Chapter: Clinical Cases in Anesthesia : Respiratory Failure
What are the four primary causes of hypoxemia and how are they distin-guished? Which is most likely in this patient, and how would you treat it?
The arterial blood gas (ABG) is pH 7.35, PCO2
37 torr, and PO2 54 torr on control-mode ventilation (CMV) with a
rate of 12 breaths per minute, a tidal volume of 650 mL, an FiO2 of
0.5, and a positive end-expiratory pressure (PEEP) of 5 cm H2O. Peak
inspira-tory pressures (PIP) are 26 cm H2O. What are the four
primary causes of hypoxemia and how are they distin-guished? Which is most
likely in this patient, and how would you treat it?
The four primary causes of hypoxemia are
hypoventilation, shunt, ventilation/perfusion (V/Q) mismatch, and diffusion
impairment.
Hypoventilation
Hypoventilation means that there is a reduction
in fresh gas flow to the alveoli such as is seen in a circuit disconnect. The
hallmark feature is an increased arterial PCO2. Two basic equations
relate to this condition:
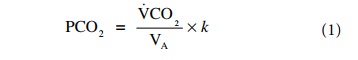
where VCO2 is the CO2
produced, VA is the alveolar venti-lation, and k is a constant equal to 0.863. This means that if the alveolar
ventilation is halved, the PCO2 doubles and vice versa.
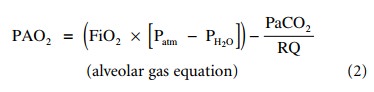
where PAO2 is the alveolar partial
pressure of oxygen, Patm is the atmospheric pressure, usually 760
mmHg at sea level (but in the low 600s in Denver, Colorado), PH2O is
the saturated pressure of water at 37°C (47 mmHg), and RQ is the respiratory
quotient, between 0.7 and 1.0 depending on the carbohydrate/lipid ratio in the
diet (usually a value of 0.8 is used). This equation predicts that even severe
hypoventilation may be overcome by administration of high FiO2.
Thus, postoperative patients with respiratory depres-sion receiving
supplemental oxygen by mask are more likely to suffer the effects of
respiratory acidosis than hypoxemia.
Shunt
A shunt exists when blood passes from the
venous circula-tion to the arterial circulation without exposure to ventilated
areas of the lung. The primary feature of a significant shunt is the failure of
the PO2 to rise to normal values with the administration of oxygen.
The PCO2 is not raised because the central chemoreceptor sensitivity
to a rise in the PCO2 and hypoxemia will both act as a stimulus for
greater ven-tilation. The degree of shunting is calculated by the shunt
equation:
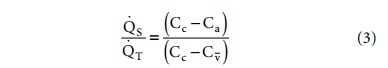
where .QS and .QT refer to
the shunt and total pulmonary blood flows, respectively, and Cc, Ca,
and Cv refer to the oxygen content of pulmonary end-capillary,
arterial and mixed venous blood, respectively. Blood oxygen content is
calculated as follows:
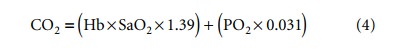
Where Hb is the hemoglobin concentration in
mg/dL, SaO2 the oxygen saturation of hemoglobin, and PO2
the partial pressure of oxygen. Hemoglobin concentration and oxygen saturation
are thus the main determinants of blood oxygen content, with the partial
pressure of oxygen playing a comparatively small role, except in special
situations such as severe anemia or hyperbaric oxygen therapy.
Ventilation/perfusion mismatch
Ventilation/perfusion mismatch is the most
common cause of hypoxemia and refers to the inefficient and incomplete transfer
of gas because of mismatching of blood flow to ventilation. In practice,
ventilation/perfusion mismatching is said to occur when hypoventilation, shunt,
and diffusion defect (to be discussed next) are excluded from the differential
diagnosis by studying the PCO2 to rule out hypoventilation, the
response to oxygen adminis-tration to rule out shunt, and a lack of history and
radi-ographic findings to suggest diffusion impairment.
Diffusion impairment
Diffusion impairment implies that there is an
incom-plete equilibrium between the gas in the alveolus and the capillary blood
because of an abnormality in the normally thin-walled and easily crossed
alveolar–capillary barrier. This may occur in a variety of chronic lung
diseases such as interstitial fibrosis, asbestosis, and sarcoidosis. Although
diffusion capacity may be measured in a pulmonary func-tion laboratory setting
using carbon monoxide, bedside quantification of diffusion-limited hypoxemia
using stan-dard laboratory tests is impossible.
In the case of this patient, the alveolar to
arterial (A–a) gradient is 256, while the PaO2/FiO2 ratio
is 108. The most likely cause of hypoxemia is V/Q mismatch. The PCO2
is normal, virtually excluding hypoventilation. Shunt can be excluded if an
increase in PO2 is seen after increasing the FiO2.
Several etiologies could account for the V/Q mismatch: pre-existing lung
disease, fluid overload after an operation with major fluid shifts, aspiration
pneumonitis, and atelec-tasis after general anesthesia with neuromuscular
blockade and one-lung ventilation. Increasing the FiO2 will help
raise the PO2, but an FiO2 greater than 0.6 for prolonged
periods of time should be avoided, if possible, because of the risk of oxygen
toxicity. Increasing the PEEP, as hemodynamically tolerated, can recruit
additional alveoli, reduce the shunt component, and redistribute lung water to
areas that do not participate in gas exchange. Diuresis is indicated if fluid
overload is clinically suspected or confirmed by invasive monitoring.
Related Topics