Chapter: Microbiology and Immunology: Bacterial Genetics
Transfer of DNA Between Bacterial Cells
Transfer of DNA Between Bacterial Cells
The genetic information can be transferred from one bacterium to another. There are three general methods for genetic exchange in bacteria: (a) transformation, (b) transduction, and (c) conjugation.
Transformation
Transformation is a process of the transfer of DNA itself
fromone bacterium to another. This may occur either in nature or in a
laboratory. In nature, DNA is released from a bacterium by lysis, which may be
taken up by recipient bacterium that must be competent. This natural process of
transfer of genetic material appears to play no role in disease. In laboratory
condi-tions, DNA may be extracted from one type of bacterium and introduced
into genetically different bacteria. The cell walls of bacteria in vitro are made more permeable for DNA
uptake by using substances, such as calcium chloride.
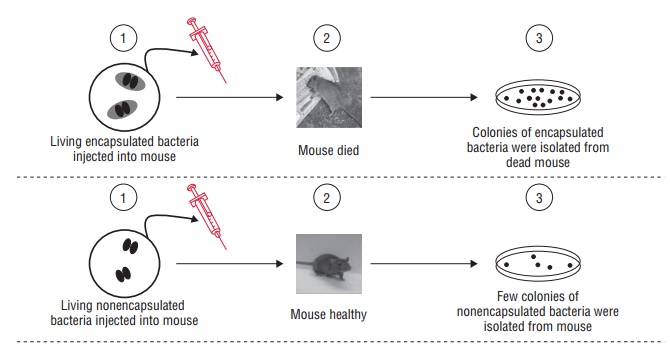
Griffith (1922) in his classical experiment on mice demon-strated
that neither of the mice died when injected separately with a live,
noncapsulated Pneumococcus (nonvirulent) and heat-killed, capsulated
Pneumococcus (nonvirulent), but the mice died when they were injected with a mixture
of both these strains. From the dead mice, he could isolate live, capsulated
pneumococci, which were virulent. He demon-strated that some factor in
heat-killed, capsulated pneumo-cocci had transferred the material for capsule
synthesis in the noncapsulated strains of the bacteria, making them virulent
(Fig. 7-1).
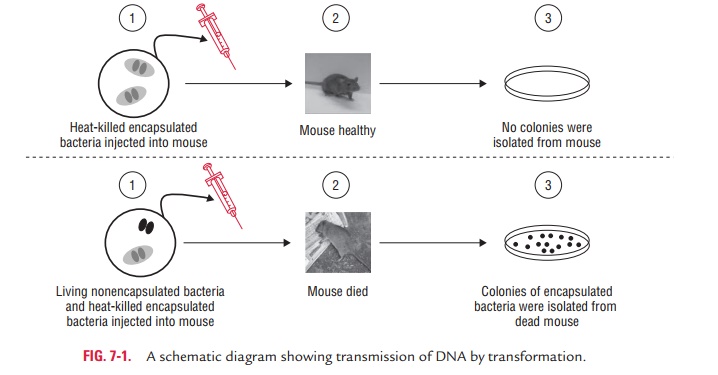
McLeod and McCarthy in 1944 demonstrated that DNA extracted from
encapsulated, smooth pneumococci could transform nonencapsulated, rough
pneumococci into capsulated, smooth organisms. They demonstrated the
transforming principle of DNA. The experimental use of transformation was the
first experiment to reveal important information about DNA and was the first
example of genetic exchange in bacteria.
Another bacterium where
transformation is observed
is Haemophilus influenzae.
Transduction
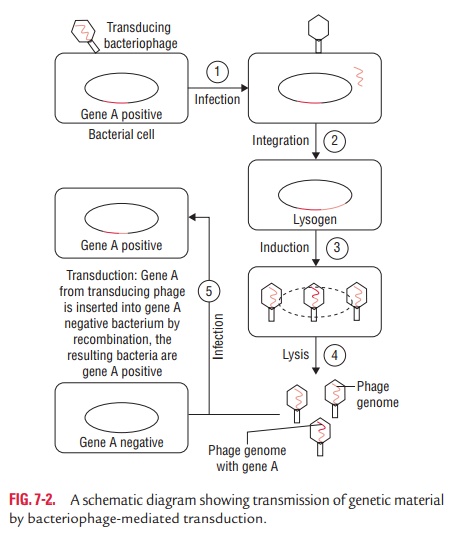
The transfer of a portion of DNA from one bacterium to another
mediated by a bacteriophage is known as transduc-tion. During replication of
virus within the cell, a piece ofbacterial DNA is incorporated into the
bacteriophage and is carried into the recipient bacterium at the time of
infection. The phage DNA within the recipient bacterial cell integrates into
the cell DNA during a process called lysogenic conversion. The process of
lysogenic conversion confers a new property to the bacterial cell; for example,
by lysogenic conversion non-pathogenic bacteria can become pathogenic.
Bacteriophages encode diphtheria toxin, botulinum toxin, cholera toxin, and
erythrogenic toxin and can be transferred from one bacte-rium to another by
transduction (Fig. 7-2). Transduction is of two types: (a) generalized transduction and (b) specialized transduction.
◗ Generalized transduction
This occurs when a small fraction of the phage virions produced
during lytic cycle are aberrant and contain a random fragment of the bacterial
genome instead of phage DNA. Each individ-ual transducing phage carries a
different set of closely linked genes, representing a small segment of the
bacterial genome. Transduction mediated by populations of such phages is called
generalized
transduction. Each part of the bacterial genome hasapproximately the
same probability of being transferred from donor to recipient bacteria.
Generalized transduction involves any segment of the donor DNA at
random. This occurs because the cell DNA is fragmented after such infection and
pieces of same DNA, the same size as viral DNA, are incorporated into the
bac-terial DNA. This occurs at a frequency of about 1 in every 1000 viruses.
Generalized transduction may be complete or abortive:
Complete transduction is characterized by
production ofstable recombinants that inherit donor genes and retain the ability
to express them.
Abortive transduction refers to the transient
expressionof one or more donor genes without formation of recombi-nant progeny.
The donor DNA fragment does not replicate in abortive transduction, and only
one bacterium contains the donor DNA fragment among the progeny of the
origi-nal transductant. The donor gene products become progres-sively diluted
in all other progeny after each generation of bacterial growth until the donor
phenotype can no longer be expressed.
On selective medium, abortive transductants produce minute colonies
that can be distinguished easily from colonies of stable transductants. The
frequency of abortive transduction is typically one to two times more than the
frequency of generalized trans-duction. This indicates that most cells infected
by generalized transducing phages do not produce recombinant progeny.
◗ Specialized transduction
Specialized transduction results from lysogenization of the
recipient bacterium by the specialized transducing phage and expression of the
donor genes. Specialized transducing phages
The specialized transducing phages are rare recombinants that lack
part of the normal phage genome. They contain part of the bacterial chromosome
present adjacent to the site of pro-phage attachment. Many specialized
transducing phages are defective. They cannot complete the lytic cycle of phage
growth in infected cells unless helper phages are present to provide missing
phage functions.
Specialized transduction differs from generalized transduc-tion in
many ways. The former is mediated only by specific temperate phages and only a
few specific donor genes can be transferred to recipient bacteria.
Conjugation
Conjugation is a process of transfer of DNA from the donor
bacterium to the recipient bacterium during the mating of two bacterial cells.
In conjugation, direct contact between the donor and recipient bacteria leads
to formation of a cytoplasmic bridge between them and transfer of part or all
of the donor genome to the recipient (Fig. 7-3). Conjugation takes place
between two closely related species and occurs mostly in Gram-negative
bacteria. Conjugation also occurs in Gram-positive bacteria.
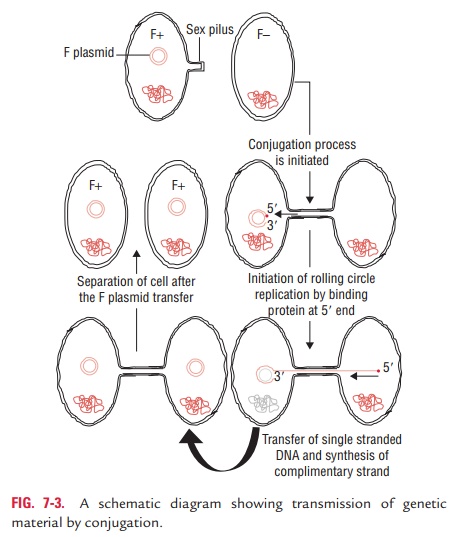
Donor ability of bacteria is determined by specific conju-gative
plasmids called fertility (F1) plasmids or sex plasmids. The F plasmid controls the mating
process of bacteria. Pilus is the most important protein that forms the sex
pilus or conjugation tube. The sex pilus produces a bridge between conjugating
cells in Gram-negative bacteria. Mating occurs between the donor male bacterium
carrying the F factor (F1) and the recipient female bacterium that does not contain F
fac-tor (F2). It begins when the pilus of F1 bacterium attaches to a receptor on the
surface of a female (F2) bacterium. The cells are then brought into direct contact by the
link in the pilus. This is followed by an enzymatic cleavage of the F factor
DNA in which one strand of bacterial DNA is transferred into the recipient cell
through the conjugation bridge. The synthesis of the complementary strand to
form a double-stranded F-factor plasmid in both the donor and recipient cells
completes the process of conjugation. The recipient cell becomes F1 male that is capable of
transmitting the plasmid to other F2 cells.
High-frequency recombination
(Hfr): Long length of DNAcan be transferred by process of conjugation. Hfr
strain is a type of F+ cells that have an F plasmid integrated into the
bacterial DNA. Hence they acquire the capability of transferring the
A whole chromosome can be transferred
if it is integrated with F plasmid. In this process, the single strand of DNA
that enters the recipient F2 cell contains a part of the F factor at one end, followed by the
bacterial chromosome, and then by the remainder of the F factor. The bacterial
genes adjacent to the leading piece of F factor are the most frequently
transferred. The newly acquired DNA recombines with the recipient DNA and
becomes an integral component of genetic material. The complete transfer of the
bacterial DNA is usually completed in approximately 100 minutes.
In matings between F+ and F− bacteria, only the F plasmid is transferred
with high efficiency to recipients. Chromosomal genes are transferred with very
low efficiency, which is medi-ated by the spontaneous Hfr mutants in F1 populations. In matings
between Hfr and F2 strains, the segment of the F plas-mid containing the tra region
is transferred last, after the entire bacterial chromosome has been
transferred. Most recombi-nants produced after matings between Hfr and F2 cells fail to inherit the
entire set of F-plasmid genes and are phenotypi-cally F2. In matings between F1 and F2 strains, the F plasmid
spreads rapidly throughout the bacterial population and most recombinants are F1.
Conjugation also occurs in Gram-positive bacteria. Gram-positive
donor bacteria produce adhesions that cause them to aggregate with recipient
cells, but sex pili are not involved. In some Streptococcus spp., recipient bacteria produce extracellular sex
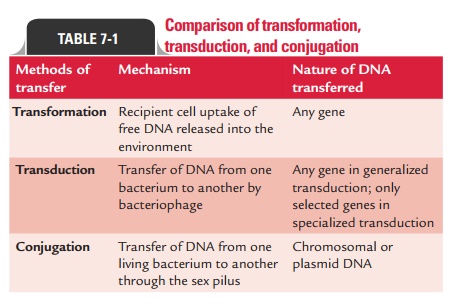
pheromones that facilitate conjugation. Table 7-1 shows a comparison of
transformation, transduction, and conjugation.
Recombination
After the DNA is transferred from one donor bacterium to the
recipient through transformation, transduction, or conjugation, it combines
with the chromosome of the bacterium by a process called recombination.
Recombination is of two types: homologous and nonho-mologous.
Homologous recombination takes place between two pieces of DNA showing
extensive homologous regions. This results in pairing up and exchange of pieces
by the pro-cesses of breakage and reunion. The nonhomologous recom-bination
takes place between two pieces of DNA showing little or no homology.
Related Topics