prolonged or excessive uterine
bleeding
that occurs at regular intervals (the loss of 80 mL or more of blood that lasts
for more than 7 days)
Metrorrhagia—irregular
menstrual bleeding or bleeding between periods
Menometrorrhagia—frequent
menstrual bleeding that is excessive and irregular in amount and duration
American College of Obstetricians and
Gynecologists. Manage-ment of anovulatory bleeding. ACOG Practice Bulletin 14.
Washington, DC: American College of Obstetricians and Gyne-cologists; 2000.
HYPOTHALAMIC–PITUITARY DYSFUNCTION
Release of hypothalamic
gonadotropin-releasing hor-mone (GnRH) occurs in a pulsatile fashion, modulated
by catecholamine secretion from the central nervous system and by feedback of
sex steroids from the ovaries. When this pulsatile secretion of GnRH is
disrupted or altered, the anterior pituitary gland is not stimulated to secrete
follicle-stimulating hormone (FSH) and luteinizing hor-mone (LH). The result is
an absence of folliculogenesis despite estrogen production, no ovulation, and
lack of corpus luteum with its usual production of estrogen and progesterone.
Because of the lack of sex hormone produc-tion with no stimulation of the
endometrium, there is no menstruation.
Alterations in catecholamine
secretion and metabo-lism in sex steroid hormone feedback or an alteration of
blood flow through the hypothalamic–pituitary portal plexus can disrupt the
signaling process that leads to ovu-lation. This latter disruption can be
caused by tumors or infiltrative processes that impinge on the pituitary stalk
and alter blood flow.
The most common causes of
hypothalamic–pituitary dysfunction are presented in Box 35.2. Most hypothalamic–
pituitary amenorrhea is of functional origin and can be corrected by modifying
causal behavior, by stimulating gonadotropin secretion, or by giving exogenous
human menopausal gonadotropins.
The physician cannot
differentiate hypothalamic– pituitary causes of amenorrhea from ovarian or
genital outflow causes by medical history or even physical exam-ination alone.
However, there are some clues in the med-ical history and physical examination
that would suggest a hypothalamic–pituitary etiology for amenorrhea. A his tory
of any condition listed in Box 35.2 should cause the physician to consider
hypothalamic–pituitary dysfunction.
Box 35.2
Causes of Hypothalamic–Pituitary Amenorrhea
Functional Causes
Weight
loss
Excessive
exercise
Obesity
Drug-Induced Causes
Marijuana
Psychoactive
drugs, including antidepressants
Neoplastic Causes
Prolactin-secreting
pituitary adenomas Craniopharyngioma Hypothalamic hamartoma
Psychogenic Causes
Chronic
anxiety
Pseudocyesis
Anorexia
nervosa
Other Causes
Head
injury
Chronic
medical illness
The definitive method to identify hypothalamic–pituitary dysfunction is to measure FSH, LH, and prolactin levels in the blood. In these conditions, FSH and LH levels are in the low range. The prolactin level is normal in most conditions, but is elevated in prolactin-secreting pituitary adenomas.
OVARIAN DYSFUNCTION
In ovarian failure, the ovarian
follicles are either exhausted or are resistant to stimulation by pituitary FSH
and LH. Asthe ovaries cease functioning,
blood concentrations of FSH and LH increase. Women with ovarian failure
experience thesymptoms and signs of estrogen deficiency. A summary of causes is
presented in Box 35.3.
ALTERATION OF THE GENITAL OUTFLOW TRACT
Obstruction of the genital
outflow tract prevents overt menstrual bleeding even if ovulation occurs.
Box 35.3
Causes of Ovarian Failure
Chromosomal Causes
Turner
syndrome (45,X gonadal dysgenesis)
X
chromosome long-arm deletion (46,XX q5)
Other Causes
Gonadotropin-resistant
ovary syndrome (Savage syndrome)
Premature
natural menopause
Autoimmune
ovarian failure (Blizzard syndrome)
Most
cases ofoutflow obstruction result from congenital abnormalities in the development and canalization of the müllerian
ducts. Imperforatehymen and no uterus or vagina are the most
common anomalies that result in primary amenorrhea. Surgical cor-rection of an
imperforate hymen allows for menstruation and fertility. Less-commonly
encountered anomalies, such as a transverse vaginal septum, are more difficult
to correct, and even with attempted surgical correction, menstruation and
fertility are often not restored.
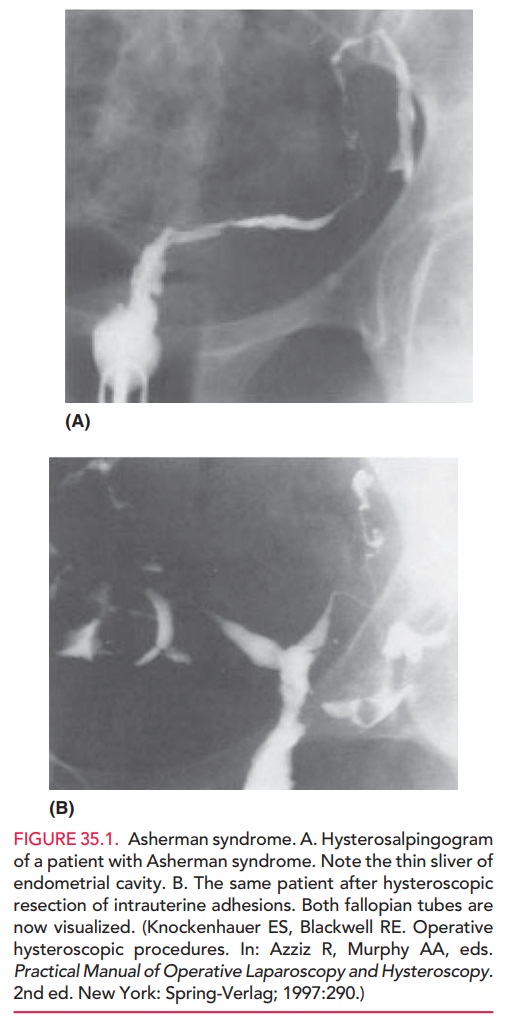
Scarring
of the uterine cavity (Asherman syndrome) is the most frequent anatomic cause
of secondary amenorrhea (Fig. 35.1). Women who undergo
dilation and curettage (D&C) for retained products of pregnancy (especially
when infection is present) are at risk for developing scar-ring of the
endometrium. Cases of mild scarring can be corrected by surgical lysis of the
adhesions performed by hysteroscopy and D&C. However, severe cases are
often refractory to therapy. Estrogen therapy should be added to the surgical
treatment postoperatively to stimulate endometrial regeneration of the denuded
areas. In some cases, a balloon or intrauterine (contraceptive) device may be
placed in the uterine cavity to help keep the uterine walls apart during
healing.
Treatment of Amenorrhea
The first step is to establish a cause for the amenorrhea. The progesterone “challenge test” is commonly used to deter-mine whether or not the patient has adequate estrogen, a competent endometrium, and a patent outflow tract. An injection of 100 mg of progesterone in oil or a 5-day to 14-day course of oral medroxyprogesterone acetate or micronized progesterone is expected to induce proges-terone withdrawal bleeding within a few days after com-pleting the oral course. If bleeding does occur, the patient is likely to be anovulatory or oligo-ovulatory. If withdrawal bleeding does not occur, the patient may be hypoestro-genic or have an anatomic condition such as Asherman syndrome or outflow tract obstruction.
Hyperprolactinemia
associated with some pituitary adeno-mas (or other medical conditions) results
in amenorrhea and galactorrhea (a
milky discharge from the breast).Approxi-mately 80% of all
pituitary tumors secrete prolactin, causing galactorrhea, and these patients
are treated with either cabergoline (Dostinex) or the dopamine agonist
bromocriptine (Parlodel). In approximately 5% of patients with hyperprolactinemia
and galactorrhea, the underlying etiology is hypothyroidism. A low serum
thy-roxine (T4) level eliminates negative feedback signaling to the
hypothalamic–pituitary axis. As a result, TRH (thyrotropin-releasing hormone)
levels increase. Positive feedback signaling that stimulates dopamine secretion
is also absent, causing a decrease in dopamine levels. Elevated TRH stimulates
release of prolactin from the pituitary gland. The reduced dopamine secretion
results in elevated levels of TSH (thyroid-stimulating hormone) and prolactin.
In patients who desire pregnancy,
ovulation can be induced through the use of clomiphene citrate, human
menopausal gonadotropins, pulsatile GnRH, or aromatase inhibitors. In patients
who are oligo-ovulatory or anovula-tory (polycystic ovary syndrome), ovulation
can usually be induced with clomiphene citrate. In patients with
hypogo-nadotropic hypogonadism, ovulation can be induced with pulsatile GnRH or
human menopausal gonadotropins. Women with genital tract obstruction require
surgery to create a vagina or to restore genital tract integrity. Menstruation
will never be established if the uterus is absent. Women with premature
menopause may require exogenous estrogen therapy.



Related Topics