Chapter: Medical Immunology: Infections and Immunity
Abnormal Consequences of the Immune Response
ABNORMAL CONSEQUENCES OF THE IMMUNE RESPONSE
In the vast majority of situations the immune response has a protective effect that allows the organism to recover from infection without major illness and without long-term sequelae. However, there are well-known examples of deleterious effects triggered by an exaggerated or misdirected immune response.
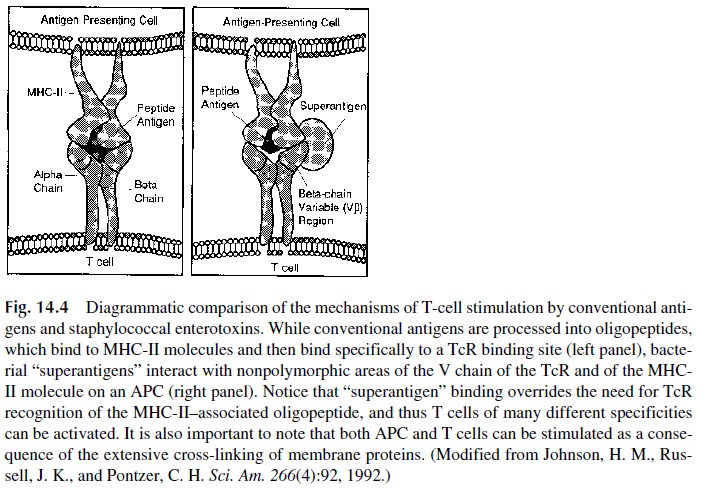
The Activation of T Lymphocytes by Bacterial “Superantigens”
A variety of bacterial exotoxins, such as staphylococcal enterotoxins-A and -B (SE-A and SE-B), staphylococcal toxic shock syndrome toxin-1 (TSST-1), exfoliating toxin, and streptococcal exotoxin A, as well as other unrelated bacterial proteins (such as streptococ-cal M proteins), have been characterized as “superantigens.”
Superantigens are defined by their ability to stimulate T cells without being pro-cessed. The stimulation of T cells is polyclonal; thus the designation of superantigen is a misnomer, but it has gained popularity and is widely used in the literature. The better stud-ied superantigens are the staphylococcal enterotoxins, which are potent polyclonal activa-tors of murine and human T lymphocytes, inducing T-cell proliferation and cytokine re-lease. TSST-1 also appears to activate monocytes and is a potent B-cell mitogen, inducing B-cell proliferation and differentiation.
The stimulatory effects of superantigens are a consequence of the direct and si-multaneous binding to the nonpolymorphic area of class II MHC on professional acces-sory cells (macrophages and related cells) and to the Vβ chain of the α/β TcR (Fig. 14.4). For example, staphylococcal enterotoxins bind exclusively to specific subfamilies of Vβ chains that are expressed only by certain individuals. When expressed, these Vβ chain regions can be found on 2–20% of a positive individual’s T cells, and the cross-linking of the TcR2 and of the APC by the enterotoxin activates all T cells (both CD4 and CD8+ ) expressing the specific Vβ region recognized by the enterotoxin. The mas-sive T-cell activation induced by superantigens results in the release of large amounts of IL-2, interferon-γ , lymphotoxin-α (LTα ), and TNF. After the initial burst of cytokine re-lease, the stimulated T cells either undergo apoptosis or become anergic. This effect could severely disturb the ability of the immune system to adequately respond to bacte-ria releasing superantigens.
Patients infected by bacteria able to release large amounts of superantigens (e.g., S.aureus releasing enterotoxins or TSST-1 and group A Streptococcus releasing exotoxin A)may develop septic shock as a consequence of the systemic effects of these cytokines. These systemic effects include fever, endothelial damage, profound hypotension, dissemi-nated intravascular coagulation, multiorgan failure, and death.
Infection as a Consequence of the Uptake of Antigen-Antibody Complexes
The immune response, in some cases, facilitates the access of infectious agents to cells in which they will be able to proliferate. For example, macrophages are often infected by in-tracellular organisms that are ingested as a consequence of opsonization. Babesia rodhaini, a bovine intraerythrocytic parasite, penetrates the host’s cells after it has bound comple-ment, particularly C3. Absorption of C3b-containing circulating antigen-antibody com-plexes to the CR1 expressed by red cells allows the parasite to gain access to the red cell, which becomes its permanent location. Epstein-Barr virus (EBV) normally infects B cells through a CR-2–related protein. However, if dimeric IgA reactive with the virus is pro-duced and released into the mucosal secretions, the resulting IgA-EBV complex is able to infect mucosal cells through the poly-Ig receptor, which binds dimeric IgA . Mucosal infection in the nasopharynx can eventually acquire malignant characteristics.
Postinfectious Tissue Damage
Several examples of the pathogenic role of an anti-infectious immune response have been characterized. The following are some of the best known examples:
Immune complex–induced inflammation.Antigen-antibody complexes, if formed inlarge amounts, can cause disease by being trapped in different capillary net-works and leading to inflammation. The clinical expression of immune com-plex–related inflammation depends on the localization of the trapped com-plexes: vasculitis and purpura, when the skin is predominantly affected, glomerulonephritis if trapping takes place on the glomerular capillaries, or arthritis when the joints are affected . Viruses are often in-volved in the formation of circulating antigen-antibody complexes.
Immune destruction of infected cells and tissues.An immune response directedagainst an infectious agent may be the main cause of damage to the infected tis-sue. For example, in subacute sclerosing panencephalitis, a degenerative dis-ease of the nervous system associated with persistent infection with the measles virus, the response against viral epitopes expressed in infected neurons is believed to be the primary mechanism of disease. Also, in some forms of chronic active hepatitis the immune response directed against viral epitopes expressed by infected hepatocytes seems to cause more tissue damage than the infection itself.
Cross-reactions with tissue antigens.These have been proposed as the basis for theassociation of streptococcal infections with rheumatic carditis and glomeru-lonephritis. Antibodies to type 1 streptococcal M protein cross-react with epi-topes of myocardium and kidney mesangial cells and cause inflammatory changes in the heart and glomeruli, respectively.
Related Topics