Chapter: Essential Clinical Immunology: Immunological Aspects of Renal Disease
Acute Poststreptococcal Glomerulonephritis
ACUTE POSTSTREPTOCOCCAL GLOMERULONEPHRITIS
APSGN is an immune complex disease that follows
pharyngitis and skin infec-tions from nephritogenic strains of group A
streptococci. Most affected patients are children in developing countries.
About ten days after streptococcal pharyngitis or twenty-one days after
streptococcal skin infection (impetigo), susceptible indi-viduals may develop
microscopic hema-turia or full-blown acute nephritis (red cell casts,
proteinuria, edema, hyperten-sion, and acute renal failure). Diagnosis is made
by history of throat or skin infec-tion. In addition to throat or wound
cul-ture, serology for ASO, anti-DNAase B and antihyaluronidase may be obtained
to confirm the infection. Circulating immune complexes appear early in the
course of the disease and are associated with hypo-complementemia. The majority
of affected children recover and kidney biopsy is not always indicated. When
kidney biopsy is obtained, prominent findings are compat-ible with a diffuse
proliferative nephritis picture (hypercellularity and marked neu-trophilic
infiltration). On immunofluo-rescence, there is granular deposition of
complements and IgG in the glomerular tuft. On electron microscopy, subepithe-lial
dense deposits, or “humps,” are con-sidered hallmarks of PSGN. Damage to the
epithelial cells appears to be respon-sible for the proteinuria noted in this
renal disease. Subendothelial deposits are also seen, which are primarily
responsible for complement activation and influx of inflammatory cells.
Over the past two decades, many attempts to
identify the nature of the strep-tococcal antigen in the immune complex have
been made by several investigators.
Two prominent proteins have been impli-cated in the
pathogenesis of APSGN: strep-tokinase (Ska) and SPEB. Ska is produced by all
strains of group A streptococci, but SPEB appears to be secreted in abundance
in nephritogenic strains causing APSGN. Ska cleaves cell-surface-bound plas-minogen
to active plasmin. Plasmin then cleaves fibrin into degradation products, which
are also seen in inflammatory pro-cesses. Bound plasmin apparently cannot be
inhibited by endogenous antiproteases, thereby causing unopposed formation of
fibrin degradation products. The role of streptokinase, however, was in doubt
when immunostaining studies on kidney biopsy specimens using rabbit anti-Ska
antibody failed to detect this protein.
SPEB, or streptococcal proteinase, how-ever,
behaves as a cysteine protease, which can activate endothelial metalloprotease
and cause tissue destruction. Although structurally unrelated to streptokinase,
SPEB can also bind plasmin to form a com-plex that can activate the complement
cas-cade system. SPEB and other streptococcal pyrogenic exotoxins (SPEA and
SPEC) are considered superantigens, which can stimulate T-cell proliferation.
The result is excessive production of cytokines, which mediate inflammation and
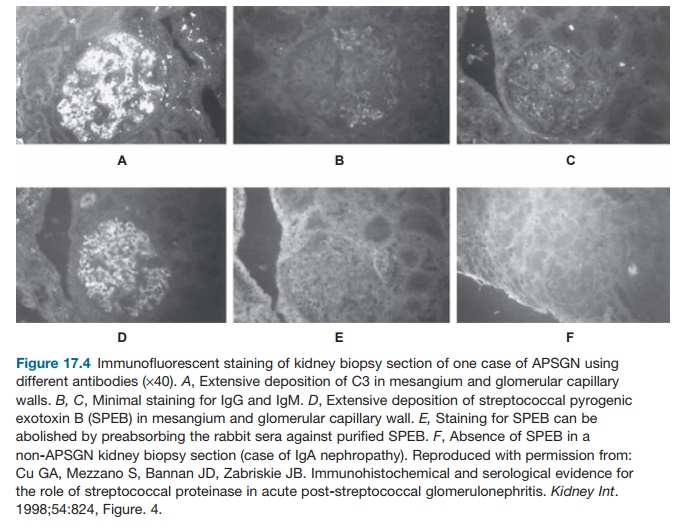
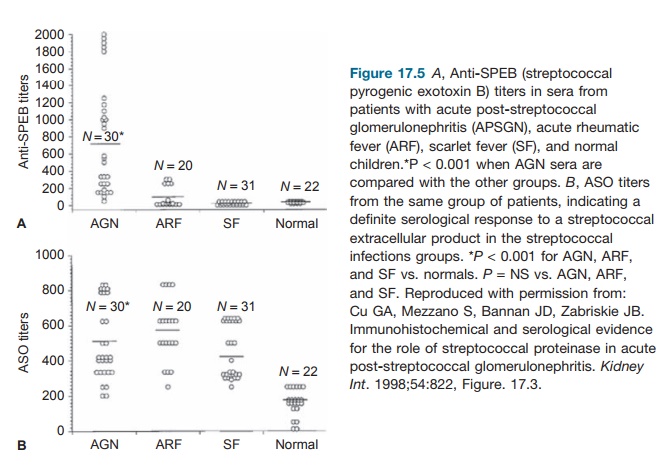
tissue injury. As demonstrated by Cu and coworkers, immu-nohistochemical and
serologic studies (see Figures 17.4 and 17.5) on kidney biopsy specimens and
sera from affected patients have shown strong and specific reactivity to SPEB
in the biopsies and in the sera of these patients. This suggests a significant
role of this protein in the pathogenesis of APSGN. This observation was
recently
APSGN follows a relatively benign course. The
majority of cases will recover spontaneously with only less than 1 percent of
children and slightly higher percentages of adults needing long-term dialysis
treatment. Hematuria may resolve in three to six months. Proteinuria may persist
for years, which may be later asso-ciated with hypertension and renal
insufficiency.
Related Topics